Professional Documents
Culture Documents
Cellular Injury, Adaptation and Cell Death
Uploaded by
Jessica Febrina Wuisan100%(1)100% found this document useful (1 vote)
195 views8 pagespathology
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this Documentpathology
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
100%(1)100% found this document useful (1 vote)
195 views8 pagesCellular Injury, Adaptation and Cell Death
Uploaded by
Jessica Febrina Wuisanpathology
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 8
1 of 8 Page
CELLULAR INJURY, ADAPTATION & CELL DEATH
NOTES:
Ex: In systemic hypertension, increase BP heart has to work against increase in
peripheral resistance cardiac muscle adapts by increasing in cell size =
Hypertrophy
When there is persistent hypertension (uncontrolled), myocardial cell exceeds its limit
of adaptation will decompensate and undergo changes (ex. lysis of cells) =
Irreversible cell injury (usually in the form of coagulative necrosis in the heart
Myocardial Infarction)
Usually apoptosis is not seen in cardiac muscle
CELLULAR ADAPTATIONS OF GROWTH AND DIFFERENTIATION
Occurs when excessive physiologic stresses or some pathologic
stimuli result in a new but altered state that preserves the viability
of the cell
Adaptations are reversible responses to physiologic stress and
pathologic stimuli; Takes several forms
A. ATROPHY
- Decrease in cell size and number, leading to decrease in size of
tissue/organ; Shrinkage in the size of the cell by loss of cell integrity and
substance (mitochondria, myofilaments, ER)
- Mechanism: result from decrease in protein synthesis as result of
a decrease in metabolic activity and increase in protein
degradation via ubuquitin-proteosome pathway
Ubiquitin ligase ubiquitin attaches to cellular protein degradation
Atrophy may also be accompanied by increased AUTOPHAGY (self eating)
wherein a starved cell eats its own components to survive.
- Causes:
1) Decrease workload (disuse atrophy) immobilization
- Immobilized fractured bone or complete bed rest cause
skeletal muscle atrophy; reversible once activity is
resumed
- Skeletal muscle fibers decrease in cell size and number
which can lead to increased bone resorption then to
osteoporosis
2) Loss of innervations muscle atrophy (paralyzed patients)
3) Decrease blood supply (ex. arteriosclerosis of the BV
supplying the brain cerebral ischemia cerebral
atrophy)
4) Inadequate nutrition
- Marasmus (profound protein-calorie malnutrition) is
associated with the use of skeletal muscle as a source
of energy after other reserves have been depleted
resulting in muscle wasting (Cachexia)
5) Loss of endocrine stimulation
- A) Postmenopause: Loss of estrogen stimulation after
menopause results in physiologic atrophy of the
endometrium/vaginal epithelium and breast (atrophy of
the lining epithelium of the uterus = becomes thin and
shiny); B) Aging
6) Pressure atrophy
- Tissue compression for any given time
- Ex. Benign tumors can cause atrophy in the surrounding
tissues
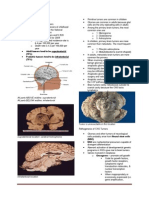
ATHEROSCLEROTIC CEREBROVASCULAR DISEASE resulting in reduced blood
supply; Loss of brain substance narrows the gyri and widens the sulci
Physiologic Atrophy: Occurs during normal development; Ex. Atrophy of notochord
during fetal development
B. HYPERTROPHY
- Increase in the size of cells that results to increased organ size
- Mechanism: results of increased production of cellular proteins
(structural components)
- New cells are not present, only larger ones
- Hypertrophy can be accompanied by hyperplasia among normally
dividing cells; But non-dividing cells undergoes HYPERTROPHY
when stressed.
1) PHYSIOLOGIC HYPERTROPHY
1. Hormonal stimulation
Ex. Increase in uterine size during pregnancy due
to estrogenic hormones; breast and sex organs
during puberty
2. Increased functional demand
Ex. Exercise
2) PATHOLOGIC HYPERTROPHY
1. Abnormal hormone levels
Ex. Enlargement of thyroid gland, due excessive
levels of T3 and T4 (Goiter)
2. Increased functional Demand
Ex. Chronic hemodynamic overload in left
ventricular hypertrophy as seen in hypertension
and valvular disease
ADDITIONAL:
The most common stimulus for muscle hypertrophy is an increase in workload.
Mechanisms of Hypertrophy:
- The coordinated actions of mechanical sensors, growth factors (GFs) and
vasoactive agents increase the synthesis of muscle proteins responsible for
hypertrophy.
- Hypertrophy can also be associated with the switch of contractile protein from
adult to fetal or neonatal forms (Kumar, Abbas, Fausto, & Aster, 2009, p. 7).
- Occurs in non-dividing cells like myocardial and skeletal muscle fibers
C. HYPERPLASIA
- Increase in the number of cells in an organ/tissue resulting to
increase mass of organ/tissue
- Mechanism: due to the growth factor-driven proliferation of mature
cells and sometimes by increased output of new cells from tissue
stem cells
- local production of growth factors, levels of growth factor receptors on the
responding cells, or activation of particular intracellular signaling pathways
that leads to production of transcription factors that turn on many cellular
genes (encoding growth factors, receptors for growth factors, and cell cycle
regulators) and the net result is cellular proliferation
- Occurs if the cell population is capable of dividing
1) PHYSIOLOGIC HYPERPLASIA
a) Hormonal pregnant uterus; proliferation of glandular
epithelium of breast at puberty or pregnancy which is
usually accompanied by hypertrophy
2 of 8 |Page
b) Compensatory liver regeneration after partial
hepatectomy via transforming growth factor alpha
(counteracted by transforming growth factor beta);
In cases when there is massive destruction of
liver cells (ex. In viral hepatitis), liver cell
cannot regenerate itself because of the
destruction of CT framework necessary for
regeneration; resulting to
Compromise proliferation hepatocytes
regenerates from the intrahepatic stem cells
2) PATHOLOGIC HYPERPLASIA
- Most forms are caused by excess hormones or growth
factor
- The process remains controlled because the
hyperplasia regresses if the stimulation is eliminated
- Hyperplasia is distinct from cancer. But pathologic
hyperplasia constitutes fertile soil in which cancerous
proliferation may eventually arise.
a) Excessive hormonal stimulation
Hyperestrinism endometrial hyperplasia (precursor
of carcinoma if there are atypical changes) CA
b) Effect of growth factors on target organs
Ex. Mitogenic factors hyperplasia in wound healing;
Papilloma virus skin wart
Additional:
- During Menstrual Period, there is a rapid burst of proliferative activity in
the epithelium stimulated by pituitary hormones and ovarian estrogen
- It is brought to a halt by rising level of progesterone usually 10-14 days
after the end of menstrual period
- *In some instances, the balance of Estrogen and Progesterone is
disturbed resulting to absolute or relative increase in the amount of
ESTROGEN with consequent HYPERPLASIA of endometrial glands and
this is the common cause of ABNORMAL MENSTRUAL BLEEDING
- BENIGN PROSTATIC HYPERPLASIA in response to androgens. But this
abnormality remains controlled because it causes no mutation in genes
that regulate cell division
D. METAPLASIA
- Reversible change wherein one adult cell type is replaced by
another adult cell type
- Usually involves epithelial cells and mesenchymal cells. The most
common epithelial metaplasia is COLUMNAR TO SQUAMOUS
- Mechanism: result of a reprogramming of stem cells that are
known to exist in normal tissues, or of undifferentiated
mesenchymal cells present in connective tissue. The precursor
cell differentiate along a new pathway brought about by signals
generated by cytokines, GFs, and extracellular matrix components
in the cells environment.
