Professional Documents
Culture Documents
Chemmatters Feb2010 Green Gasoline
Uploaded by
Mohammad FaizCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Chemmatters Feb2010 Green Gasoline
Uploaded by
Mohammad FaizCopyright:
Available Formats
A
mericans pump on average 18
million gallons of gasoline per
hour into their cars, but less
than half of it comes from oil produced in
the United States. To make up for the dif-
ference, the United States imports oil from
politically unstable regions in the Middle
East, Africa, and South America. The United
States could lessen its reliance on imports
by drilling more oil, but that may only be
a temporary solution because many
scientists expect that the worlds oil
resources will eventually dry up.
Another solution is to use crops and
plants to make gasoline. It might
be hard to imagine, but
chemists can convert
corn stalks, sawdust, and
grasses into gasoline. They
do it by breaking down the plant mol-
ecules and reshuffling their parts into
the same molecules found in gasoline.
This new type of gasoline, called green gas-
oline, would have many advantages. In addi-
tion to reducing U.S. dependence on foreign
countries, it would also produce less pollution
than oil-derived fuels, because the plants used
to produce green gasoline would absorb some
of the pollutants. Also, we would not have to
worry about running out of fuel because green
gasoline is renewable: The plants used to
make it can be grown over and over again.
There are other plant-derived fuels, or bio-
fuels, such as ethanol, which is produced by
the fermentation of plant sugars by yeast, and
biodiesel, which is made from vegetable oil or
animal fats. But the advantage of green gaso-
line is that it can directly replace oil-derived
gasoline.
We wouldnt have to buy new cars or build
new refineries and pipelines just to use green
gasoline, says chemical engineer Jennifer
Holmgren of UOP, a company in Des Plaines,
Ill., that develops oil-refining technologies.
In as little as 5 years, you might drive up
to a filling station and, without even realizing
it, fill up your car with gasoline that came
directly from plants. At least that is what some
researchers foresee. The challenge will be to
figure out how to make large quantities of green
gasoline because, up until now, only small
quantities have been made in the laboratory.
Non-green gasoline
Gasoline derived from oil has been the main
choice for transportation fuel during the past
century. It is stable, noncorrosive, and packs
a high-energy punch. The energy in gasoline
is stored in molecules called hydrocarbons,
which are long chains or rings made of carbon
and hydrogen atoms. When given a spark, the
carbon and hydrogen atoms combine with
oxygen in the air, resulting in carbon dioxide,
water, and a lot of energy.
An example of this is the burning of octane
(C8H18), one of the hydrocarbons found in
gasoline:
2 C8H18 + 25 O2 16 CO2 + 18 H2O + energy
This reac-
tion is actually
an explosion: The
amount of energy in
a pound of gasoline is
more than 10 times that
found in a pound of trinitro-
toluene, a common explosive.
In a car, this energy heats the
carbon dioxide, water vapor, and
other gases present in the engine,
which causes them to expand and to
push on the engines pistons.
Most of the time, gasoline inside a
cars engine is not totally broken down
to carbon dioxide and
water. Some hydro-
carbons
remain, and some of the
fragments end up as carbon
monoxi de (CO), a very toxic gas. The
combustion process also can create other
pol l ut ant s, such as nitrogen oxides (NO
and NO2), that lead to smog and acid rain.
A good deal of this pollution has been
reduced in modern cars. Most of them con-
tain a device called a catalytic converter that
filters the pollutants before they are released
in a cars exhaust pipe. But this device can-
not prevent the emission of carbon dioxide, a
gas that mixes in with the atmosphere, traps
heat from the sun, and contributes to global
climate change.
C
H
H
C
H
H
C
H
H
C
H
H
C
H
H
C
H
H
C
H
H
C
H
H H
H
By Michael Schirber
Green Gasoline
Fuel from Plants
ChemMatters, FEBRUARY 2010 13
M
I
K
E
C
I
E
S
I
E
L
S
K
I
I
S
T
O
C
K
Octane
Green gasoline can help lessen the prob-
lem, because plants absorb carbon dioxide
and convert it into organic compounds by
using energy from sunlight. The carbon diox-
ide is therefore recycled, going from the air to
the plants to green gasoline, and then back to
the air, when green gasoline is burned.
