Professional Documents
Culture Documents
Fukuyama Group - Group Meeting Problems 07/17/2011: Low Yield Under Basic Condition
Uploaded by
Anonymous rhUNqC1s0Original Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Fukuyama Group - Group Meeting Problems 07/17/2011: Low Yield Under Basic Condition
Uploaded by
Anonymous rhUNqC1s0Copyright:
Available Formats
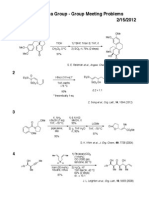
Fukuyama Group - Group Meeting Problems
07/17/2011
1
O
O HO
BF4
+
PhI
Me
KOCEt3 (3 eq)
[Fe2(CO)9]* (2 eq)
benzene
95 C;
THF
78 C to rt
DBU (20 eq)
95 C
74%
O O
51%
* low yield under basic condition
E. M. Carreira et al., Angew. Chem. Int. Ed., 50, 2962 (2011)
2
O
Dy(OTf)3
PhHN
CH3CN, 80 C
OH
PhN
90%
J. R. Alaniz et al., Angew. Chem. Int. Ed., 50, 7167 (2011)
3
Me
N
Ph
O
CO2H
TFAA (3 eq*)
Ph3PEtBr
n-BuLi, THF
AcOEt
0 C to rt
20 C to rt;
AcOH, 80 C
87%
(* theoretically 2 eq)
87%
Me
Ph
CF3
N
Me
M. Kawase et al., Org. Lett., 12, 4776 (2010)
4
SiMe3
Boc
BocNHOH (2.5 eq)
NaIO4 (2.5 eq)
OsO4, NMO
SmI2
HO
MeOH/ H2O
20 C
THF/ t-BuOH/ H2O
20 C
THF
78 C
HO
52%
82%
60%
NH OH
*1 OH
H
NBoc
OH OH
*1 stereochemistry
not determined
Y. Landais et al., Org. Lett., 10, 4195 (2008)
Fukuyama Group - Group Meeting Problems
07/24/2011
1
H H H O
MeO
BHT, PhH
H
235 C, 88%
H
O
1) MeMgBr, CuBrMe2S
Me2S, THF, 20 C
66%, 14:1 dr
H O
BHT, PhH
MeO
120 C, 96%
2) TMSCHN2
MeOH/PhH, rt, 93%
H
CO2Me
Me
M. L. Snapper et al., Org. Lett., 3, 2819 (2001)
O
2
O
[Rh(OCOC7H15)2]2 (cat.)
DMAD
N2
MeO2C
CH2Cl2
rt
MeO2C
H
N
70%
A. Padwa et al., Tetrahedron Lett., 34, 7853 (1993)
HO
LTA
CHCl3
-40 C
Tricyclic Compound
(C17H24O3)
1) CrO3
H2SO4/H2O/acetone
rt, 96%
2) OsO4, THF
rt, 79%
O
O
3) p-TsOHH2O
toluene, reflux
85%
61%
T. R. R. Pettus et al., J. Am. Chem. Soc., 133, 1603 (2011)
4
Ph
O
Ph
O
NIS (2.5 eq)
H2O (5.0 eq)
MeCN
40 to 0 C
62%*
1) DDQ
MeCN-H2O (10:1)
60 C
58% (36% SM)
CAN
MeCN-H2O (1:1)
rt;
2) NaH2PO4, NaClO2
2-methyl-2-butene
t-BuOH-H2O (5:1), rt
92%
K2CO3
O
I
O
O
80%
(*18% other diastereomixture)
Y. Kita et al., Angew. Chem. Int. Ed., 44, 734 (2005)
Fukuyama Group - Group Meeting Problems
07/31/2011
1
O
N2
O
N
Me
1) allyl alcohol
Rh2(OAc)4
CH2Cl2, rt
3) H(OH)NMeHCl
pyridine, MeOH
reflux
2) xylenes
reflux
85% (2 steps)
4) imidazoleHCl
EtOH, reflux
81% (2 steps)
O
MeN
H
O
N
Me
J. L. Wood et al., Tetrahedron, 66, 6647 (2010)
2
OH
NMe
OAc
N
H
OH
NMe
N
H
TFA, rt
61%
* : Diastereomixture
S. Blechert et al., J. Am. Chem. Soc., 126, 3534 (2004)
3
O
OH
O
Br
MeO2C
Pd(PPh3)4, i-Pr2NH
microwave
benzene
160 C
60%
1) DMP
CH2Cl2, rt
77%
O O
O
O
2) cat. RuCl3, NaIO4
CCl4-CH3CN-H2O
50%
CO2Me
J. Suffert et al., Angew. Chem. Int. Ed., 50, 3285 (2011)
TBSO
SmI2, HMPA
N
Me
i-PrOH
THF, 35 C
99%
1) CsF, PhN(Tf)2
DME
2) Pd(OAc)2, PPh3
HCO2H, DMF
87% (2 steps)
O
LDA
THF, 78 C;
MeI
NMe
Me
67%
Y. Landais et al., Org. Lett., 10, 4441 (2008)
You might also like