- May represent adaptive substitution of cells that are sensitive to
stress by cell types better able to withstand the adverse
environment
1) SQUAMOUS METAPLASIA
- Chronic respiratory tract irritation (cigarette smokers);
normal ciliated and bronchi replaced by stratified
squamous columnar epithelium of trachea epithelium
- Stones in excretory ducts, pancreas and bile ducts,
vitamin A deficiency; secretory columnar to stratified
squamous; mucus secretion and ciliary function are lost
2) OSTEOBLASTIC OR CHONDROBLASTIC METAPLASIA
of fibroblasts
3) APOCRINE METAPLASIA (seen in specimens of breast
mass with fibrocystic change; most likely are with apocrine
metaplasia is benign)
- If stimuli predisposing to hyperplasia persist dysplasia/cancer
may occur.
Additional:
- BARRETs ESOPHAGUS: ESOPHAGEAL SQUAMOUS to intestinal-like
COLUMNAR under the influence of refluxed gastric acid
(Glandular/Adeno carcinoma)
- ENDOCERVIX: NORMAL COLUMNAR to STRATIFIED SQUAMOUS
E. DYSPLASIA
- Epithelial or mesenchymal cells undergo proliferation and atypical
cytologic changes involving cell size, shape and organization (loss
of polarity, inc. mitoses, inc. N/C ratio, irreg. or thickening of
nuclear membrane, hyperchromasia, chromatin clumps)
- Sometimes, cancer precursors (cervix and respiratory tract)
- In cervical cancer, desquamation is observed at the
squamocolumnar junction.
CELL INJURY
Occurs when a cells exposure to injurious agents or stress which
exceeds its capability for adaptive response
Can be reversible or irreversible
If irreversible, it can lead to cell death.
CAUSES OF CELL INJURY:
1) Hypoxia - deficiency in oxygen which causes cell injury by
decreasing aerobic oxidative respiration; Causes are
Ischemia, Cardiorespiratory failure (Inadequate oxygenation
of blood), Anemia (decreased oxygen carrying capacity),
Carbon Monoxide poisoning (block oxygen carriage), severe
blood loss
2) Physical agents - mechanical trauma, extremes of temp.,
sudden changes in atmospheric pressure, radiation, electric
shock
3) Chemical agents and drugs - hypertonic conc. of glucose/
salt, high oxygen conc., poisons, environmental and air
pollutants, insecticides/ herbicides, CO and asbestos,
alcohol, narcotics, etc.
4) Infectious agents - viruses, bacteria, rickettsiae, fungi,
parasites
5) Immunologic reactions - anaphylaxis, autoimmune
diseases
6) Genetic derangements - genetic injury (Down's syndrome
caused by chromosomal anomaly), sickle cell anemia inborn
errors of metabolism, accumulation of damaged DNA or
misfolded proteins
7) Nutritional imbalances - protein-calorie deficiency, vitamin
def., atherosclerosis
FOUR COMMON CAUSES OF CELL INJURY:
1) Hypoxia
2) Free radicals and activated O2 species
3) Some chemicals
4) Viruses
Duration of hypoxia needed to produce irreversible cellular injury
varies according to:
1) Cell type
Susceptibility of Cells to Ischemic Necrosis.
High 3-5 minutes Neurons
Intermediate 30 minutes
2 hours
Myocardium, hepatocyte, renal
epithelium
Low Many hours Fibroblast, epidermis, skeletal
muscle
2) Nutritional state
3) Hormonal status
3 of 8 |Page
MECHANISMS OF CELLULAR INJURY:
1. DEPLETION OF ATP PRODUCTION AND SYNTHESIS
- Associated with hypoxic and toxic injury
- Major causes are reduction of oxygen supply, mitochondrial
damage and actions of toxins
- Effects of depletion of ATP to 5% to 10% of normal levels are:
Decreased plasma membrane energy-dependent sodium
pump; Sodium enters and accumulates inside cells and
potassium diffuses out, causing cell swelling and dilation of
the ER
Altered cellular energy metabolism
- If supply of oxygen to cells is reduced
- Resulting to decreased cellular ATP and increased AMP
- Increased rate of anaerobic glycolysis will cause
accumulation of lactic acid and inorganic phosphates,
reducing intracellular pH and decreased activity of many
cellular enzymes
Influx of calcium
due to pump failure: leads to influx of Ca
2+
Decreased protein synthesis: due to prolonged or worsening
depletion of ATP, structural disruption of the protein synthetic
apparatus occurs
2. MITOCHONDRIAL DAMAGE
- May be caused by increased cytosolic Ca
2+
, reactive oxygen
species and O2 deprivation
- Two major consequences:
o Formation of mitochondrial permeability transition pore
loss of mitochondrial membrane potential failure of
oxidative phosphorylation decrease in ATP necrosis
o Leakage of proteins (ex. cytochrome c) that activate
apoptosis into the cytosol
3. Ca
2+
INFLUX AND LOSS OF Ca
2+
HOMEOSTASIS
- Ischemia and certain toxins causes this intracellular calcium
causes cell injury by several mechanisms:
Opening of mitochondrial permeability transition pore
Activation of a number of enzymes (phospholipases,
proteases, endonucleases)
Induction of apoptosis by direct activation of caspases and
by increasing mitochondrial permeability
4. ACCUMULATION OF OXYGEN-DERIVED FREE RADICALS
- Reactive Oxygen Species (ROS)-produced normally in cells
during mitochondrial respiration and energy generation, but
degraded and removed by cellular defense systems; also
produced by leukocytes, particularly neutrpphils and
macrophages
- Oxidative stress- condition that is a result of excessive free
radicals when production of ROS increases or the scavenging
systems are ineffective (implicated in cell injury, cancer, aging,
some degenerative diseases like Alzheimers)
5. MEMBRANE DAMAGE
- in ischemic cells, membrane defects may be the result of ATP
depletion and calcium-mediated activation of phospholipases
- direct damage may also be caused by bacterial toxins, viral
proteins, lytic complement components, physical and chemical
agents
Biochemical Mechanisms Contributing to Membrane Damage:
- ROS lipid peroxidation
- Decreased phospholipids synthesis due to defective
mitochondrial function or hypoxia
- Increased phospholipid breakdown due to activation of
endogenous phospholipases by increased cytosolic Ca
2+
; lipid
breakdown products may insert into the lipid bilayer of membrane
or exchange with membrane phospholipids, causing changes in
permeability and electrophysiologic alterations
- Cytoskeletal abnormalities due to activation of proteases by
increased cytosolic Ca
2+
; results to detachment of the cell
membrane from cytoskeleton, rendering it susceptible to
stretching and rupture
Consequences of Membrane Damage
- mitochondrial membrane damage = opening of mitochondrial
permeability transition pore
- plasma membrane damage = loss of osmotic balance
- injury to lysosomal membranes = leakage of enzymes into the
cytoplasm and activation of acidic hydrolases, resulting to
enzymatic digestion of proteins, RNA, DNA, and glycogen, and
cell dies by necrosis
6. DAMAGE TO DNA AND PROTEINS
- If damage is too severe, the cell initiates a suicide program that
results in death by apoptosis
NOTES:
- Atherosclerosis ischemia = decrease in oxygen supply as a result of decrease in
blood supply first to be affected is the aerobic respiration of mitochondria
decrease in oxidative phosphorylation decrease in ATP
- Increase in Glycolysis lactic acid formation decrease pH
- If oxygen is restored, all of these disturbances are REVERSIBLE
- Membrane damage central factor in pathogenesis of irreversible cell injury
A. ISCHEMIC/HYPOXIC INJURY
Most common type of cell injury in clinical medicine
Features of Ischemic/Hypoxic Injury:
1) Increased membrane permeability
2) Decreased mitochondrial function
- Cells aerobic respiration affected first (decreased
oxidative phosphorylation and decreased ATP generation)
Critical events leading to irreversible hypoxic cell injury:
1) Inability to reverse mitochondrial dysfunction upon
reoxygenation
2) Cell membrane damage - central factor in the pathogenesis
of ICI to irreversible cellular injury
- Calcium is an important mediator of biochemical changes
leading to cell death.
