Professional Documents
Culture Documents
Energy Ans
Uploaded by
kevinamyOriginal Description:
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Energy Ans
Uploaded by
kevinamyCopyright:
Available Formats
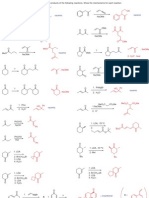
CHEM211 Problem Set
Reaction Energetics
1) For the molecules below:
a) Draw the most stable and least stable conformations.
b) Calculate the difference in energy between the conformations.
c) Calculate the equilibrium constant (Keq) for conversion between the conformations at room
temperature (70 oF, 21 oC, 294 K).
OH
(H3C)2HC CH3 H3CH2C CH(CH3)2
t-Bu Cl
H3CH H CH3
H H3C H HH CH3 H H CH3
OH
H H H3C H Cl
H3C HH CH3 H3C HCH3 CH3 t-Bu
Cl
CH3
H3C H HCH3 H3C CH3 H3CCH3 t-Bu
H H
H3CH HH H CH3 HH H3C H
H HCH3 H CH3 OH
CH3
4 + 6 + 6 = 16 kJ/mol 11 - 4 = 7 kJ/mol 2x(11.4) - 4x(3.8) 2x(1.0) + 2x(2.1)
= 7.6 kJ/mol = 6.2 kJ/mol
∆Go = -RT lnKeq Keq = 5.7 x 10 -2
Keq = 1.4 x 10 -3 Keq = 4.46 x 10 -2 Keq = 7.92 x 10 -2
2) Sketch an energy diagram to match each of the following descriptions:
a) Fast reaction; ∆Go = -120 kJ/mol.
b) Slow reaction; ∆Go = 20 kJ/mol.
c) Very slow reaction; Keq = 1.36×10 –3.
d) Fast reaction; Keq = 3.0.
e) A two step reaction; first step is fast; second step is slow; ∆Go = -80 kJ/mol.
f) ∆G1‡ = 200 kJ/mol; ∆G2‡ = 50 kJ/mol; ∆Go = -40 kJ/mol.
TS
E TS E
∆G
SM
∆ Go ∆G
= -120
P
SM ∆ Go
P = +20
reaction coordinate reaction coordinate
TS
E ∆G E TS
∆G
P SM
P
∆Go = 16 o
∆G = -2.7
SM
reaction coordinate reaction coordinate
TS1
TS2
TS2 ∆G2 = 50
I
∆ G2
TS1 ∆ G1
I E = 200
E
∆ G1
SM
SM
∆ Go
= -80 ∆ Go
= -40
P P
reaction coordinate reaction coordinate
3) For each energy diagram in (2), lable starting material(s) (SM), product(s) (P), intermediate(s) (I),
transition state(s) (TS‡), activation energy(s) (∆
∆G‡) and free energy (∆
∆Go).
4) For the reactions below, calculate ∆Go, ∆Ho and ∆So.
