Professional Documents
Culture Documents
Why Is The Ocean Salty
Uploaded by
Christina Jayun JangOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Why Is The Ocean Salty
Uploaded by
Christina Jayun JangCopyright:
Available Formats
Why is the ocean salty?
Ocean water is salty because it contains high concentrations of dissolved minerals known collectively as salts. Around 3.5% of ocean water is comprised of salts, depending on where in the world one is; equatorial waters tend to be saltier, while northern waters are slowly becoming fresher. There are a number of factors which make the ocean salty, and scientists are very interested in the salt content of the ocean, because it contributes to the flow of currents through the ocean, in a process known as thermohaline circulation. One of the reasons that the ocean is salty has to do with the ocean's floor, which contains a huge assortment of minerals and dissolved organic material which is slowly eroded and stirred up by the movements of the ocean. As the ocean eats away at the ocean floor, it increases the salt content. The ocean floor is also constantly renewing itself, another way to make the ocean salty, as seafloor spreading releases even more dissolved minerals into the water, in the form of emissions from hydrothermal vents and cracks in the seafloor. Another thing that makes the ocean salty is the water runoff which pours into the ocean. This might seem counterintuitive to you, as rivers, streams, and lakes probably taste fresh to you. However, this water also contains dissolved salts, although the concentration is lower than that in the ocean. These salts make their way to the ocean with the river water, which eventually evaporates from the ocean to fall back to Earth again as rain, repeating the process. The reason these salts do not build up in things like lakes is because inland bodies of water have an outlet. What makes the ocean salty is the slow concentration of salts over time, because the salts in the ocean have nowhere to go. Lakes and streams, on the other hand, are constantly recalculating their water. To find out what happens when a lake has no outlet, you can look at the Dead Sea, which has a famously high concentration of salts.
HOW SALTY IS THE OCEAN?
How salty the ocean is, however, defies ordinary comprehension. Some scientists estimate that the oceans contain as much as 50 quadrillion tons (50 million billion tons) of dissolved solids. If the salt in the sea could be removed and spread evenly over the Earth's land
surface it would form a layer more than 500 feet thick, about the height of a 40story office building. The saltiness of the ocean is more understandable when compared with the salt content of a fresh-water lake. For example, when 1 cubic foot of sea water evaporates it yields about 2.2 pounds of salt, but 1 cubic foot of fresh water from Lake Michigan contains only one one-hundredth (0.01) of a pound of salt, or about one sixth of an ounce. Thus, sea water is 220 times saltier than the fresh lake water. What arouses the scientist's curiosity is not so much why the ocean is salty, but why it isn't fresh like the rivers and streams that empty into it. Further, what is the origin of the sea and of its "salts"? And how does one explain ocean water's remarkably uniform chemical composition? To these and related questions, scientists seek answers with full awareness that little about the oceans is understood.
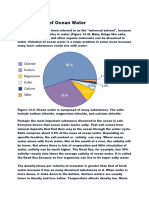
Illustration: Sources of salts in the ocean.
THE SALTIEST WATER
The saltiest water occurs in the Red Sea and the Persian Gulf, where rates of evaporation are very high. Of the major oceans, the North Atlantic is the saltiest. Within the North Atlantic, the saltiest part is the Sargasso Sea, an area of about 2 million square miles, located about 2,000 miles west of the Canary Islands. The Sargasso Sea is set apart from the open ocean by floating brown seaweed "sargassum" from which the sea gets its name. The saltiness of this sea is due in part to the high water temperature (up to 83 F) causing a high rate of evaporation and in part to its remoteness from land; because it is so far from land, it receives no fresh-water inflow. Low salinities occur in polar seas where the salt water is diluted by melting ice and continued precipitation. Partly landlocked seas or coastal inlets that receive substantial runoff from precipitation falling on the land also may have low salinities.