NOTES:
IRREVERSIBLE INJURY is associated morphologically with
1) SEVERE SWELLING of MITOCHONDRIA
2) EXTENSIVE DAMAGE to PLASMA MEMBRANE
3) SWELLING of LYSOSOMES
Leakage of intracellular enzymes and other proteins across the abnormally permeable
plasma membrane and into the blood provide important clinical indicators of cell
death
Elevated serum levels of CKMB and Troponin are early signs of MI and may be seen
before the infarct is detectable morphologically
ADDITIONAL:
Ischemia-Reperfusion Injury
- Under certain circumstances, when blood flow is restored to cells that have
been ischemic but have not died, injury is paradoxically exacerbated and
proceeds at an accelerated pace
- Reperfused tissues may sustain loss of cells in addition to the cells that are
irreversibly damaged at the end of ischemia
- New damaging processes during reperfusion:
4 of 8 |Page
a) New damage may be initiated by reoxygenation by oxygen free radicals
from infiltrating leukocytes
b) Ischemic injury is associated with inflammation, as a result of production
of cytokines and adhesion molecules by hypoxic parenchymal and
endothelial cells, which recruit circulating neutrophils
c) Activation of the complement system
Mechanism of Cell Injury by Activated Oxygen Species
Lipid Peroxidation Initiated by Hydroxyl Radical.
B. FREE RADICALS AND ACTIVATED OXYGEN SPECIES
Free radicals are chemical species that have a single unpaired
electron.
1) Superoxide (O2) - inactivated by superoxide dismutase
(SOD) or spontaneously
2) Hydrogen peroxide (H2O2)
3) Hydroxyl radicals
Main effect of O2 species
1) Lipid peroxidation
2) Oxidative modification of proteins: Formation of sulfhydryl
bonds of proteins causing abnormal protein folding
3) Mutation in genetic code
Fate of Free Radicals:
1) Spontaneous decay
2) Termination of inactivation of free radicals
a) Antioxidants block the initiation of free radical
formation and terminate radical damage
Vitamin E
Sulfhydryl containing compounds such as
cysteine, glutathione and D-penicillamine
Serum proteins such as ceruloplasmin, ferritin and
transferring which bind to free iron
b) Enzymes
Superoxide dismutase (SOD - H2O2 system)
converts O2 to H2O2
Catalase which decomposes H2O2 to O2 and H2O
Glutathione peroxidase catalyzes free radical
breakdown
They may be generated by:
Redox reactions during normal metabolic processes
Absorption of radiant energy
During inflammation via redox reactions which use NADPH
oxidase
Enzymatic metabolism of exogenous chemicals or drugs (ex.
CCl4)
Transition metals such as Fe and Cu
Nitric Oxide (NO)
C. CHEMICAL INJURY
This can be observed from a decrease in glucose leading to
electrolyte imbalance
1) DIRECTLY combine with molecular components or cellular
organelle. Example:
a. Mercuric chloride - Mercury binds to sulfhydryl groups of
the cell membrane proteins, causing an increase in
membrane permeability and inhibition of ATPase-dependent
transport; The greatest damage is usually to the cells that
use, absorb, excrete, or concentrate the chemicals (GI
TRACT and KIDNEY)
b. Cyanide - Poisons mitochondrial cytochrome oxidase and
this inhibits oxidative phosphorylation
***Many antineoplastic chemotherapeutic agents and antibiotic
drugs also induce cell damage by direct cytotoxic effects
2) INDIRECTLY - Chemicals not biologically active in their native
form and has to be converted to toxic metabolites which act on
target cells by:
a) Direct covalent binding to membrane protein and lipids
(cytochrome P-450 mixed function oxidases in the smooth
ER of the liver and other organs)
b) Formation of reactive free radicals and subsequent lipid
peroxidation (is the most important membrane injury result)
o Ex. CCl4-induced liver necrosis and fatty change occur
because conversion of CCl4 by cytochrome P450 to
CCl3 (a highly reactive free radical), which causes lipid
peroxidation and damages many cellular structures.
o Acetaminophen, an analgesic drug, is converted to a
toxic product during detoxification in the liver.
Large doses of acetaminophen/paracetamol
diminish reduced glutathione levels,
consequently decreasing free radical breakdown
Sequence of Events Leading to Fatty Change and Cell Necrosis in CCl4 Toxicity
D. VIRUS-INDUCED CELL INJURY
1. DIRECT CYTOPATHIC EFFECT - Replicating virus particles which
interfere with the cells metabolism, leading to cell damage such as:
Cell lysis
Cytoskeletal alterations
Syncytial or Multinucleated giant cells (ex. measles and
herpes virus)
Inclusion bodies contain virions or viral proteins;
intranuclear, intracytoplasmic or both
2. IMMUNE MEDIATED CELL INJURY
5 of 8 |Page
MORPHOLOGY OF INJURED CELLS
Reversible (non-lethal) Irreversible (lethal)
Cellular swelling. First manifestation.
Occurs when (a) cell is incapable of
maintaining fluid and ionic homeostasis
and (b) energy-dependent ion pumps on
plasma membrane have failed.
Fatty change. Occurs in hypoxic, toxic or
metabolic injury. Manifested as lipid
vacuoles.
Cell
necrosis/death
TWO MECHANISMS OF CELL DEATH:
A. NECROSIS
sum of morphologic changes that follow cell death in a living
tissue or organ, most commonly due to hypoxia
a major morphologic manifestations of ICI
with inflammation
Morphologic changes in cell death:
A. Nuclear changes:
1. Pyknosis (nuclear shrinkage and increased basophilia).
2. Karyorrhexis (pyknotic nucleus fragments).
3. Karyolysis (basophilia of chromatin fades).
B. Cytoplasmic changes: inc. eosinophilia; vacuolated, moth-eaten
appearance; calcification
- More glassy, homogeneous appearance.
- Dead cells appear as phospholipid masses called myelin figures.
