Professional Documents
Culture Documents
Retro Synthesis
Uploaded by
Pramod AryalCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Retro Synthesis
Uploaded by
Pramod AryalCopyright:
Available Formats
1 / 20
Novel Approach to Retrosynthesis Automatic Generation of Transform Libraries
V. Eigner-Pitto, J. Eiblmaier, H. Kraut, H. Saller and P. Loew.
InfoChem GmbH, Landsberger Strasse 408, Munich, 81241, Germany
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
2 / 20
Why Retrosynthesis?
Is there a shorter synthesis path to my target? How can I synthesize this compound?
Can we increase the yield of this process by choosing another synthesis strategy?
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
3 / 20
Retrosynthesis: Expected Features
Main Challenge: Automatic generation of chemically meaningful
precursors and their reasonable validation 1. Evaluation of Alternative Synthesis Pathways
Sorting criteria are necessary for broad representations with many expanding paths: Yield Type of transformation (e.g. ring-closures or construction reactions preferred, etc.)
2. Support of Multistep Synthesis
Necessary to guide the synthesis from the target to all existing precursors that can be obtained from commercially available substances
3.
Organized Visualization
Allows a broad view over the different strategies. A tree view, i.e., gives an organized representation of the synthesis paths to the target
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
4 / 20
Synthesis-Planning Tools
1. Use of known compounds and published reactions
i.e. MDL (CrossFire Commander), Symyx (Reaction Pathfinder), InfoChem (STS) ! Handle multistep synthesis ! Order in synthesis paths - Sorting criteria ! Shows synthesis options in your database (tree representation) " Structure and corresponding reaction(s) must be in database
2.
Intellectual retrosynthesis systems
! New molecules (not in the database) ! Alternative routes to known molecules ! Reaction transformation / Synthesis strategy to specify ! Transform based
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
5 / 20
Intellectual Retrosynthesis Approaches
2a. Transforms compiled manually from literature
! accurate, highly specific " tedious, expensive, restrictive
2b.
Transforms based on enumerated bond changes
! fully automated " unspecific, combinatorial explosion
Examples:
LHASA, SECS / CASP ... (Corey, Wipke) WODCA, EROS ... (Gasteiger) SYNGEN ... (Hendrickson) AIPHOS ... (Sasaki) .....
ICIC Nmes, October 22-25, 2006
InfoChem GmbH 2006
Dr. V. Eigner-Pitto
6 / 20
RetroSynthesis Concept Overview
1. Pre-processing
Template (2 levels):
1 14 2 4 7 13 8
11
12
R1
10 6 3
R2
9
Automatic transform extraction (ICMAP/CLASSIFY)
Transform:
R e m o v e b o n d 1 -2 M a k e n e w s in g le b o n d b e t w e e n a to m s 3 a n d 4
Decrease bond order of double bond 2=4 by 1. Make new single bond between atoms 2 and 6. Add group to atom 3: -OH
R e m o v e b o n d 1 -3 Remove atoms M ake ne w s in bond g le between b ond b e t w3 eand e n 6. atom s 3 a nd 5
Reaction database
Transform Library
2. RetroSynthesis
O H H O
e. g. SPRESI proprietary databases commercial databases
RetroSyn
Lookup stored examples
Target molecule
Reaction 1: Reaction 2:
Precursor(s) 1 Precursor(s) 2
Transform x
O
Transform x
H H O
Transform y
Transform y
...
Reaction n:
Precursor(s) n
Transform z
Target molecule
Transform z
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
7 / 20
Reaction Database Requirements
1. Correct Reaction Mapping
Description of the transformation occuring during the reaction Determination of reaction centers
2.
Reaction Classification
Automatic identification of the structural environment around the reacting centers Determination of unique identifiers (ClassCodes)
O O + N N
Reaction Type Amination:
.3. .3.
O
.2. .1.
O
.5.
+ O
.4.
N
.1.
.5. .2.
F F F
O O O + N
F F F
O N
N
.4.