You might also like
- Primary Water TheoryDocument7 pagesPrimary Water TheoryPrimary Water100% (15)
- Seawater Density & SalinityDocument3 pagesSeawater Density & SalinityKarthik MathivananNo ratings yet
- Oceans and OceanographyDocument13 pagesOceans and OceanographymkprabhuNo ratings yet
- Watercycle (Streams, Rivers, Lakes and Oceans): 2nd Grade Science Workbook | Children's Earth Sciences Books EditionFrom EverandWatercycle (Streams, Rivers, Lakes and Oceans): 2nd Grade Science Workbook | Children's Earth Sciences Books EditionNo ratings yet
- A Simple Experiment Illustrates This. Fill Three Glasses With Water From TheDocument3 pagesA Simple Experiment Illustrates This. Fill Three Glasses With Water From TheRay Jhon OrtizNo ratings yet
- 7 - Why Is The Ocean SaltyDocument7 pages7 - Why Is The Ocean SaltyA dumNo ratings yet
- Why Is The Ocean SaltyDocument2 pagesWhy Is The Ocean SaltyintermazeNo ratings yet
- Why Sea Water Is Salty?Document7 pagesWhy Sea Water Is Salty?jakkiNo ratings yet
- GhadaDocument2 pagesGhadaNoso CrestyNo ratings yet
- Online AssignmentDocument9 pagesOnline AssignmentgeethaNo ratings yet
- Online Assignment PreethuiDocument9 pagesOnline Assignment PreethuigeethaNo ratings yet
- Why Is The Sea SaltyDocument2 pagesWhy Is The Sea SaltyBudiSetiawanNo ratings yet
- Why Seawater is SaltyDocument2 pagesWhy Seawater is SaltynvndrptrNo ratings yet
- Group 4: Dela Torre, Krishia Mae Lacanaria, John Theo Maniti, Joshua Assignment 6 Water Salinity Definition of SalinityDocument4 pagesGroup 4: Dela Torre, Krishia Mae Lacanaria, John Theo Maniti, Joshua Assignment 6 Water Salinity Definition of SalinityJoshua ManitiNo ratings yet
- SeawaterDocument7 pagesSeawateryusufeka178No ratings yet
- OceanDocument21 pagesOceanRajalaxmi pNo ratings yet
- Estuaries, Fjords & Saltwater EcosystemsDocument12 pagesEstuaries, Fjords & Saltwater EcosystemsJohn Lloyd SalinasNo ratings yet
- Dissolved GassesDocument9 pagesDissolved GassesVictoria Ann TasiniNo ratings yet
- "The Oceans": Group 5 OceanographyDocument53 pages"The Oceans": Group 5 OceanographyAlger Dalman ClerigoNo ratings yet
- The Water Cycle: Presented By: Emma Dhalia CalaguiDocument21 pagesThe Water Cycle: Presented By: Emma Dhalia CalaguiDhaliaCalaguiNo ratings yet
- Grasping The OceanDocument17 pagesGrasping The Oceansapphire09No ratings yet
- Page 188-193Document7 pagesPage 188-19305 陳宗榮 RYAN GERALD CHUANo ratings yet
- Estuaries PDFDocument19 pagesEstuaries PDFsiladharmaNo ratings yet
- Distribution of Water Resources The On EarthDocument16 pagesDistribution of Water Resources The On EarthKaizen ShoriNo ratings yet
- Distribution of Water Resources on Earth in 38 CharactersDocument16 pagesDistribution of Water Resources on Earth in 38 CharactersKaizen ShoriNo ratings yet
- Distribution of Water On EarthDocument16 pagesDistribution of Water On EarthKaizen ShoriNo ratings yet
- Composition and Distribution of the World's OceansDocument3 pagesComposition and Distribution of the World's OceanspriyanshNo ratings yet
- Movements of WaterDocument6 pagesMovements of WaterJhaJha AgcaoiliNo ratings yet
- Lakes - Brief ReviewDocument1 pageLakes - Brief Reviewpolyborahnew2001No ratings yet