Types of Necrosis
A. COAGULATION NECROSIS
o Most common type; protein denaturation
o Retain cell outline, lose nucleus acidophilic, opaque
"tombstone"
o Architecture of dead tissues is preserved for a span of at least
some days
Presumably, the injury denatures not only the structural
proteins but also enzymes and so block the proteolysis of
dead cells resulting to EOSINOPHILIC, ANUCLEATE CELLS
which may persist for days or weeks
Ultimately, the necrotic cells are removed by phagocytosis of
the cellular debris by infiltrating leukocytes and by digestion
of the dead cells by the action of lysosomal enzymes of
leukocytes
o Usually follows sudden, severe ischemia; ischemia by obstruction
of vessel may lead to this in all organs except the brain.
o Localized area of this necrosis called an infarct
o kidney, heart, adrenals, liver
B. LIQUEFACTION NECROSIS
o Involves digestion of dead cells leading to formation of liquid
viscous mass.
o Bacterial or fungal infections that stimulate leukocytes to
release hydrolytic enzymes in cells
o Autolysis and heterolysis prevail over protein denaturation
o Characteristic of brain infarct - reasons for the Liquefaction
necrosis of the brain are not yet known
o Pus - Creamy yellow material due to dead leukocytes
o Bacterial infection, suppuration, amebic liver abscess
C. FAT NECROSIS
o Not a specific pattern of necrosis.
o Involves fat destruction due to pancreatic lipases that
invaded the pancreas or peritoneal cavity.
Pancreatic enzymes leak out of acinar cells and liquefy
the membranes of fat cells in the peritoneum
The released lipases split the TG esters contained
within fat cells
The Fas, so derived, combine with Calcium to produce
grossly visible chalky-white areas(fat saponification) due
to combining of calcium with released fatty acids.which
enables the identification of the lesion
o Histologic examination: Takes form of shadowy outlines of
necrotic fat cells with basophilic Ca deposits surrounded by
an inflammatory reaction.
o Seen in (1) acute pancreatic necrosis (acute pancreatitis)
triglycerides from damaged fat cells decompose to produce
FFA which complex with calcium to form soap. (2) trauma
breasts
D. CASEOUS NECROSIS
o Collection of fragmented or lysed cells and amorphous
granular debris within an inflammatory border
o Coag. and liquefactive necrosis;
o Derived from the friable white appearance of the area of
necrosis characteristic of a focus of inflammation known
as a GRANULOMA
o Gross: soft, friable, whitish-gray (cheesy)
o Microscopic: amorphous, granular pinkish debris
o Most often in foci of tuberculous Infection; cheese-like
E. GANGRENOUS NECROSIS
o NOT a pattern of cell death.
o Coag. n. + liquefactive action of bacterial and WBCs
o Refers to part of body (usually a limb) that has lost its blood
supply and undergoes necrosis.
o Wet gangrene occurs if bacterial infection is present leading
to more liquefactive characteristics. (pred. Liquef., swollen,
red, odorous; intestine, AP, GB)
o Dry gangrene (pred. Coag. pattern; dry, black exts)
F. FIBRINOID NECROSIS
o CT and arterial walls are infiltrated by eosinophilic hyaline
material which shows some of characteristics of fibrin
o Occurs when complexes of ANTIGENS and ANTIBODIES
are deposited in the walls of arteries
o These deposits plus the leaked out fibrin, results in a bright
pink and amorphous appearance in H&E stains called the
FIBRINOID(fibrin-like)
o Seen in immunologically mediated vasculitis syndromes.
B. APOPTOSIS
Pathway of cell death that is induced by a tightly regulated suicide
program .
There is activation of enzymes that degrade the cells' own nuclear
DNA and nuclear and cytoplasmic proteins.
Breaks into fragments called apoptotic bodies.
Plasma membrane remains intact though altered to induce
phagocytosis of apoptotic bodies (as a result, no leaking occurs)
No ATP depletion.
Does not elicit inflammation.
Involved only in living tissue.
6 of 8 |Page
Physiologic Causes
For elimination of cells that are no longer needed as well as to
maintain steady population levels of cells in tissues
Embryogenesis involves programmed destruction of cells in
implantation, organogenesis, developmental involution, and
metamorphosis.
Involution of hormone-dependent tissues upon hormone
withdrawal such as endometrial cell breakdown during the
menstrual cycle.
Cell loss in proliferating cell populations (i.e. immature
lymphocytes in the bone marrow that do no express the right
antigen receptors)
Elimination of harmful self-reactive lymphocytes.
Pathologic Causes
For elimination of cells injured beyond repair.
DNA damage due to radiation, hypoxia, free radicals, etc.
Accumulation of misfolded proteins due to mutations in genes
leading to ER stress and then to apoptosis.
Cell death due to infections like viral infections (HIV) or immune
responses (viral hepatitis) via cytotoxic T lymphocytes
Pathologic atrophy in parenchymal organs due to duct obstruction
such as in the pancreas or kidney.
Morphology
1) Cell Shrinkage.
2) Chromatin condensation.- most characteristic feature of apoptosis
3) Formation of cytoplasmic blebs and membrane-bound apoptotic
bodies.
4) Phagocytosis of apoptotic cells or cells bodies by macrophages
(phagocytes).
Biochemical Features
Activation of Caspases
o Caspases are a family of cysteine proteases that activate
apoptosis.
o Caspase-8 and caspase-9 are initiators
o Caspase-3 and caspase-6 are executioners.
o Must undergo enzymatic cleavage to become active.
DNA and Protein Breakdown
o Calcium and magnesium dependent endonuclease activity
breaks down DNA.
o Changes of movement of phospholipids on the membrane
(i.e. phosphatidylserine) from inner to outer leaflet are
recognized by phagocytes.
Mechanism
Initiation Phase
Caspases become catalytically active.
Occurs via intrinsic (mitochondrial) and extrinsic (death receptor-
initiated) pathways.
o Intrinsic Pathway.
Involves release of mitochondrial proteins (i.e.
cytochrome c) into cytoplasm intiating apoptosis.
Anti-apoptotic Bcl family regulates release.
Sensor proteins Bim, Bid, and Bad detect stress or
damage and activate pro-apoptotic effectors Bax and
Bak which allows mitochondrial leakage.
Cytochrome c binds to Apaf-1 forming an apoptosome
that binds to initiator caspase-9 producing an auto-
amplification process of the pathway.
IAP (inhibitors of apoptosis) are inhibited.
o Extrinsic Pathway
Involves death receptors (TNF receptor family) such as
TNFR1 and Fas (CD95).
Ligands attach to receptors forming cytoplasmic death
domains forming binding sites. Adapter proteins bind to
these sites and their death domains bind to and allows
activation of caspase-8 (caspse-10 in humans) leading
to apoptosis.
Execution Phase
Executioner caspases (caspase-3 and -6) are activated by either
initiation pathways and degrade cytoplasmic structures and DNA.
Dysregulated Apoptosis
Defective apoptosis allows increased cell survival.
o May involve mutation in p53 which fails to activate cell death
leading to an increase in potentially harmful cells like
lymphocytes.
Increased apoptosis leads to excessive cell death.
o Neurodegenerative diseases involving loss of neurons
caused by mutations or misfolded proteins.
o Ischemic injury.
o Virus-infected cells.