Cl
Cl
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
8 / 20
Definition of Template (Retron)
SPRESI Reaction Reg-No.: 3179546
Kita Yasuyuki, Yoshida Yutaka, Mihara Sachiko, Fang Dai-Fei, Higuchi Kazuhiro, Furukawa Akihiro, Fujioka Hiromichi Efficient pinacol rearrangement mediated by trimethyl orthoformate Tetrahedron Lett., 38 (1997) 48, 8315-8318
CH3 HO OH O
CH3
CH3 O O
CH3 O
CH3
Template (level 2)
Template (level 3)
ICIC Nmes, October 22-25, 2006
Template (level 4)
InfoChem GmbH 2006
Dr. V. Eigner-Pitto
9 / 20
Definition of Transform
Template:
1 2 3 5
CH3
8
O
4
Indications:
Remove bond between atom 5 and 6 Decrease bond order of double bond 3=4 by 1 Make new single bond between atoms 3 and 6 Add group to atom 5: -OH
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
10 / 20
Pre-processing Step (Transform Library Building)
Reaction Database Transform Library
O O + N
O N
11
12
R1
10
R2
9 6
O O + N
O N
Transform Generator
O
Template (2 levels):
1 14 2
3 7 4
5 8 13
R em ove bo nd 1-3 M a k e n e w s in g le b o n d b e t w e e n a t o m s 3 a n d 5
Transform:
b on d 1-2 M a k e ne w s Decrease in g le b bond o nd order b eof tw double e e n bond a t o2=4 m sby 31.a n d 4
Make new single bond between atoms 2 and 6. Add group to atom 3: -OH
R e3m ov e Remove bond between atoms and 6.
F F F
O O O + N
F F F
O N
Cl
Cl
Generation of substructure index
Template Index
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
11 / 20
Templates Search
Rated Templates
Template Index Target Molecule
ICFSE
Rating C Rating B Rating A
Strategy Parameters
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
12 / 20
Strategy Parameters and Precursors Rating
Strategy parameters for precursor-rating consider the following criteria:
formation of a new carbon-carbon bond bond change within a functional group formation of a new ring/aromatic bond formation of a new bond between two symmetric fragments formation of a new bond near the center of the molecule number of generated precursors
These criteria are weighted depending on the chosen configuration:
sets of pre-defined strategy parameters are available sets may be defined from user
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
13 / 20
Precursors Generation from Target
Rated Templates Target Precursors
RetroSyn
Transform Transform
Transform
Rating c Rating B Rating A
Transform Library
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
14 / 20
Multistep Synthesis
Best First Search
Rating A
Precursors Step 1
O Br O O
Precursors Step 2
Rating AA
Mg
Br
Catalog
OH
Target
O
OH
O
O
O
+
O
Rating AB
NH2
OH
Limits:
steps precursors
Rating B
OH
O
N N
Rating BA
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
15 / 20
Prototype: Query Form
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
16 / 20
Reaction Databases Used in Prototype
SPRESI Synthesis Name Reactions EROS SOS CAC
2.5 mio reactions from SPRESI reactions with 50% minimum yield 140.000 reactions from SPRESI all examples for a large set of Name Reactions 68.000 reactions from J. Wileys Encylopedia of Reagents for Organic Synthesis 120.000 reactions from Thiemes Science of Synthesis 6.600 reactions from Springers Comprehensive Asymmetric Catalysis
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
17 / 20
Prototype: Suggested Precursors
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
18 / 20
Prototype: Synthesis Tree
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
19 / 20
Conclusions
Very large pool of data available (SPRESI 4 million reactions) Automatic transform generation from reaction databases Suggestions based on real (analogous) reactions Verification of suggested synthesis reactions, through link to reaction database / literature Availability-check of precursors in catalogs System will be integrated in a retrieval system providing common search tools
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
Dr. V. Eigner-Pitto
20 / 20
Thank you!