- Fukuyama Group - Group Meeting Problems 11/10/2012: PH BF KDocument3 pagesFukuyama Group - Group Meeting Problems 11/10/2012: PH BF KAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 08/01/2012: Otes MeDocument3 pagesFukuyama Group - Group Meeting Problems 08/01/2012: Otes MeAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 09/07/2011: O N O Me MeDocument2 pagesFukuyama Group - Group Meeting Problems 09/07/2011: O N O Me MeAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 10/06/2012: J. Rodriguez Et Al., Org. Lett., 12, 4212 (2010)Document3 pagesFukuyama Group - Group Meeting Problems 10/06/2012: J. Rodriguez Et Al., Org. Lett., 12, 4212 (2010)Anonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 08/08/2011: T-Buok, N-Buli N-Buli (1.1 Eq)Document3 pagesFukuyama Group - Group Meeting Problems 08/08/2011: T-Buok, N-Buli N-Buli (1.1 Eq)Anonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 01/11/2013: T-BuliDocument4 pagesFukuyama Group - Group Meeting Problems 01/11/2013: T-BuliAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 06/02/2010: O P N O Ome OmeDocument2 pagesFukuyama Group - Group Meeting Problems 06/02/2010: O P N O Ome OmeAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 1/17/2012: Bicyclic CompoundDocument1 pageFukuyama Group - Group Meeting Problems 1/17/2012: Bicyclic CompoundAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 2/15/2012: Et Si Siet O D Thf-Hmpa - 78 °C 80% Et Si Siet Oh DDocument1 pageFukuyama Group - Group Meeting Problems 2/15/2012: Et Si Siet O D Thf-Hmpa - 78 °C 80% Et Si Siet Oh DAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 12/12/2012: CO Me NDocument2 pagesFukuyama Group - Group Meeting Problems 12/12/2012: CO Me NAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 10/01/2011: N-Buli (2 Eq), - 20 °CDocument2 pagesFukuyama Group - Group Meeting Problems 10/01/2011: N-Buli (2 Eq), - 20 °CAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 09/05/2012: P-Tsoh (Cat.)Document2 pagesFukuyama Group - Group Meeting Problems 09/05/2012: P-Tsoh (Cat.)Anonymous rhUNqC1s0No ratings yet
- Química OrganicaDocument6 pagesQuímica OrganicaVllsSNo ratings yet
- 3,4 AdciojnDocument4 pages3,4 AdciojnCalamidad Loco PeligroNo ratings yet
- My PresentationDocument44 pagesMy PresentationNilaHudaBaqirNo ratings yet
- ANTHONY CRASTO-Brevetoxin SynthesisDocument58 pagesANTHONY CRASTO-Brevetoxin SynthesisAnthony Melvin Crasto Ph.DNo ratings yet
- Benzotriazole 2000Document33 pagesBenzotriazole 2000eraborNo ratings yet
- Convenient Access To An Alkenyl (Biscarbene) - Bridged Heterobinuclear (Ru W) Complex. X-Ray Structure of ( (H - C Me H) Ru C (Ome) (CHCHC H CH Chco Me) - (CL) (Pme) ) (PF)Document3 pagesConvenient Access To An Alkenyl (Biscarbene) - Bridged Heterobinuclear (Ru W) Complex. X-Ray Structure of ( (H - C Me H) Ru C (Ome) (CHCHC H CH Chco Me) - (CL) (Pme) ) (PF)Robeul AwalNo ratings yet
- Wjoc 2 1 1 PDFDocument8 pagesWjoc 2 1 1 PDFWalid Ebid ElgammalNo ratings yet
- Mcclory-Lit-03 16 09Document58 pagesMcclory-Lit-03 16 09Vasudevan SubramaniyanNo ratings yet
- Shenvi Sep 04Document10 pagesShenvi Sep 04patilamardip007No ratings yet
- Synthesis of Schiff Bases by Aromatic Amine Condensation With 3,3 - Bithiophenes-2,2 and 4,4 - DicarbaldehydesDocument5 pagesSynthesis of Schiff Bases by Aromatic Amine Condensation With 3,3 - Bithiophenes-2,2 and 4,4 - DicarbaldehydesYsabel Huaccallo AguilarNo ratings yet
- CCSC 2022 02418 File002Document63 pagesCCSC 2022 02418 File002migenyasuyoshiNo ratings yet
- 1987 Mayr - Acid - and Base-Catalyzed Ring-Opening Reactions or A Sterically Hindered Epoxide PDFDocument3 pages1987 Mayr - Acid - and Base-Catalyzed Ring-Opening Reactions or A Sterically Hindered Epoxide PDFHernán AstudilloNo ratings yet
- jm9b01579 Si 001Document36 pagesjm9b01579 Si 001Jagjeet GujralNo ratings yet