NECROSIS AND APOPTOSIS
Necrosis Apoptosis
Stimuli Hypoxia, toxins Physiologic & Pathologic
Histology Cellular swelling
Coagulation necrosis
Disruption of
organelles
Single cells shrinkage
Chromatin condensation
Apoptic bodies
DNA Breakdown Random, diffuse Internucleosomal
Mechanisms ATP depletion
Membrane injury
Free radical damage
Gene activation
Endonuclease
Tissue Reaction Inflammation No inflammation
Phagocytosis of
apoptotic bodies
FEATURES
Cell size Enlarged (swelling) Reduced (shrinkage)
Nucleus Pyknosis
karyorrhexis
karyolysis
Fragmentation into
nucleosome-size
fragments
Plasma
membrane
Disrupted Intact; altered structure,
especially orientation of
lipids
Cellular contents Enzymatic digestion;
may leak out of cell
Intact; may be released
in apoptotic bodies
Adjacent
inflammation
Frequent No
Physiologic or
pathologic role
Invariably pathologic
(culmination of
irreversible cell
injury)
Often physiologic,
means of eliminating
unwanted cells; may be
pathologic after some
forms of cell injury,
especially DNA damage
CELL INJURY: SUBCELLULAR ALTERATION
A. Lysosomal catabolism
o Primary Lysosomes
Membrane-bound intracellular organelles containing
a variety of hydrolytic enzymes which are synthesized
by RER and packaged into vesicles by Golgi
apparatus
o Secondary Lysosomes
Fusion of primary lysosomes and membrane-bound
vacuoles containing material to be digested
Phagolysosomes
Ways to Breakdown Phagocytosed Material:
Heterophagy
- Materials from the external environment are taken up
through endocytosis, phagocytosis and pinocytosis
- Process commonly exhibited by neutrophils and
macrophages
Autophagy
- Removal of damaged organelles during cell injury and
cellular differentiation
- Common in cells undergoing atrophy due to nutritional
deprivation or hormonal involution
7 of 8 |Page
B. Hypertrophy of SER - Adaptive response to medications
C. Mitochondrial alterations - Changes in size, shape and number
D. Abnormalities of cytoskeleton, contractile proteins, membrane
skeleton
Chediak-Higashi syndrome defective microtubule polymerization
Colchicine disrupts microtubules, blocks mitosis in metaphase
Cytochalasin B inhibits microfilament action and phagocytosis
Immotile cilia syndrome microtubule defect in respiratory cilia
Intermediate filament accumulations Mallory body (alcoholic
hyaline in liver) and neurofibrillary tangle (brain in Alzheimer's disease)
E. Membrane skeleton - Seen in hereditary spherocytosis
INTRACELLULAR ACCUMULATIONS
- Sign of metabolic derangement due to abnormal amounts of
various substances
- Four major mechanisms:
Abnormal metabolism metabolic rate inadequate to remove
normal substance (ex. fatty change)
Enzyme deficiency one can't metabolize certain substances
(ex. inborn errors of metabolism like glycogen storage diseases)
Inability to degrade or transport abnormal exogenous
substances (ex. hemosiderin, carbon pigments, silica) to
other sites
Defects in protein folding and an inability to degrade the
abnormal protein efficiently. (ex. Accumulation of mutated
1-antitrypsin in liver cells, various mutated proteins in the
degenerative disorders of the CNS)
A. Lipids
Fatty Change
o Steatosis
Abnormal accumulation of triglycerides within the
parenchymal cell
Often seen in the liver since it is the major organ
involved in fat metabolism, but may also be seen in
the heart, muscle and kidney.
Fatty change appears as clear vacuoles
o Causes:
Alcohol most common cause in adults
Protein malnutrition
Diabetes Mellitus
Pregnancy acute fatty change
Obesity
Some chronic diseases
Hepatotoxins
Anoxia
o Mechanism:
Excessive entry of FFA into the liver brought about
by starvation and corticosteroids
Increased esterification of FA to triglycerides due
to:
increased alpha-glycerophosphate (ex.
alcohol poisoning)
enhanced FA synthesis
decrease in FA oxidation
Reduced apoprotein synthesis leading to a
decrease in fat mobilization (ex. CCl4 and protein
malnutrition)
Impaired lipoprotein secretion from the liver (oretic
acid)
Other lipid accumulations:
1. Atherosclerosis
- Smooth muscle cells and macrophages in the
intima and large arteries are filled with lipid
vacuoles. These may rupture and release lipid into
the extracellular space. Extracellular esters may
crystallize in the shape of long needles, producing
distinctive clefts in tissue sections.
2. Xanthomas
- clusters of foamy cells in the subepithelial
connective tissue of the skin and in tendons which
form tumorous masses
3. Cholesterolosis
- Focal accumulations of cholesterol-laden
macrophages in the lamina propria of the
gallbladder. Mechanism of accumulation is unknown
4. Niemann-Pick disease, type C
- A lysosomal storage disease
- Caused by mutations affecting an enzyme involved
in cholesterol trafficking
- Results in cholesterol accumulation in multiple
organs
B. Proteins
- Protein accumulations appear as rounded eosinophillic
droplets in the cytoplasm.
- Abnormal proteins deposit primarily in extracellular spaces in
some disorders such as amyloidosis
- Causes of morphologically visible protein accumulation:
Reabsorption droplets in the proximal renal tubules
o In disorders with heavy protein leakage
across the glomerular filter, renal absorption
of protein into vesicles is increased.
o Protein appears as pink hyaline droplets
within the cytoplasm of the tubular cell.
*this process is reversible. Once the proteinuria (protein loss
in the urine) diminishes, the protein droplets are metabolized
and disappear.
Defective intracellular transport and secretion of
critical proteins
Accumulation of cytoskeletal proteins
Aggregation of abnormal proteins
o Abnormal or misfolded proteins may deposit in
tissues interfere with normal functioning
o Deposits may be intracellular, extracellular, or
both, and may directly or indirectly be the
cause of the pathologic changes.
C. Glycogen
- Excessive deposits of glycogen are due to an abnormality in
either glucose or glycogen metabolism
- Glycogen appears as clear vacuoles within the cytoplasm.
- Causes:
1. Diabetes Mellitus
Glycogen is found in renal epithelial cells of PCT,
liver cells, beta cells of islets of Langerhans and
heart muscle.
2. Glycogen storage diseases or Glycogenoses
Enzymatic defects in the synthesis or breakdown of
glycogen result to massive accumulation of glycogen
and, eventually, cell injury and death.