InfoChem GmbH:
www.infochem.de www.spresi.de info@infochem.de
Dr. V. Eigner-Pitto
InfoChem GmbH 2006
ICIC Nmes, October 22-25, 2006
You might also like
- Understanding Power Dynamics and Developing Political ExpertiseDocument29 pagesUnderstanding Power Dynamics and Developing Political Expertisealessiacon100% (1)
- Resonance and Inductive Effects in Organic ChemistryDocument36 pagesResonance and Inductive Effects in Organic Chemistryeagl33yeNo ratings yet
- Lecture 5 - Stereoelectronic Effects On ReactivityDocument19 pagesLecture 5 - Stereoelectronic Effects On Reactivityhoang12310100% (1)
- Lecture Notes 2 Nano MaterialsDocument21 pagesLecture Notes 2 Nano MaterialsHuzaifa ShabbirNo ratings yet
- Adverb Affirmation Negation LessonDocument4 pagesAdverb Affirmation Negation LessonMire-chan Bacon100% (1)
- Hyd Schematic 2658487205 - 005Document3 pagesHyd Schematic 2658487205 - 005Angelo Solorzano100% (2)
- Adv RetrosynthesisDocument29 pagesAdv RetrosynthesisSurya Dewi Wahyuningrum100% (1)
- Adv Retrosynthesis PDFDocument29 pagesAdv Retrosynthesis PDFericaNo ratings yet
- Synthesis Review - Undergraduate Organic Synthesis GuideDocument19 pagesSynthesis Review - Undergraduate Organic Synthesis GuidePhạm Thị Thùy NhiênNo ratings yet
- Shodhganga Introduction To Synthon ApproachDocument22 pagesShodhganga Introduction To Synthon ApproachSantosh ButleNo ratings yet
- Protecting Group HandoutDocument5 pagesProtecting Group HandoutRafaelle Sanvictores SilongNo ratings yet
- CHM 1321 Assignment 5 Answers: 1) Name The Following CompoundsDocument15 pagesCHM 1321 Assignment 5 Answers: 1) Name The Following CompoundsSara Yuen100% (1)
- Retro Synthetic Analysis GuidelinesDocument12 pagesRetro Synthetic Analysis GuidelinesaukidoNo ratings yet
- Inductive EffectDocument38 pagesInductive EffectJoe JNo ratings yet
- Organic Chemistry IIDocument7 pagesOrganic Chemistry IIRoberto SIlvaNo ratings yet
- tổng hợp nghịchDocument81 pagestổng hợp nghịchruakon_ldt9527100% (1)
- Protective GroupDocument26 pagesProtective Groupchhan4No ratings yet
- Protecting Groups Guide for Amines and AlcoholsDocument45 pagesProtecting Groups Guide for Amines and AlcoholsSait MalkonduNo ratings yet
- Introduction of Organic Chemistry by Eyes of Ajnish Kumar Gupta (AKG)Document24 pagesIntroduction of Organic Chemistry by Eyes of Ajnish Kumar Gupta (AKG)ajju_208180% (5)
- Symmetry NotesDocument8 pagesSymmetry NoteslillyammalNo ratings yet
- Photochemistry & Pericyclic ReactionsDocument119 pagesPhotochemistry & Pericyclic Reactions24kemist_108741039No ratings yet
- Hetero-Cyclic CompoundsDocument69 pagesHetero-Cyclic CompoundsNaveed SajidNo ratings yet
- RetrosintesisDocument82 pagesRetrosintesisfren100% (1)
- Organometallic Chemistry: Carbonyl ComplexesDocument44 pagesOrganometallic Chemistry: Carbonyl ComplexesMior Afiq100% (1)
- Mitsunobu and Related Reactions - Advances and ApplicationsDocument101 pagesMitsunobu and Related Reactions - Advances and ApplicationsSuresh BabuNo ratings yet
- Multi-Step Organic SynthesisDocument6 pagesMulti-Step Organic SynthesisPhạm Thị Thùy NhiênNo ratings yet
- 1 Alkene Practice Problems MOC PDFDocument8 pages1 Alkene Practice Problems MOC PDFsamkarthik47No ratings yet
- Why Bother With Organic Synthesis?: Chemists Need To Make Them!Document34 pagesWhy Bother With Organic Synthesis?: Chemists Need To Make Them!FarhanAkramNo ratings yet