- Group Meeting Problems 2023/04/08: O Me Me Me OH Me Me Me O HODocument7 pagesGroup Meeting Problems 2023/04/08: O Me Me Me OH Me Me Me O HOroccenxuNo ratings yet
- Synthesis of The Steroidal Natural Product Ouabain and Aglycone Ouabagenin and Aglycone OuabageninDocument23 pagesSynthesis of The Steroidal Natural Product Ouabain and Aglycone Ouabagenin and Aglycone OuabageninNober Sandy LayukNo ratings yet
- Camphorsultam CleavageDocument3 pagesCamphorsultam Cleavageleda_prandiNo ratings yet
- 1990 Annonaceous Acetogenins - A ReviewDocument42 pages1990 Annonaceous Acetogenins - A ReviewJuan PizanoNo ratings yet
- MS 204Document4 pagesMS 204Suyog patilNo ratings yet
- Synthesis, Characterization, and Organic Chemistry of An Edge-Bridged Half-Open TitanoceneDocument8 pagesSynthesis, Characterization, and Organic Chemistry of An Edge-Bridged Half-Open TitanoceneANTONIO C.MNo ratings yet
- Lysergic Kurokawa PDFDocument15 pagesLysergic Kurokawa PDFBrandon TimmNo ratings yet
- Molecules 17 11046Document10 pagesMolecules 17 11046garciaalvarezjosheveth528No ratings yet
- 2007 Synthesis of 1,5-Dinitroaryl-1,4-Pentadien-3-Ones Under Ultrasound IrradiationDocument5 pages2007 Synthesis of 1,5-Dinitroaryl-1,4-Pentadien-3-Ones Under Ultrasound IrradiationJESUS DAVID BOLA‹O JIMENEZNo ratings yet
- Preview 8Document94 pagesPreview 8Rohan PrajapatiNo ratings yet
- Ring Opening of Cyclic Anhydrides: Synthesis of Achiral Half-Esters Using Lewis AcidsDocument4 pagesRing Opening of Cyclic Anhydrides: Synthesis of Achiral Half-Esters Using Lewis AcidsNaveed UR RehmanNo ratings yet
- Tbtu MechanismDocument6 pagesTbtu MechanismFangel IodohemioneNo ratings yet
- Preparation of Diethyl Malonate Adducts From Chalcone Analogs Containing A Thienyl RingDocument7 pagesPreparation of Diethyl Malonate Adducts From Chalcone Analogs Containing A Thienyl RingGabriel PekárekNo ratings yet
- Tetrahedron Letters Volume 40 Issue 5 1999 (Doi 10.1016/s0040-4039 (98) 02540-4) Francisco JoséRomero-Salguero Jean-Marie Lehn - Synthesis of Multitopic Bidentate Ligands Based On Alternating PyDocument4 pagesTetrahedron Letters Volume 40 Issue 5 1999 (Doi 10.1016/s0040-4039 (98) 02540-4) Francisco JoséRomero-Salguero Jean-Marie Lehn - Synthesis of Multitopic Bidentate Ligands Based On Alternating PyJoakin BahamondesNo ratings yet
- 16 Sharpless Asymmetric EDocument4 pages16 Sharpless Asymmetric ENicolò MarnoniNo ratings yet
- Scrobipalpuloides Absoluta: Sex Pheromone of Tomato Pest (Lepidoptera: Gelechiidae)Document14 pagesScrobipalpuloides Absoluta: Sex Pheromone of Tomato Pest (Lepidoptera: Gelechiidae)Giovana Mamani HuayhuaNo ratings yet
- An Efficient Synthesis of Racemic TolterodineDocument2 pagesAn Efficient Synthesis of Racemic TolterodineJignesh TrivediNo ratings yet
- 07.Mmc ExperimentalDocument14 pages07.Mmc ExperimentalNurul Putry SyahrulNo ratings yet
- Org Syn Collective Vol 9. Page 88Document4 pagesOrg Syn Collective Vol 9. Page 88rrgodboleNo ratings yet
- Fitoterapia: Dong Pei, Jun-Xi Liu, Duo-Long DiDocument6 pagesFitoterapia: Dong Pei, Jun-Xi Liu, Duo-Long DiDuong Pham QuangNo ratings yet
- Ja0449269 Si 001Document17 pagesJa0449269 Si 001Origamist KryaNo ratings yet
- Typical Procedure For The Synthesis of XanthatesDocument10 pagesTypical Procedure For The Synthesis of XanthatesraminNo ratings yet