Ex. Von Gierke's disease, McArdle's syndrome,
Pompe's disease
D. Pigments
- Colored substances, some of whichare normal constituents
of the cell (eg.melanin), wheras others are abnormal and
accumulate in cells only under special circumstances
Exogenous Pigments
Carbon
o Ubiquitous air pollutant
8 of 8 |Page
o Picked up by macrophages within the alveoli and
transported to lymph nodes at the tracheobronchial
region
o Accumulation causes anthracosis in lungs and
involved lymph nodes
Blackened lung tissue
Tattoo
o Localized pigmentation of the skin
o Inoculated pigments are phagocytosed by dermal
macrophages which reside in the skin for the duration
of the persons life.
o The pigments do not usually evoke any inflammatory
response
Endogenous pigments
1. Lipofuscin
is an insoluble pigment, also known as lipochrome or
wear and tear (aging) pigment
Appears as a yellow brown, finely granular cytoplasmic,
often perinuclear pigment
Composed of polymers of lipids and phospholipids in
complex with protein
Important as a telltale sign of free radical injury and lipid
peroxidation
Seen in cells undergoing slow, regressive changes and
is particularly prominent in the liver and heart of patients
with severe malnutrition and cancer cachexia
2. Melanin
is an endogenous, non hemoglobin derived, brown black
pigment formed when tyrosinase catalyzes the oxidation of
tyrosine to dihydroxyphenylalanine in melanocytes
is the only endogenous brown-black pigment
3. Hemosiderin
Is a hemoglobin derived, golden yellow to brown, granular or
crystalline pigment that serves as one of the major forms of
iron
In cells, iron is stored in association with a protein,
apoferritin, to form ferritin micelles .When there is local or
systemic excess of iron, ferritin forms henosiderin granules
which are easily seen in the light microscope
Hemosiderosis - systemic overload of iron, wherein
hemosiderin is deposited in many organs and
tissues
Does not damage the parenchynmal cells or
impair organ functions
main causes are: increased absorption of dietary
iron, hemolytic anemia, and repeated blood
transfusions .
hemochromatosis most extreme accumulation of
iron associated with liver, hear and pancreatic
damage resulting in liver fibrosis, heart failure and
diabetes mellitus.
4. Bilirubin
Normal major pigment found in bile.
Derived from hemoglobin but contains no iron
Evident in liver and kidney
Jaundice: common clinical disorder caused by excesses of
bilirubin within cells and tissues
Kernicterus : damage to the brain centers of infants
caused by increased levels of unconjugated bilirubin.
5. Hematin (Hemozoin)
o Also called malaria pigment
o Found in the liver and spleen
o (-) Prussian Blue
6. Copper
o Toxic levels of copper accumulate in Wilsons
disease, causing hepatolenticular degeneration
PATHOLOGIC CALCIFICATION- is the abnormal tissue deposition of
calcium salts together with smaller amounts of iron, magnesium and
other mineral salts
Dystrophic Calcification:
Deposition usually occurs in dying tissues. Encountered in areas
of necrosis, whether they are coagulative, casous, or liquefactive
type and in the foci of enzymatic necrosis of fat.
Occurs despite normal serum levels of calcium and in the absence
of derangements in calcium metabolism.
Calcium salts appear macroscopically as fine, white granules or
clumps and often felt as gritty deposits
Always present in atheromas of advanced atherosclerosis
Pathogenesis:
Ca
2+
is concentrated in membrane-bound vesicles through a
process initiated by membrane damag
o Ca
2+
binds to phospholipids present in the vesicle
membrane.
o Phosphates associated with the membrane generate
phosphate groups which bind to Ca
2+
.
o Calcium-phosphate binding repeats.
o Structural changes occur in the arrangement of calcium and
phosphate groups generating a microcrystal which then
propagate and lead to MORE calcium deposition
Eg. Psammoma bodies seen in mesothelioma
Metastatic Calcification
May occur in normal tissues whenever there is hypercalcemia
Can occur widely throughout the body but principally affects the
interstitial tissues of the gastric mucosa, kidneys, lungs, systemic
arteries and pulmonary veins.
In all these sites calcium salts morphologically resemble
those described in dystrophic calcification. They may occur
as noncrystalline amorphous deposits or at times
hydroxyapatite crystals
May cause massive deposits in the kidney (nephrocalcinosis)
may cause renal damage
Hyaline Change
An alteration within or in the extracellular space which gives a
homogenous, glassy, pink appearance in routine H&E sections
A. Intracellular
Can be observed in the proximal convoluted tubules,
Russell bodies, viral inclusions and alcoholic hyaline
B. Extracellular
Examples are scars, hyaline arteriosclerosis, hyalinized
glomeruli (chronic renal disease) and amyloid (positive
Congo red stain; bipolar refringence)
In long term hypertension, the walls of arterioles,
especially in the kidney, become hyalinized.
CELLULAR AGING
Is the result of a progressive decline in cellular function and
viability caused by genetic abnormalities and the accumulation of
cellular and molecular damage due to effects of exogenous
influences.
The balance between cumulative metabolic damage and the
response to that damage could determine the rate at which we
age.
The known changes that contribute to cellular aging include:
o Decreased cellular replication
o After a fixed number of divisions, all somatic cells
become arrested in a terminally non dividing state
known as senescence .
o Accumulation of metabolic and genetic damage
Eg . reactive oxygen species, byproducts of oxidative
phosphorylation , causes covalent modification of
proteins lipids and nucleic acids .
Increased oxidative damage could result from repeated
environmental exposure to such influences as ionizing
radiation, mitochondrial dysfunction or reduction of
antioxidant defense mechanisms with age.
You might also like
- Pathology Cell InjuryDocument57 pagesPathology Cell InjuryMajd MustafaNo ratings yet
- Introduction to Pathology: Study of Disease at the Cellular LevelDocument44 pagesIntroduction to Pathology: Study of Disease at the Cellular Level53-Deepankar SutradharNo ratings yet
- Cell Injury & AdaptationDocument22 pagesCell Injury & AdaptationUmam LoyalNo ratings yet
- Cellular Adaptations and Cell DeathDocument4 pagesCellular Adaptations and Cell DeathShuaib SiddiquiNo ratings yet
- Apoptosis PPT, Pathological AnatomyDocument15 pagesApoptosis PPT, Pathological AnatomyN J3 CNo ratings yet