- Organic ChemistryDocument20 pagesOrganic ChemistryGirish RaguvirNo ratings yet
- Chem 206: Introduction to Frontier Molecular Orbital TheoryDocument22 pagesChem 206: Introduction to Frontier Molecular Orbital TheoryeraborNo ratings yet
- Problems On Named ReactionsDocument103 pagesProblems On Named ReactionsBapu ThoratNo ratings yet
- SOLVED CSIR UGC JRF NET CHEMICAL SCIENCES PAPER 1 (PART-BDocument22 pagesSOLVED CSIR UGC JRF NET CHEMICAL SCIENCES PAPER 1 (PART-BpolamrajuNo ratings yet
- Pericyclics-2014 Handout PDFDocument79 pagesPericyclics-2014 Handout PDFnavchemNo ratings yet
- Organometallic CompoundsDocument40 pagesOrganometallic CompoundsHalida SophiaNo ratings yet
- Lecture 1Document11 pagesLecture 1Fang GaoNo ratings yet
- Disconnection in Organic SynthesisDocument31 pagesDisconnection in Organic SynthesisSwami Prabhu100% (2)
- NMR SpectrosDocument29 pagesNMR Spectroshareesh13h100% (1)
- 01 StereochemistryDocument6 pages01 StereochemistryGundum Bodyz100% (1)
- Chemistry of Natural Products PDFDocument21 pagesChemistry of Natural Products PDFhosseini_9864No ratings yet
- Inorganic Chemistry Exam 1/3, March 31, 2011Document4 pagesInorganic Chemistry Exam 1/3, March 31, 2011曾鈞浩No ratings yet
- Periodic Table IPEDocument15 pagesPeriodic Table IPEAdiChemAdi100% (4)
- TautomerismDocument3 pagesTautomerismShubham JainNo ratings yet
- 242Document11 pages242John Michael CueNo ratings yet
- Electron Delocalization and ResonanceDocument9 pagesElectron Delocalization and ResonanceMariana LizethNo ratings yet
- Retrosynthesis SolutionsDocument7 pagesRetrosynthesis SolutionsScott Hendricks100% (1)
- Inductive and Resonance EffectDocument20 pagesInductive and Resonance EffectNehalPatelNo ratings yet
- IR Spectroscopy Problem Set 1Document0 pagesIR Spectroscopy Problem Set 1Jules BrunoNo ratings yet
- Organic Chemistry Midterm 1 Dir+eff++keyDocument1 pageOrganic Chemistry Midterm 1 Dir+eff++keyNorma Leticia RamosNo ratings yet
- Metal Complexes or Coordination Compounds: Kfecn 4K Fe CNDocument90 pagesMetal Complexes or Coordination Compounds: Kfecn 4K Fe CNPavan Boro100% (1)
- Organic ChemistryDocument14 pagesOrganic ChemistryStuteeNo ratings yet
- Edexcel A2 Chemistry Paper 5Document386 pagesEdexcel A2 Chemistry Paper 5AbdulRahman Mustafa100% (1)
- Solid State PDFDocument35 pagesSolid State PDFAniruddha KawadeNo ratings yet
- Retrosynthetic Analysis PDFDocument6 pagesRetrosynthetic Analysis PDFHelmi FauziNo ratings yet
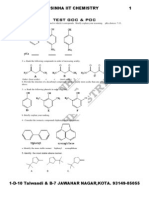
- Test1 Goc & Poc Tough by S.K.sinha See Chemistry Animations atDocument3 pagesTest1 Goc & Poc Tough by S.K.sinha See Chemistry Animations atmyiitchemistry100% (1)
- Hybrid Retrosynthesis: Organic Synthesis using Reaxys and SciFinderFrom EverandHybrid Retrosynthesis: Organic Synthesis using Reaxys and SciFinderNo ratings yet
- Medicinal Chemistry—III: Main Lectures Presented at the Third International Symposium on Medicinal ChemistryFrom EverandMedicinal Chemistry—III: Main Lectures Presented at the Third International Symposium on Medicinal ChemistryP. PratesiNo ratings yet
- Exercises in Organic Synthesis Based on Synthetic DrugsFrom EverandExercises in Organic Synthesis Based on Synthetic DrugsNo ratings yet
- Transition Metal ToxicityFrom EverandTransition Metal ToxicityG. W. RichterNo ratings yet
- L Chemical Shifts in 13CDocument1 pageL Chemical Shifts in 13Ctamilsam1986No ratings yet