290 K 19
Br2 Br Keq = 1.24 x 10
Br
∆Go = -RT lnKeq = -(8.315 J/K mol)(290K) ln (1.24×1019) = -106,000 J/mol = -106 kJ/mol
∆Ho = ∆Hf bonds broken – ∆Hf bonds formed = ∆Hf C=C + ∆Hf Br-Br – 2(∆Hf C-Br) – ∆Hf C-C =
611 + 193 – 2(285) – 376 = -142 kJ/mol
∆Go = ∆Ho – T∆So
∆So = - (∆Go – ∆Ho)/T = - (-106 – (-142))/290 = -0.124 kJ/K mol = -124 J/K mol
HBr Keq = 3.96 x 10 15
Br
∆Go = -RT lnKeq = -(8.315 J/K mol)(290K) ln (3.96×1019) = -87,000 J/mol = -87 kJ/mol
∆Ho = ∆Hf C=C + ∆Hf H-Br – ∆Hf C-H – ∆Hf C-Br – ∆Hf C-C =
611 + 366 – 420 – 285 – 376 = -104 kJ/mol
∆So = - (∆Go – ∆Ho)/T = - (-87 – (-104))/290 = -0.060 kJ/K mol = -60 J/K mol
Br Br Keq = 0.13
O OH
∆Go = -RT lnKeq = -(8.315 J/K mol)(290K) ln (0.13) = 4900 J/mol = 4.9 kJ/mol
∆Ho = ∆Hf C-H + ∆Hf C-Br + ∆Hf C-C – ∆Hf O-H – ∆Hf C=C =
420 + 285 + 376 – 437 – 611 = 33 kJ/mol
∆So = - (∆Go – ∆Ho)/T = - (4.9 – 33)/290 = 0.097 kJ/K mol = 97 J/K mol
You might also like
- Haloalkanes PDFDocument6 pagesHaloalkanes PDFthc8477No ratings yet
- Chemistry 108M Final Exam Practice 1Document8 pagesChemistry 108M Final Exam Practice 1Norma Leticia RamosNo ratings yet
- 3.2.1 Enthalpy ChangesDocument9 pages3.2.1 Enthalpy ChangesSofia YNo ratings yet
- Chapter 7 - Structure and Synthesis of Alkenes PDFDocument40 pagesChapter 7 - Structure and Synthesis of Alkenes PDFSam0% (1)
- 11chemistry OMEGA PDFDocument96 pages11chemistry OMEGA PDFChirAgNo ratings yet
- Semi-Empirical Methods: CHEM 430Document42 pagesSemi-Empirical Methods: CHEM 430chama_gozNo ratings yet
- 10 Grand Canonical EnsembleDocument12 pages10 Grand Canonical EnsembleAnonymous FRKcnDx7No ratings yet
- EnolateansDocument1 pageEnolateanskevinamyNo ratings yet
- Chemical Kinetics Mechanisms and Rate Laws ExplainedDocument29 pagesChemical Kinetics Mechanisms and Rate Laws ExplainedAribazChemNo ratings yet
- T1-1 TDocument30 pagesT1-1 TFRENCHONLYNo ratings yet
- Organometallic CompoundsDocument66 pagesOrganometallic CompoundsJon Ho100% (1)
- Caieee04fisica PDFDocument15 pagesCaieee04fisica PDFRafaelNo ratings yet
- Organic 6 CDocument26 pagesOrganic 6 CDr.Rajarshi PatelNo ratings yet
- Reactions of Alkenes: CC HX C HX C Markovnikov's OrientationDocument8 pagesReactions of Alkenes: CC HX C HX C Markovnikov's OrientationMarc RitzNo ratings yet
- Bioinorganic HandoutDocument63 pagesBioinorganic HandoutAL__52No ratings yet
- Practical Organic Chemistry (I) : October 2017Document44 pagesPractical Organic Chemistry (I) : October 2017NUR ALOMNo ratings yet
- CH 44 Organic Reactions - Supp Ex 1 (Updated)Document4 pagesCH 44 Organic Reactions - Supp Ex 1 (Updated)伊貝P-No ratings yet
- Chem 212 Alkyl Halide Problems 3Document1 pageChem 212 Alkyl Halide Problems 3kevinamyNo ratings yet
- HW1 Solns KineticsDocument10 pagesHW1 Solns Kineticsapb91781No ratings yet
- Tutorial Sheets For Thermodynamics 02Document3 pagesTutorial Sheets For Thermodynamics 02Aditya raj sachdevNo ratings yet
- Thermochemistry Answers RemovedDocument11 pagesThermochemistry Answers Removedapi-327309463No ratings yet
- Cy 101 Uv-Vis and Ir NewDocument66 pagesCy 101 Uv-Vis and Ir NewSomesh MohapatraNo ratings yet
- Chem 212 Alkyl Halide Problems 4Document1 pageChem 212 Alkyl Halide Problems 4kevinamyNo ratings yet