Green gasoline is not the only biofuel that
recycles carbon dioxide this way, but it has
the advantage that it carries the same energy
punch as gasoline.
Packing energy in
green gasoline
Plants store the energy of the sun in organic
molecules called carbohydrates, which include
sugars and cellulosethe main constituent of
a plant cell wall. This energy can be extracted
directly by burning the plants. For example,
in the 19th century, some steamships were
powered by burning wood. But burning wood
or other plant materials to run your car is not
practical. They do not flow into the engine like
liquid fuels do, and you would need to carry
three times as much weight in wood to go the
same distance as with gasoline.
We use gasoline because it packs a lot of
energy, says John Regalbuto, director of the
Catalysis and Biocatalysis
Program at the National Sci-
ence Foundation, Arlington,
Va. Gasoline takes less
space and is more efficient.
The amount of energy per
pound contained in plants
can be increased by
removing oxygen from the
carbohydrate molecules. Octane and other
hydrocarbons, as mentioned above, burn by
breaking bonds between carbon and hydrogen
atoms, which then grab onto oxygen atoms.
Carbohydrates burn in the same way, but they
already contain some oxygen. In short, if you
remove as many oxygen atoms as you can
from a molecule, it will pack more energy.
As an example, the sugar glucose (C
6H12O6)
is similar in shape to cyclohexane (C6H12), a
hydrocarbon found in gasoline. But glucose
contains six additional oxygen atoms, which
reduces its energy content. You would need
roughly three pounds of glucose to get the
same amount of energy as one pound of
cyclohexane.
C C
H H
H
H
H
H
H
H
H
H
H
H
C
C C
C
O
CH
2
OH
OH
OH
OH
OH
H
H
H
H
H
Glucose Cyclohexane
Cellulose
Turning cellulose into gasoline: Cellulose (a, hydrogen atoms omitted for clarity) is heated to 500 C, breaking it apart into smaller molecules (b).
These molecules bind to a porous substance called zeolite (c). As they bind to the zeolite, these molecules undergo chemical reactions that remove
oxygen atoms from them. These reactions result in aromatic molecules (such as toluene and indene), which are components of gasoline, along with
other final products, such as carbon dioxide and water (d).
(a) (b) (c) (d)
14 ChemMatters, FEBRUARY 2010 www.acs.org/chemmatters
A
N
T
H
O
N
Y
F
E
R
N
A
N
D
E
Z
;
A
D
A
P
T
E
D
F
R
O
M
G
R
A
S
S
O
L
I
N
E
A
T
T
H
E
P
U
M
P
,
B
Y
G
E
O
R
G
E
W
.
H
U
B
E
R
A
N
D
B
R
U
C
E
E
.
D
A
L
E
,
S
C
I
E
N
T
I
F
I
C
A
M
E
R
I
C
A
N
,
J
U
L
Y
2
0
0
9
.
U
O
P
L
L
C
C
C
O
C
H
H
H
H
H
OH
CH
2
OH
OH
C
C
C
O
C
H
H
H
H
H
OH
CH
2
OH
OH
C
C
C C O
O
C
H
H
H
H
H
OH
CH
2
OH
OH
C
C O C O
carbon
oxygen
indene
toluene
carbon
dioxide
carbon
monoxide
carbon
monoxide
toluene
toluene
carbon
dioxide
zeolite
Scientists at the University of Wisconsin,
Madison, have successfully stripped oxygen
atoms from glucose and other sugars to cre-
ate hydrocarbons similar to cyclohexane. The
resulting liquid is essentially gasoline, but it
is green because it is extracted from plants,
not oil.
Virent Energy Systems, a renewable energy
company based in Madison, is working on this
sugar-to-hydrocarbon process and is planning
to produce 100 million barrels of green gaso-
line in 57 years.
Energy from plant
leftovers
Carbohydrates, such as glucose, are
typically derived from sugarcane or corn. But
using these food crops for the production of
green gasoline runs the risk of driving up food
prices. It would be better to make green gaso-
line with plant materials that are not used.