- Anand.B: Current Challenging Diseases OF Rural AreaDocument10 pagesAnand.B: Current Challenging Diseases OF Rural Areatamilsam1986No ratings yet
- Anand.B: Current Challenging Diseases OF Rural AreaDocument10 pagesAnand.B: Current Challenging Diseases OF Rural Areatamilsam1986No ratings yet
- HPLC Trouble ShootingDocument47 pagesHPLC Trouble Shootingtamilsam1986No ratings yet
- RetrosynthesisDocument20 pagesRetrosynthesistamilsam19860% (1)
- Anand.B: Current Challenging Diseases OF Rural AreaDocument10 pagesAnand.B: Current Challenging Diseases OF Rural Areatamilsam1986No ratings yet
- Whirlpool FL 5064 (ET)Document8 pagesWhirlpool FL 5064 (ET)long_kongo100% (1)
- ADL MATRIX STRATEGY FOR BPCL'S GROWTHDocument17 pagesADL MATRIX STRATEGY FOR BPCL'S GROWTHSachin Nagar100% (1)
- Lux MeterDocument4 pagesLux MeterNmg KumarNo ratings yet
- WORK ORDER TITLEDocument2 pagesWORK ORDER TITLEDesign V-Tork ControlsNo ratings yet
- The Importance of WritingDocument4 pagesThe Importance of WritingBogdan VasileNo ratings yet
- The Eukaryotic Replication Machine: D. Zhang, M. O'DonnellDocument39 pagesThe Eukaryotic Replication Machine: D. Zhang, M. O'DonnellÁgnes TóthNo ratings yet
- Information HandoutsDocument6 pagesInformation HandoutsPooja Marwadkar TupcheNo ratings yet
- Tithi PRAVESHADocument38 pagesTithi PRAVESHAdbbircs100% (1)
- CommunicationDocument5 pagesCommunicationRyan TomeldenNo ratings yet
- Alaris 8210 and 8220 SpO2 Module Service ManualDocument63 pagesAlaris 8210 and 8220 SpO2 Module Service ManualNaveen Kumar TiwaryNo ratings yet
- Exhaust Brake PDFDocument2 pagesExhaust Brake PDFFeliciaNo ratings yet
- REFLEKSI KASUS PLASENTADocument48 pagesREFLEKSI KASUS PLASENTAImelda AritonangNo ratings yet
- HR MasterlistDocument6 pagesHR MasterlistLeychelle AnnNo ratings yet
- Super-Critical BoilerDocument32 pagesSuper-Critical BoilerAshvani Shukla100% (2)
- CorentineDocument559 pagesCorentinejames b willardNo ratings yet
- Events of National Importance 2016Document345 pagesEvents of National Importance 2016TapasKumarDashNo ratings yet
- Electromagnetic Braking SystemDocument14 pagesElectromagnetic Braking SystemTanvi50% (2)
- CHB1 Assignmen5Document2 pagesCHB1 Assignmen5anhspidermenNo ratings yet
- Lec08 (Topic 4 Define Classes)Document33 pagesLec08 (Topic 4 Define Classes)huaiencheengNo ratings yet
- Maths Note P1 and P3Document188 pagesMaths Note P1 and P3Afeefa SaadatNo ratings yet
- 2002 AriDocument53 pages2002 AriMbarouk Shaame MbaroukNo ratings yet
- CH 11 & CH 12 John R. Schermerhorn - Management-Wiley (2020)Document16 pagesCH 11 & CH 12 John R. Schermerhorn - Management-Wiley (2020)Muhammad Fariz IbrahimNo ratings yet
- What Is A Lecher AntennaDocument4 pagesWhat Is A Lecher AntennaPt AkaashNo ratings yet
- UN Habitat UPCL Myanmar TranslationDocument254 pagesUN Habitat UPCL Myanmar TranslationzayyarNo ratings yet
- (Math 6 WK 5 L9) - Problems Involving Addition and or Subtraction of DecimalsDocument43 pages(Math 6 WK 5 L9) - Problems Involving Addition and or Subtraction of DecimalsRhea OciteNo ratings yet
- The Godfather Term One Sample Basic Six Annual Scheme of Learning Termly Scheme of Learning WEEK 1 - 12Document313 pagesThe Godfather Term One Sample Basic Six Annual Scheme of Learning Termly Scheme of Learning WEEK 1 - 12justice hayfordNo ratings yet
- Math 101Document3 pagesMath 101Nitish ShahNo ratings yet