- Chemical Kinetics (M) PDFDocument41 pagesChemical Kinetics (M) PDFNalla Umapathi Reddy75% (4)
- Chapter 20 & 21 - Wade - NDDocument93 pagesChapter 20 & 21 - Wade - NDyonggyeNo ratings yet
- Spectros PDFDocument28 pagesSpectros PDFbalajiNo ratings yet
- Lecture Notes Chapter-12-Aldehydes, Ketones & Carboxylic AcidsDocument26 pagesLecture Notes Chapter-12-Aldehydes, Ketones & Carboxylic AcidsSHUBHAMNo ratings yet
- Physics Brochure PDFDocument12 pagesPhysics Brochure PDFKatrina PanaliganNo ratings yet
- Assignment 1Document3 pagesAssignment 1Andrew_Wong_8492No ratings yet
- 13B Heat of FusionDocument8 pages13B Heat of FusionManish KumarNo ratings yet
- Organic Chemistry Chapter 19: AminesDocument40 pagesOrganic Chemistry Chapter 19: Aminesmelg16No ratings yet
- Aldehydes Ketones HaccDocument66 pagesAldehydes Ketones HaccRammohan VaidyanathanNo ratings yet
- Organic Chemistry Final Exam - Questions OnlyDocument9 pagesOrganic Chemistry Final Exam - Questions OnlybrookNo ratings yet
- Sheet 2Document2 pagesSheet 2Ahmed Rabie Abd Elazeem100% (1)
- The S-Block Elements: The Elements in Which Last Electron Enters The S-Subshell Are Called As S-Block ElementsDocument36 pagesThe S-Block Elements: The Elements in Which Last Electron Enters The S-Subshell Are Called As S-Block ElementstheDarknight2050 (Karthik Sunil)No ratings yet
- Chapter 8 - Alkene ReactivityDocument23 pagesChapter 8 - Alkene ReactivitySimran DhunnaNo ratings yet
- Reactions of AlcoholsDocument49 pagesReactions of AlcoholsayushNo ratings yet
- Chem 212 Alkyl Halide Problems 2Document1 pageChem 212 Alkyl Halide Problems 2kevinamy100% (1)
- CHE 229 Lab Report 1Document7 pagesCHE 229 Lab Report 1Stephanie CarreraNo ratings yet
- Aromatic Compounds and Uses of BenzeneDocument44 pagesAromatic Compounds and Uses of BenzenePaarijat DubeyNo ratings yet
- Organometallic ChemistryDocument31 pagesOrganometallic ChemistrySadiaKhan100% (1)
- Organic Chemistry 2 Practice Exam 1Document15 pagesOrganic Chemistry 2 Practice Exam 1KaybidoNo ratings yet
- Alkyl Halides & Aryl Halides-02 - Solved ProblemsDocument13 pagesAlkyl Halides & Aryl Halides-02 - Solved ProblemsRaju SinghNo ratings yet
- Molecular Orbital Therory-Diatomic MoleculesDocument25 pagesMolecular Orbital Therory-Diatomic MoleculesDnyaneshwar ShindeNo ratings yet
- Chemistry 3 Electrochemistry PDFDocument15 pagesChemistry 3 Electrochemistry PDFinfinilifeNo ratings yet
- Organometallic ChemistryDocument24 pagesOrganometallic ChemistryFatma TaherNo ratings yet
- Alkanes - Alkenes - Alkynes - DPP 2Document3 pagesAlkanes - Alkenes - Alkynes - DPP 2Vishal_93No ratings yet
- Stereochemistry Basic Concepts Useful Notes For StudentsDocument26 pagesStereochemistry Basic Concepts Useful Notes For StudentsReddappaNo ratings yet
- G R Reduction AlkaneDocument43 pagesG R Reduction AlkaneManthan HaritashNo ratings yet
- Stereochemistry: 4 Edition Paula Yurkanis BruiceDocument43 pagesStereochemistry: 4 Edition Paula Yurkanis Bruicenrguerrerod100% (1)
- 12 August Mock TestDocument65 pages12 August Mock Testapi-26674800No ratings yet
- Chapter 9 PDFDocument12 pagesChapter 9 PDFKelsi Kyla Peralta0% (1)