- OH O O: Griffiths, Russell Jon, PCT Int. Appl., 2013124682, 29 Aug 2013Document3 pagesOH O O: Griffiths, Russell Jon, PCT Int. Appl., 2013124682, 29 Aug 2013kasun1237459No ratings yet
- Synthesis of Carboxylic Acids, Esters, Alcohols and Ethers Containing A Tetrahydropyran Ring Derived From 6-Methyl-5-Hepten-2-OneDocument10 pagesSynthesis of Carboxylic Acids, Esters, Alcohols and Ethers Containing A Tetrahydropyran Ring Derived From 6-Methyl-5-Hepten-2-OnebiologiNo ratings yet
- AKD BuchwaldhartwigDocument39 pagesAKD BuchwaldhartwigMurali Venkat NagNo ratings yet
- Cooper1979 PDFDocument3 pagesCooper1979 PDFJ Venkat RamanNo ratings yet
- Chem 215 Myers: Birch ReductionDocument7 pagesChem 215 Myers: Birch ReductionPrasanna AndojuNo ratings yet
- Org Let. Sauropunol - SupportingDocument63 pagesOrg Let. Sauropunol - SupportingDebanjan DasNo ratings yet
- Identification of 2cmethylenelophenol From Heartwood of Azadirachta ZndicaDocument2 pagesIdentification of 2cmethylenelophenol From Heartwood of Azadirachta ZndicaRigotti BrNo ratings yet
- Drugs of The Future 2002, 27 (2) 143-158Document16 pagesDrugs of The Future 2002, 27 (2) 143-158Rajesh TammanaNo ratings yet
- Spectrochimica Acta Part A: Molecular and Biomolecular SpectrosDocument8 pagesSpectrochimica Acta Part A: Molecular and Biomolecular SpectrosAdrian Bermudez LoeraNo ratings yet
- Methyl Pothoscandensate, A New Ent-18 (4 ! 3) - Abeokaurane From Pothos ScandensDocument7 pagesMethyl Pothoscandensate, A New Ent-18 (4 ! 3) - Abeokaurane From Pothos Scandensaji gumelarNo ratings yet
- Friday Problem Set: Maria Duran 11/19/2010Document56 pagesFriday Problem Set: Maria Duran 11/19/2010Aba BabNo ratings yet
- Exam III Study GuideDocument2 pagesExam III Study GuideAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 01/11/2013: T-BuliDocument4 pagesFukuyama Group - Group Meeting Problems 01/11/2013: T-BuliAnonymous rhUNqC1s0No ratings yet
- 1986 - CHA - HAL - Thermodyn Prop C1-C4 Ideal GasDocument68 pages1986 - CHA - HAL - Thermodyn Prop C1-C4 Ideal GasAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 09/05/2012: P-Tsoh (Cat.)Document2 pagesFukuyama Group - Group Meeting Problems 09/05/2012: P-Tsoh (Cat.)Anonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 2/15/2012: Et Si Siet O D Thf-Hmpa - 78 °C 80% Et Si Siet Oh DDocument1 pageFukuyama Group - Group Meeting Problems 2/15/2012: Et Si Siet O D Thf-Hmpa - 78 °C 80% Et Si Siet Oh DAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 1/17/2012: Bicyclic CompoundDocument1 pageFukuyama Group - Group Meeting Problems 1/17/2012: Bicyclic CompoundAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 12/12/2012: CO Me NDocument2 pagesFukuyama Group - Group Meeting Problems 12/12/2012: CO Me NAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 10/01/2011: N-Buli (2 Eq), - 20 °CDocument2 pagesFukuyama Group - Group Meeting Problems 10/01/2011: N-Buli (2 Eq), - 20 °CAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 12/11/2010: T-BuomeDocument1 pageFukuyama Group - Group Meeting Problems 12/11/2010: T-BuomeAnonymous rhUNqC1s0No ratings yet
- Fukuyama Group - Group Meeting Problems 06/02/2010: O P N O Ome OmeDocument2 pagesFukuyama Group - Group Meeting Problems 06/02/2010: O P N O Ome OmeAnonymous rhUNqC1s0No ratings yet