- Composition of Ocean WaterDocument2 pagesComposition of Ocean Waterabontitori25No ratings yet
- Marine biochemistry lab report analyzes salinity of estuary waterDocument9 pagesMarine biochemistry lab report analyzes salinity of estuary waterMelissa GómezNo ratings yet
- The Salt MinesDocument1 pageThe Salt MinesGabriel Castillo ReyesNo ratings yet
- Assesment 1Document7 pagesAssesment 1John Lloyd SalinasNo ratings yet
- Earth'S Water: - Started As A Combination of Gasses Which Were Carried by Meteor That Collided WithDocument5 pagesEarth'S Water: - Started As A Combination of Gasses Which Were Carried by Meteor That Collided WithEim NellaNo ratings yet
- Water of The SeasDocument82 pagesWater of The SeasBeauNo ratings yet
- Sedimen EstuariesDocument71 pagesSedimen Estuariesmuhabidin sehatNo ratings yet
- Coral Reefs and Estuaries 2Document8 pagesCoral Reefs and Estuaries 2photon090109572No ratings yet
- Text Book Phan 2Document529 pagesText Book Phan 2Van Phuong TranNo ratings yet
- Sodium in The CaribbeanDocument2 pagesSodium in The CaribbeanShamir RagoonananNo ratings yet
- The Chemistry of Natural WatersDocument3 pagesThe Chemistry of Natural Waterscory kurdapyaNo ratings yet
- Lake Vostok EssayDocument8 pagesLake Vostok EssaySamNo ratings yet
- GEO101x 2016 v.2.1 Source Rock-TranscriptDocument3 pagesGEO101x 2016 v.2.1 Source Rock-TranscriptACIDNo ratings yet
- Oceanography (Inglés) Autor Federal University of Agriculture, AbeokutaDocument18 pagesOceanography (Inglés) Autor Federal University of Agriculture, AbeokutaJeshu ValleNo ratings yet
- Salt Concentration: Fresh WaterDocument31 pagesSalt Concentration: Fresh WaterAli Raza (Alee)No ratings yet
- 11th Grade Groundwater GuideDocument51 pages11th Grade Groundwater GuideJovic SasotaNo ratings yet
- 8unit1ch1slideshow2011 1Document42 pages8unit1ch1slideshow2011 1api-238589602No ratings yet
- Estuarine EcosystemDocument46 pagesEstuarine Ecosystemapi-3827285No ratings yet
- Seawater Composition: Salinity, Temperature, Dissolved Gases, and MoreDocument6 pagesSeawater Composition: Salinity, Temperature, Dissolved Gases, and MoreVisalakshi TalakokulaNo ratings yet
- SeaDocument21 pagesSeaLakshmish GopalNo ratings yet
- Aquatic Ecosystem: I. LakesDocument6 pagesAquatic Ecosystem: I. LakesJohn Paul Espiña CandoNo ratings yet
- SeaDocument12 pagesSeaEa RLNo ratings yet
- NotesDocument4 pagesNotesLokesh OriginalsNo ratings yet
- Article EST The Dying SeaDocument1 pageArticle EST The Dying SeaSyahirah SharumNo ratings yet
- Chap 8 Physical GeoDocument3 pagesChap 8 Physical GeoJuliana Mae FabrigarNo ratings yet
- CHP 6 Geog Properties of Sea WaterDocument2 pagesCHP 6 Geog Properties of Sea WaterConsult ShamimNo ratings yet
- High School Earth Science 14-26Document529 pagesHigh School Earth Science 14-26Andrei PopaNo ratings yet
- Introduction To OceanographyDocument6 pagesIntroduction To Oceanographyevident 2014No ratings yet
- Why Is Salt Spread Onto Icy Roads?: Why There Is A Spluttering Sound When Water Is Sprinkled On Hot Oil?Document31 pagesWhy Is Salt Spread Onto Icy Roads?: Why There Is A Spluttering Sound When Water Is Sprinkled On Hot Oil?Sindhuja YaminiNo ratings yet