- Practice Questions-Conformational AnalysisDocument4 pagesPractice Questions-Conformational AnalysisHarry Zgambo100% (1)
- Org Chem Final ReviewerDocument7 pagesOrg Chem Final ReviewerblessaNo ratings yet
- HANDOUTESDocument24 pagesHANDOUTESJohn Owen Dipad100% (1)
- Organic Chemistry 32-235 Practice Questions For Exam #2: 2. Consider The SDocument9 pagesOrganic Chemistry 32-235 Practice Questions For Exam #2: 2. Consider The Ssweta KushwahaNo ratings yet
- Alkyl HalidesDocument20 pagesAlkyl HalidesShivam Gupta0% (1)
- CLS AN 107 ChemCompPlasticDocument1 pageCLS AN 107 ChemCompPlasticPim ChaNo ratings yet
- Exercise With Ans FinalDocument24 pagesExercise With Ans Finald anjilappa25% (4)
- Chem 212 Electrophilic Aromatic Substitution 2Document1 pageChem 212 Electrophilic Aromatic Substitution 2kevinamyNo ratings yet
- Chem 212 Alkyl Halide Problems 2Document1 pageChem 212 Alkyl Halide Problems 2kevinamy100% (1)
- Chem 212 Alkyl Halide Problems 3Document1 pageChem 212 Alkyl Halide Problems 3kevinamyNo ratings yet
- Chem 212 Alkyl Halide Problems 4Document1 pageChem 212 Alkyl Halide Problems 4kevinamyNo ratings yet
- CarboxylicansDocument1 pageCarboxylicanskevinamyNo ratings yet
- Chem 212 Alkyl Halide Problems 5Document1 pageChem 212 Alkyl Halide Problems 5kevinamyNo ratings yet
- CH211 ALKYNES Problem Set 1. Provide The Products For TheDocument1 pageCH211 ALKYNES Problem Set 1. Provide The Products For ThekevinamyNo ratings yet
- Chem 212 Condensation Reactions 3Document1 pageChem 212 Condensation Reactions 3kevinamyNo ratings yet
- Condensation 1 AnsDocument1 pageCondensation 1 AnskevinamyNo ratings yet
- CHEM211 Problem Set Functional Groups and Reaction Types These AnswersDocument4 pagesCHEM211 Problem Set Functional Groups and Reaction Types These AnswerskevinamyNo ratings yet
- Condensation 2 AnsDocument1 pageCondensation 2 AnskevinamyNo ratings yet
- EnolateansDocument1 pageEnolateanskevinamyNo ratings yet
- Alkane Stereochemistry 1) For The Molecules Below: A) Provide ADocument3 pagesAlkane Stereochemistry 1) For The Molecules Below: A) Provide AkevinamyNo ratings yet
- Chem 212 Provide The Products For The Following Reactions. BeDocument1 pageChem 212 Provide The Products For The Following Reactions. BekevinamyNo ratings yet
- Stereochemistry Problem Set 1) Indicate Whether The Following Structures AreDocument1 pageStereochemistry Problem Set 1) Indicate Whether The Following Structures ArekevinamyNo ratings yet
- MSansDocument2 pagesMSanskevinamyNo ratings yet
- Naming Alkanes AnsDocument1 pageNaming Alkanes AnskevinamyNo ratings yet
- Giao Ly For KidsDocument2 pagesGiao Ly For KidskevinamyNo ratings yet
- NMRansDocument1 pageNMRanskevinamyNo ratings yet
- SpecansDocument2 pagesSpecanskevinamyNo ratings yet
- Synthesis 3 AnsDocument1 pageSynthesis 3 AnskevinamyNo ratings yet
- Pharmaceutical Chemistry Answer Key Blue PacopDocument30 pagesPharmaceutical Chemistry Answer Key Blue PacopJeannie UyNo ratings yet
- JTB Pulse - Str1 - Data2 - FCADocument2 pagesJTB Pulse - Str1 - Data2 - FCAsutandri siregar100% (1)
- Bragg Williams ApproximationDocument18 pagesBragg Williams ApproximationKunal Garg100% (1)