Current research is looking at agricultural
and forestry leftovers, such as cornstalks
and sawdust, and nonfood crops, such as
switchgrass and other prairie grasses that can
be grown without much
irrigation or fertilizer.
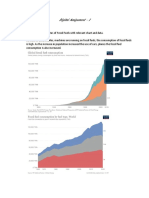
More than 1 billion
tons of plant leftovers are
available each year in the
United States, according
to a study by the U.S.
Department of Agriculture
and the U.S. Department
of Energy. This could
produce 100 billion gal-
lons per year of green gasolineabout half the
current U.S. annual consumption of gasoline
and diesel.
One of the scientists working on the conver-
sion of plant leftovers into gasoline is George
Huber, a professor of chemical engineering
at the University of Massachusetts, Amherst.
He and his colleagues have developed a
technique that can turn almost any plant sub-
stance into gasoline.
The plant materials are first placed in a
reactor at 500 C. This breaks down the cel-
lulose and other tough plant parts into smaller
molecules, called anhydro sugars. These
molecules then go through a porous structure
called a zeolite, which is made of aluminum
and silica. The pores are designed to be just
big enough for the anhydro sugar molecules
to enter.
If the size of the pores is too small, the
molecules cant get in, Huber says. If they
are too big, too many molecules crowd in, and
unwanted reactions
occur. This can
result in a carbon-
rich material that
clogs up the zeolite.
The pores in
Hubers zeolites are
about six atoms
wide, which is room
enough for an anhy-
dro sugar molecule
to enter and attach
to the surface,
where it loses
its oxygen and re-forms into a ring-shaped
hydrocarbon similar to cyclohexane but with
less hydrogen atoms. The full processfrom
breakdown in the reactor to hydrocarbon for-
mationtakes just a few seconds.
Holmgren and her UOP colleagues are
working on a similar project. They start by
placing plant materials into a vessel that
whips up a tornado of hot sand. This swirling
sandstorm rapidly heats the plant matter to
500 C, thereby cooking it into an oily liquid.
This liquid is then processed into gasoline and
other fuels.
Other researchers, such as Lanny
Schmidt, professor of chemical engineer-
ing and materials science, and colleagues
at the University of Minnesota, Twin Cities,
are heating plant ingredients to higher tem-
peratures (700 C1,000 C) to produce a
gas mixture of carbon monoxide (CO) and
hydrogen (H
2). These molecules of gas are
then reassembled into hydrocarbons, such
as octane, with water as a byproduct:
8 CO + 17 H
2
C
8
H
18
+ 8 H
2
O
The future
All of these processes remain under devel-
opment. We are still in the early days of
green gasoline, says Robert Anex, associate
professor of agricultural and biosystems engi-
neering at Iowa State University in Ames.
So far, there are logistics problems, such
as how to collect cornstalks from widely
separated farms and bring them to one central
facility. And although the chemistry works well
in a laboratory, it is not yet clear whether the
same processes will work on a large scale.
But green gasoline holds a lot of promise,
and scientists, engineers, and policymakers
are now starting to realize it. Says Holmgren,
Energy is the most important challenge fac-
ing us today, and chemistry will play a key
role in making sure we have an alternative
source of energy into the next 100 years.
SELECTED REFERENCES
Huber, G.; Dale, B. Grassoline at the Pump.
Scientific American, July 2009.
Kintisch, E. The Greening of Synfuels. Science,
April 18, 2008.
Savage, N. Making Gasoline from Bacteria.
Technology Review, Aug 1, 2007:
http://www.technologyreview.com/read_article.
aspx?ch=specialsections&sc=biofuels&id=191
28&a [Sept 2009]
Development of Cellulosic Biofuels: Video lecture
by Chris Somerville, director of the Energy
Biosciences Institute at the University of
California, Berkeley: http://tinyurl.com/
grassoline [Sept 2009]
Green Gasoline: A Renewable Petroleum Alternative
from Plants: http://www.nsf.gov/news/newsme-
dia/greengasoline/index.jsp#added [Sept 2009]
Michael Schirber is a science writer who
lives in Lyon, France.This is his first article in
ChemMatters.
Virents Liquid Fuel Laboratories
George Huber poses with
a vial of green gasoline
compounds.
