Professional Documents
Culture Documents
ATP Is The Universal Currency of Free Energy in Biological Systems
Uploaded by
AdistyDW0 ratings0% found this document useful (0 votes)
164 views1 pageatp
Original Title
ATP is the Universal Currency of Free Energy in Biological Systems
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this Documentatp
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
164 views1 pageATP Is The Universal Currency of Free Energy in Biological Systems
Uploaded by
AdistyDWatp
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 1
ATP Is the Universal Currency of Free Energy in Biological Systems
ATP is an energy-rich molecule because its triphosphate unit contains two
phosphoanhydride bonds
A large amount of free energy is liberated when ATP is hydrolyzed to
adenosine diphosphate (ADP) and orthophosphate (Pi) or when ATP is
hydrolyzed to adenosine monophosphate (AMP) and pyrophosphate (PPi).
The role of ATP in energy metabolism is paramount
Penting !
Living organisms require a continual input of free energy for three major purposes: (1) the
performance of mechanical work in muscle contraction and other cellular movements, (2) the
active transport of molecules and ions, and (3) the synthesis of macromolecules and other
biomolecules from simple precursors
How does coupling to ATP hydrolysis make possible an otherwise unfavorable reaction?
Consider a chemical reaction that is thermodynamically unfavorable without an input of free
energy, a situation common to many biosynthetic reactions => In other words, coupling the
hydrolysis of ATP with the conversion of A into B has changed the equilibrium ratio of B to A by
a factor of about 108.
We see here the thermodynamic essence of ATP's action as an energy-coupling agent. Cells
maintain a high level of
ATP by using oxidizable substrates or light as sources of free energy. The hydrolysis of an ATP molecule
in a coupled
reaction then changes the equilibrium ratio of products to reactants by a very large factor, of the order of
108. More
generally, the hydrolysis of n ATP molecules changes the equilibrium ratio of a coupled reaction (or
sequence of reactions) by a factor of 108n
3. The electron donor in most reductive biosyntheses is NADPH, the reduced form of nicotinamide
adenine dinucleotide phosphate (NADP+; see Figure 14.13). NADPH differs from NADH in that the 2 -
hydroxyl group of its adenosine moiety is esterified with phosphate. NADPH carries electrons in the same
way as NADH. However, NADPH is used almost exclusively for reductive biosyntheses, whereas NADH
is used primarily for the generation of ATP. The extra phosphoryl group on NADPH is a tag that enables
enzymes to distinguish between high-potential electrons to be used in anabolism and those to be used in
catabolism.
You might also like
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
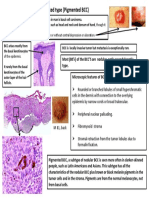
- Quick DX Basal Cell Carcinoma Pigmented TypeDocument1 pageQuick DX Basal Cell Carcinoma Pigmented TypeAdistyDWNo ratings yet
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (587)
- Mosquito Diseases Guide: Dengue, Malaria, Chikungunya & FilariasisDocument80 pagesMosquito Diseases Guide: Dengue, Malaria, Chikungunya & Filariasisalessandro linbladNo ratings yet
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (890)
- Quick DX Basal Cell Carcinoma Pigmented TypeDocument1 pageQuick DX Basal Cell Carcinoma Pigmented TypeAdistyDWNo ratings yet
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- 3D Corporate Slide Template For Keynote Presentation in Microsoft PowerPointDocument2 pages3D Corporate Slide Template For Keynote Presentation in Microsoft PowerPointashrafNo ratings yet
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (399)
- A 12-Year Retrospective Study of Invasive Amoebiasis in Western Sydney: Evidence of Local AcquisitionDocument8 pagesA 12-Year Retrospective Study of Invasive Amoebiasis in Western Sydney: Evidence of Local AcquisitionAdistyDWNo ratings yet
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (73)
- Diving Medicine: "Dikbrevet Juru Selam Tni Al" 1Document41 pagesDiving Medicine: "Dikbrevet Juru Selam Tni Al" 1AdistyDWNo ratings yet
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- Pink Pastel Free PresentationDocument10 pagesPink Pastel Free PresentationAlfianoVanDavidJonesNo ratings yet
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- 766 - HPS - Emergency TKVDocument82 pages766 - HPS - Emergency TKVAdistyDWNo ratings yet
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Jurnal PDFDocument8 pagesJurnal PDFMahdhun ShiddiqNo ratings yet
- 01 Everything That Can Be Invented Has Been Invented - Clive Maxfield PDFDocument79 pages01 Everything That Can Be Invented Has Been Invented - Clive Maxfield PDFAdistyDWNo ratings yet
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2219)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (344)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (265)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (119)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)