- Hemodynamic Disorders, Thromboembolic Disease, and ShockDocument87 pagesHemodynamic Disorders, Thromboembolic Disease, and Shockgifty100% (1)
- Neoplasia I - RecordingDocument6 pagesNeoplasia I - RecordingIS99057No ratings yet
- Metabolic Response To InjuryDocument55 pagesMetabolic Response To InjuryMuhammad NaveedNo ratings yet
- General Pathology 1Document39 pagesGeneral Pathology 1Muheto100% (2)
- REVERSIBLE CELL INJURY AND ADAPTATION MECHANISMSDocument25 pagesREVERSIBLE CELL INJURY AND ADAPTATION MECHANISMSISRAELNo ratings yet
- 07 Pathological ClacificationDocument10 pages07 Pathological Clacificationraanja2No ratings yet
- 14 Patho Head and Neck PathologyDocument10 pages14 Patho Head and Neck PathologyMartin TanNo ratings yet
- Causes, Types and Morphology of Cell Injury and DeathDocument18 pagesCauses, Types and Morphology of Cell Injury and DeathYoja GarzonNo ratings yet
- PECOMADocument25 pagesPECOMAAnan JaiswalNo ratings yet
- Cell Injury Notes-1Document63 pagesCell Injury Notes-1Stefanie VirniaNo ratings yet
- Dr. Manella Joseph Senior Lecturer/Consultant PathologistDocument41 pagesDr. Manella Joseph Senior Lecturer/Consultant PathologistNipun Shamika100% (1)
- 1.05 Qualitative 0 Quantitative Platelet DisordersDocument9 pages1.05 Qualitative 0 Quantitative Platelet DisordersShiena ArchividoNo ratings yet
- Pathology - Lab: Pathology of The Blood VesselsDocument5 pagesPathology - Lab: Pathology of The Blood VesselsRazel PerezNo ratings yet
- Chronic Inflammation Guide: Causes, Types & PathogenesisDocument47 pagesChronic Inflammation Guide: Causes, Types & PathogenesisPradeepNo ratings yet
- Inflammation CCDocument72 pagesInflammation CCmulatumeleseNo ratings yet
- Hemodynamic Disorders, Thromboembolic DiseasesDocument100 pagesHemodynamic Disorders, Thromboembolic DiseasesKate Alyssa Caton100% (1)
- Robbins Ch. 19 The Pancreas Review QuestionsDocument3 pagesRobbins Ch. 19 The Pancreas Review QuestionsPA2014100% (1)
- Pathology of Urinary SystemDocument384 pagesPathology of Urinary SystemNzau MuangeNo ratings yet
- Neoplasia Exam ReviewDocument37 pagesNeoplasia Exam ReviewSaWera AsGhar100% (1)
- General Pathology Lecture 1 Introduction & Cell InjuryDocument57 pagesGeneral Pathology Lecture 1 Introduction & Cell InjuryMaebelene MeloNo ratings yet
- Introduction to Pathology: The Four Aspects of DiseaseDocument21 pagesIntroduction to Pathology: The Four Aspects of DiseaseKC White Dela RosaNo ratings yet
- Granulomatous Inflammation ThyroidDocument55 pagesGranulomatous Inflammation ThyroidKamlesh PrajapatiNo ratings yet
- Aplastic AnaemiaDocument21 pagesAplastic AnaemiaAbhinav ReddyNo ratings yet
- The Kidney: Glomerular Diseases, Nephrotic Syndrome, and Acute Renal FailureDocument11 pagesThe Kidney: Glomerular Diseases, Nephrotic Syndrome, and Acute Renal FailureElina Drits100% (1)
- Trans 1 - Cells As A Unit of Health and DiseaseDocument12 pagesTrans 1 - Cells As A Unit of Health and DiseaseCedrick BunaganNo ratings yet
- Necrosis & ApoptosisDocument6 pagesNecrosis & ApoptosisshabeebakpNo ratings yet
- Gastrointestinal PathologyDocument14 pagesGastrointestinal PathologyRahul ShuklaNo ratings yet
- PericarditisDocument3 pagesPericarditisKhalid Mahmud Arifin0% (1)
- Chapter 10 - Diseases of Infancy and ChildhoodDocument17 pagesChapter 10 - Diseases of Infancy and ChildhoodAgnieszka WisniewskaNo ratings yet
- Neoplasia: Basic PathologyDocument12 pagesNeoplasia: Basic Pathologyandroid tricksNo ratings yet
- Neoplasm Lecture NotesDocument6 pagesNeoplasm Lecture Notesmiguel gaquitNo ratings yet
- Chapter 13 Neoplastic Proliferations of White CellsDocument16 pagesChapter 13 Neoplastic Proliferations of White CellsOmar100% (1)
- Mendelian Disorders: Classification, Examples and Inheritance PatternsDocument8 pagesMendelian Disorders: Classification, Examples and Inheritance PatternsAmna BaigNo ratings yet
- Pathology Slides 1Document28 pagesPathology Slides 1SydNo ratings yet
- Diseases of ImmunityDocument13 pagesDiseases of ImmunityRose AnnNo ratings yet
- Patho A 1. 5 Hemodynamic Disorders (Bongat, 2015)Document12 pagesPatho A 1. 5 Hemodynamic Disorders (Bongat, 2015)Grant GarcesNo ratings yet
- Hemodynamic DisordersDocument85 pagesHemodynamic DisordersTofik Mohammed100% (1)
- Ch.1 Baby Robbins OutlineDocument11 pagesCh.1 Baby Robbins OutlinePA2014100% (3)
- Genetic and Pediatric Diseases Chapter SummaryDocument16 pagesGenetic and Pediatric Diseases Chapter SummaryJustine HungNo ratings yet
- 2021 Systemic Pathology S4T1 - RBC and Bleeding Disorders PDFDocument27 pages2021 Systemic Pathology S4T1 - RBC and Bleeding Disorders PDFAlexis Bondad100% (1)
- Chapter 11 Blood Vessels 8th Ed NotesDocument7 pagesChapter 11 Blood Vessels 8th Ed NotesKyle Christopher SiaNo ratings yet
- Fatty Liver (1) - General PathologyDocument22 pagesFatty Liver (1) - General PathologyDarien LiewNo ratings yet
- The Immune Response Defends Against InfectionDocument55 pagesThe Immune Response Defends Against InfectionKoushali BanerjeeNo ratings yet
- Hypersensitivity ReactionsDocument25 pagesHypersensitivity Reactionsbpt2100% (3)
- Inflammatory Response and Vascular Permeability ChangesDocument20 pagesInflammatory Response and Vascular Permeability Changesjeffaguilar100% (2)
- Pathology Lecture 3, Cell Injury (Continued) (Lecture Notes)Document11 pagesPathology Lecture 3, Cell Injury (Continued) (Lecture Notes)Ali Al-Qudsi100% (3)
- WBC Pathology: Lecturer: Associate Professor T. A. GrekovaDocument49 pagesWBC Pathology: Lecturer: Associate Professor T. A. GrekovaFaheem MusthafaNo ratings yet
- General Pathology Bimonthly Exam Compilation Updated 2Document197 pagesGeneral Pathology Bimonthly Exam Compilation Updated 2Cherry Rahima100% (1)
- Lecture 3. Bleeding Disorders Part 1Document31 pagesLecture 3. Bleeding Disorders Part 1Kekelwa Mutumwenu Snr100% (1)
- General PathologyDocument4 pagesGeneral PathologyNudžejma KadrićNo ratings yet
- Differential Diagnosis of Painful Genital LesionsDocument7 pagesDifferential Diagnosis of Painful Genital LesionsKyra KhalidNo ratings yet
- Pathology (3) Adaptation and Cellular DepostitionDocument50 pagesPathology (3) Adaptation and Cellular Depostitionnurul dwi ratihNo ratings yet
- A) Cell Atrophy:: Cellular AdaptationDocument7 pagesA) Cell Atrophy:: Cellular AdaptationHamid AlyadumiNo ratings yet
- Dressings and BandagesDocument15 pagesDressings and BandagesJessica Febrina WuisanNo ratings yet