Switchgrass
ChemMatters, FEBRUARY 2010 15
I
S
T
O
C
K
V
I
R
E
N
T
E
N
E
R
G
Y
S
Y
S
T
E
M
S
S
T
E
P
H
E
N
A
U
S
M
U
S
B
E
N
B
A
R
N
H
A
R
T
You might also like
- Hydrogen As An Alternative FuelDocument6 pagesHydrogen As An Alternative FuelPurna Satria NugrahaNo ratings yet
- Problem: Vehicular PollutionDocument4 pagesProblem: Vehicular Pollutionapi-272945780No ratings yet
- Bio GasDocument8 pagesBio GasammumNo ratings yet
- How To Make The Most Of: Carbon DioxideDocument3 pagesHow To Make The Most Of: Carbon Dioxidetorta1No ratings yet
- Young Innovators Program 2022: Greenhouse Gas Artificial PhotosynthesisDocument10 pagesYoung Innovators Program 2022: Greenhouse Gas Artificial Photosynthesisneetus creationNo ratings yet
- Biofuel Lab ReportDocument7 pagesBiofuel Lab ReportMaxWitt100% (3)
- Alternative To Hydrocarbon Fuels EssayDocument4 pagesAlternative To Hydrocarbon Fuels EssayKhuslenNo ratings yet
- Biogas - Wikipedia, The Free Encyclopedia PDFDocument12 pagesBiogas - Wikipedia, The Free Encyclopedia PDFBrijesh GautamNo ratings yet
- Fossil Fuels to Alternative FuelsDocument5 pagesFossil Fuels to Alternative Fuelssanju tomNo ratings yet
- An Example of A Well Written Argumentative EssayDocument4 pagesAn Example of A Well Written Argumentative Essaymaiwandmoheb123No ratings yet
- 1,000 Anaerobic Digesters Treat Organic Waste WorldwideDocument20 pages1,000 Anaerobic Digesters Treat Organic Waste WorldwideilariofabbianNo ratings yet
- 5 Future Fossil Energy TechnologiesDocument14 pages5 Future Fossil Energy TechnologiesAbhinav ChuksNo ratings yet
- Hydrogen Fuel CellsDocument4 pagesHydrogen Fuel CellsSeptian JauhariansyahNo ratings yet
- Automotive EmissionsDocument30 pagesAutomotive EmissionsAmarjeet RaiNo ratings yet
- Fossil Fuels Formation Uses ConservationDocument6 pagesFossil Fuels Formation Uses ConservationjasujunkNo ratings yet
- Water Fuel Engines: An Alternative to GasolineDocument6 pagesWater Fuel Engines: An Alternative to GasolineTimothy AgustinNo ratings yet
- Biodiesel CombustionDocument29 pagesBiodiesel CombustionEr Ashish BahetiNo ratings yet
- Alternative Fuels, Known As Non-Conventional or AdvancedDocument5 pagesAlternative Fuels, Known As Non-Conventional or Advancedaerochandru.87No ratings yet
- Energy-Related Essay MESC TahriziDocument2 pagesEnergy-Related Essay MESC TahriziTahrizi AndanaNo ratings yet
- Brief Survey On Automobile FuelsDocument11 pagesBrief Survey On Automobile FuelsPintilei DanutNo ratings yet
- Generation of HHO and Its Effect on Engine PerformanceDocument5 pagesGeneration of HHO and Its Effect on Engine PerformanceNam stareNo ratings yet
- Energy WoodDocument2 pagesEnergy WoodMag FhearadhaighNo ratings yet
- Alternate FuelsDocument16 pagesAlternate FuelsharizhunkNo ratings yet
- Fluidized Bed CombustionDocument57 pagesFluidized Bed CombustionRamesh Bobba100% (1)
- Hydrogen Fuel Is Getting BuzzDocument2 pagesHydrogen Fuel Is Getting BuzzEmilio MeraNo ratings yet
- A Sustainable Approach For Carbon Dioxide FixationDocument4 pagesA Sustainable Approach For Carbon Dioxide Fixation0721673895No ratings yet
- The Team Used Xylose - A Simple Plant SugarDocument5 pagesThe Team Used Xylose - A Simple Plant SugarOvaid MehmoodNo ratings yet