- Experiment No. 1 Physical Process: Name: Ariane Mamada Date: August 11, 2022 Yr & Section: 2 - V2A ScoreDocument17 pagesExperiment No. 1 Physical Process: Name: Ariane Mamada Date: August 11, 2022 Yr & Section: 2 - V2A ScoreAriane MamadaNo ratings yet
- PH8251 - Material ScienceDocument320 pagesPH8251 - Material ScienceBalaji K100% (1)
- PROCHILL B4K Operation ManualDocument97 pagesPROCHILL B4K Operation Manualanil perala100% (1)
- Answers Bahan Pelajar Bintang Carbon Compound: Quiz 1 1 (A)Document5 pagesAnswers Bahan Pelajar Bintang Carbon Compound: Quiz 1 1 (A)airinNo ratings yet
- NTU Sample PhysicsDocument5 pagesNTU Sample PhysicsVũ LêNo ratings yet
- MCQs on AromaticityDocument10 pagesMCQs on AromaticityGaurav YadavNo ratings yet
- Manual ChromatotronDocument48 pagesManual ChromatotronmurandikesumaNo ratings yet
- Chemistry: SyllabusDocument51 pagesChemistry: SyllabusYash GuptaNo ratings yet
- Introduction to Brayton CycleDocument19 pagesIntroduction to Brayton CycleSayantan DuttaNo ratings yet
- TDS 11594139 Performance Test Standards KitDocument4 pagesTDS 11594139 Performance Test Standards KitSamuel SuNo ratings yet
- Introduction To CFPPDocument34 pagesIntroduction To CFPPVenkat CherukuriNo ratings yet
- CHEMICAL TEXTURE SERVICES HandoutsDocument4 pagesCHEMICAL TEXTURE SERVICES HandoutsMARIALUPE ESTOQUENo ratings yet
- Gcse Combined Science: Trilogy: Higher Tier Paper 5: Physics 1HDocument23 pagesGcse Combined Science: Trilogy: Higher Tier Paper 5: Physics 1HOmar AbdallaNo ratings yet
- A Detailed Lesson Plan in Grade 6 ScienceDocument10 pagesA Detailed Lesson Plan in Grade 6 ScienceMaria Daniela ViduyaNo ratings yet
- Exam Style Answers 21 Asal Physics CBDocument2 pagesExam Style Answers 21 Asal Physics CBAnshul Shah100% (1)
- XRD applications and interpretation: determining crystal structure, phase identification, quantitative analysis and more from powder diffraction patternsDocument112 pagesXRD applications and interpretation: determining crystal structure, phase identification, quantitative analysis and more from powder diffraction patternsDionisius Rinus AjiNo ratings yet
- Ammonia RecoveryDocument136 pagesAmmonia RecoveryWagus GinanjarNo ratings yet
- ASHRAE Terminology - Complete ListDocument298 pagesASHRAE Terminology - Complete Listwado11No ratings yet
- Brianna Ramirez - Greenhouse EffectDocument5 pagesBrianna Ramirez - Greenhouse EffectBriannaNo ratings yet
- Problems 1 0100103304 2-59Document1 pageProblems 1 0100103304 2-59Jj LewandyNo ratings yet
- Thermodynamic Properties of Liquid Metals PDFDocument7 pagesThermodynamic Properties of Liquid Metals PDFKaren CabezasNo ratings yet
- Natural Gas Lecture NoteDocument7 pagesNatural Gas Lecture NoteShakerMahmood100% (1)
- Lec 13Document44 pagesLec 1320-c-859No ratings yet
- Characterization of Hydrocarbons Using Different Chemical TestsDocument5 pagesCharacterization of Hydrocarbons Using Different Chemical TestsRenz Roger Esteves BuendichoNo ratings yet
- RT Level III QuestionsDocument3 pagesRT Level III QuestionsMourad AdelNo ratings yet
- DBM 114 MCQDocument29 pagesDBM 114 MCQROHIT bhuiNo ratings yet
- 5-2. Solar RefrigerationDocument13 pages5-2. Solar RefrigerationSuyash BajpaiNo ratings yet