- Acetabular FractureDocument8 pagesAcetabular FractureJessica Febrina WuisanNo ratings yet
- Juvenile Deliquency ResearchDocument14 pagesJuvenile Deliquency ResearchJessica Febrina WuisanNo ratings yet
- PalpitationsDocument3 pagesPalpitationsJessica Febrina WuisanNo ratings yet
- Anatomy LarynxDocument4 pagesAnatomy LarynxJessica Febrina Wuisan100% (1)
- Development and MilestonesDocument1 pageDevelopment and MilestonesJessica Febrina WuisanNo ratings yet
- Bio EnergeticsDocument4 pagesBio EnergeticsJessica Febrina WuisanNo ratings yet
- MalariaDocument5 pagesMalariaJessica Febrina WuisanNo ratings yet
- Nervous TissueDocument6 pagesNervous TissueJessica Febrina WuisanNo ratings yet
- MuscleDocument5 pagesMuscleJessica Febrina WuisanNo ratings yet
- Nervous SystemDocument6 pagesNervous SystemJessica Febrina WuisanNo ratings yet
- GlycolysisDocument7 pagesGlycolysisJessica Febrina WuisanNo ratings yet
- Fluid and Hemodynamics Review ManualDocument28 pagesFluid and Hemodynamics Review ManualJessica Febrina WuisanNo ratings yet
- EndometriosisDocument4 pagesEndometriosisJessica Febrina WuisanNo ratings yet
- Mood DisorderDocument6 pagesMood DisorderJessica Febrina WuisanNo ratings yet
- Acid Base DisordersDocument2 pagesAcid Base DisordersJessica Febrina WuisanNo ratings yet
- Hemostatic FunctionDocument9 pagesHemostatic FunctionJessica Febrina WuisanNo ratings yet
- Physical Examination of The SkinDocument4 pagesPhysical Examination of The SkinJessica Febrina WuisanNo ratings yet
- Wound Care, Dressing and BandagingDocument11 pagesWound Care, Dressing and BandagingJessica Febrina Wuisan100% (1)
- Dermatologic Exam GuideDocument3 pagesDermatologic Exam GuideJessica Febrina WuisanNo ratings yet
- Gait, Arms, Legs, Spine TestDocument2 pagesGait, Arms, Legs, Spine TestJessica Febrina WuisanNo ratings yet
- FeverDocument7 pagesFeverJessica Febrina WuisanNo ratings yet
- Sex Differences and Hemoglobin Levels in Relation To Stroke OutcomesDocument5 pagesSex Differences and Hemoglobin Levels in Relation To Stroke OutcomesJessica Febrina WuisanNo ratings yet
- Acid Base DisordersDocument2 pagesAcid Base DisordersJessica Febrina WuisanNo ratings yet
- CNS Tumors: Adult Tumors Tend To Be Supratentorial (70%) Pediatric Tumors Tend To Be Infratentorial (70%)Document8 pagesCNS Tumors: Adult Tumors Tend To Be Supratentorial (70%) Pediatric Tumors Tend To Be Infratentorial (70%)Jessica Febrina WuisanNo ratings yet
- Acid Base DisordersDocument2 pagesAcid Base DisordersJessica Febrina WuisanNo ratings yet
- Principles in Plastic SurgeryDocument3 pagesPrinciples in Plastic SurgeryJessica Febrina WuisanNo ratings yet
- Basic Principles in TraumaDocument5 pagesBasic Principles in TraumaJessica Febrina WuisanNo ratings yet
- DYSPNEADocument2 pagesDYSPNEAJessica Febrina WuisanNo ratings yet
- MycobacteriaDocument6 pagesMycobacteriaJessica Febrina WuisanNo ratings yet
- Nest Installation GuideDocument8 pagesNest Installation GuideOzzyNo ratings yet
- 1296169395JH 122310 - WebDocument12 pages1296169395JH 122310 - WebCoolerAdsNo ratings yet
- List of Computer Networking DevicesDocument1 pageList of Computer Networking Deviceskamit17102900100% (1)
- Unit explores Christian morality and conscienceDocument1 pageUnit explores Christian morality and conscienceRose Angela Mislang Uligan100% (1)
- VGHV NBV GH fc7fvbn BN NGCJHGDocument16 pagesVGHV NBV GH fc7fvbn BN NGCJHGRahul GNo ratings yet
- Paradise Pools Flyer With Price ListDocument5 pagesParadise Pools Flyer With Price ListKhuzaima HussainNo ratings yet
- Raspberry PiDocument19 pagesRaspberry PiAnonymous E4Rbo2s100% (1)
- Tween 80Document11 pagesTween 80fvdxrgNo ratings yet
- F588 PDFDocument8 pagesF588 PDFOscar Gutiérrez-JuncoNo ratings yet
- Applied Acoustics: André M.N. Spillere, Augusto A. Medeiros, Julio A. CordioliDocument13 pagesApplied Acoustics: André M.N. Spillere, Augusto A. Medeiros, Julio A. CordioliAbdelali MoumenNo ratings yet
- Borneo SporenburgDocument2 pagesBorneo SporenburgDorin TecuceanuNo ratings yet
- Monsterology Activity KitDocument2 pagesMonsterology Activity KitCandlewick PressNo ratings yet
- Protreat Hydro EngrgDocument6 pagesProtreat Hydro EngrgAmitkumar SinghNo ratings yet
- 1.1 Hardware and SoftwareDocument13 pages1.1 Hardware and SoftwareNurhanis SyazwaniNo ratings yet
- Mar For M: I MMQ SeriesDocument28 pagesMar For M: I MMQ SeriesIpal Febri NartaNo ratings yet
- 1830PSS R36 QuickReferenceGuide 8DG60888JAAADocument66 pages1830PSS R36 QuickReferenceGuide 8DG60888JAAAFelippe CanatoNo ratings yet
- Great Gatsby Study NotesDocument69 pagesGreat Gatsby Study NotesLara Westwood100% (2)
- Large and Medium Manufacturing Industry Survey Report 2018Document778 pagesLarge and Medium Manufacturing Industry Survey Report 2018melakuNo ratings yet
- Home Contents Vehicle Boat Cover Policy Sample Westpac NZDocument27 pagesHome Contents Vehicle Boat Cover Policy Sample Westpac NZRobin Rutter-BaumannNo ratings yet
- Outline 1. Background of Revision: JEITA CP-1104BDocument4 pagesOutline 1. Background of Revision: JEITA CP-1104BkksdnjdaNo ratings yet
- Fodor Hungary Between East and WestDocument22 pagesFodor Hungary Between East and WestFatih YucelNo ratings yet
- MACRO-ETCHING SOLUTIONS FOR ALUMINIUM ALLOYSDocument1 pageMACRO-ETCHING SOLUTIONS FOR ALUMINIUM ALLOYSsensoham03No ratings yet
- History of Cancer ChemotherapyDocument9 pagesHistory of Cancer ChemotherapyJoydeep MajumdarNo ratings yet
- Hobby 01: COD. 9942062.01 REV. 00Document9 pagesHobby 01: COD. 9942062.01 REV. 00Alexander SharamiginNo ratings yet
- Integrating Kuleana Rights and Land Trust PrioritesDocument50 pagesIntegrating Kuleana Rights and Land Trust PrioritesKeala Rodenhurst-JamesNo ratings yet
- Mahindra Powertrain - Market StrategyDocument4 pagesMahindra Powertrain - Market StrategyEshan KapoorNo ratings yet
- English Qualification TestDocument5 pagesEnglish Qualification TestMoh FaisolNo ratings yet
- Grammarism Ed Ing Adjectives Test 4 1189424Document2 pagesGrammarism Ed Ing Adjectives Test 4 1189424Royal Stars Drama AcademyNo ratings yet
- Kelas 1 AlphabetTITLE Kelas 1 Numbers ConversationTITLE Kelas 2 Feelings Body PartsDocument54 pagesKelas 1 AlphabetTITLE Kelas 1 Numbers ConversationTITLE Kelas 2 Feelings Body PartsArti Hikmatullah Perbawana Sakti BuanaNo ratings yet
- Purification of Morphologically and Functionally Intact Human Basophils To Near HomogeneityDocument9 pagesPurification of Morphologically and Functionally Intact Human Basophils To Near HomogeneitySinaí GutierrezNo ratings yet