- FUELSDocument35 pagesFUELSJayson DayaoNo ratings yet
- Alternative FuelsDocument1 pageAlternative FuelsNatasha_Noshy__9100No ratings yet
- Algae Biofuel Case Study: Emerging Companies Pursuing CO2 ReductionDocument5 pagesAlgae Biofuel Case Study: Emerging Companies Pursuing CO2 ReductionabsinitNo ratings yet
- HydrogenDocument24 pagesHydrogenmeet003No ratings yet
- Dnv-Position Paper Co2 Utilization Tcm4-445820Document20 pagesDnv-Position Paper Co2 Utilization Tcm4-445820lhphong021191No ratings yet
- Energy CrisisDocument8 pagesEnergy CrisisJosielyn TarsNo ratings yet
- Hydrogen Will Never Be A Full Solution To Our Green Energy Problems (Contradict)Document2 pagesHydrogen Will Never Be A Full Solution To Our Green Energy Problems (Contradict)LauraNo ratings yet
- Alternative FuelDocument6 pagesAlternative FuelKinna VnezhNo ratings yet
- Vehicle emission control techniquesDocument6 pagesVehicle emission control techniquesmsaqibraza93No ratings yet
- Where Do We Put It?: Trap It in RocksDocument4 pagesWhere Do We Put It?: Trap It in RocksChunchu AnilNo ratings yet
- Ensayo InglesDocument4 pagesEnsayo InglesEdison Ramirez ChaparroNo ratings yet
- Lesson 6-Saving The Planet (Continuation)Document10 pagesLesson 6-Saving The Planet (Continuation)Allyssa TrinidadNo ratings yet
- Fired Up With IdeasDocument4 pagesFired Up With IdeasH ProductionNo ratings yet
- The Economics of Co Separation and CaptureDocument18 pagesThe Economics of Co Separation and CaptureJonah VaughnNo ratings yet
- Economics Class 12 ProjectDocument10 pagesEconomics Class 12 ProjectabcindNo ratings yet
- Using Glycerin and Biomass to Create Renewable Energy PelletsDocument7 pagesUsing Glycerin and Biomass to Create Renewable Energy Pelletsanon_312581309No ratings yet
- Removing CO2 from Air with Artificial TreesDocument2 pagesRemoving CO2 from Air with Artificial TreesferfrascaNo ratings yet
- End of Diesel EngineDocument3 pagesEnd of Diesel EngineAlex PrasadNo ratings yet
- Fuel Cell As Alternative FuelDocument11 pagesFuel Cell As Alternative FuelGaurao WarungaseNo ratings yet
- Investigation of Engine Internal Combustion To Reduce Exhaust Emission: A ReviewDocument14 pagesInvestigation of Engine Internal Combustion To Reduce Exhaust Emission: A ReviewTJPRC PublicationsNo ratings yet
- A Study On: Carbon Recycling and Carbon CredtingDocument12 pagesA Study On: Carbon Recycling and Carbon CredtingMargaret JacksonNo ratings yet
- Hydrogen Energy How It Works Benefit of Hydrogen EnergyDocument3 pagesHydrogen Energy How It Works Benefit of Hydrogen EnergyUnatti AgarwalNo ratings yet
- Hydrogen EnergyDocument2 pagesHydrogen Energyvipin1001No ratings yet
- Hydrogen FuelDocument39 pagesHydrogen FuelSushanta PradhanNo ratings yet
- Thermal Lab OpenDocument9 pagesThermal Lab OpenNikhil KattiNo ratings yet
- Hydrogen As A Fuel Pros N ConsDocument4 pagesHydrogen As A Fuel Pros N ConsBrian MasauliNo ratings yet
- Alternate Fuel For Maritime UseDocument4 pagesAlternate Fuel For Maritime Useprateek prakashNo ratings yet
- Mec 13Document1 pageMec 13api-3761679No ratings yet
- Proposal Esssy Final Edit2Document5 pagesProposal Esssy Final Edit2api-457909911No ratings yet
- Green GazDocument3 pagesGreen GazMansour MohamedTNo ratings yet