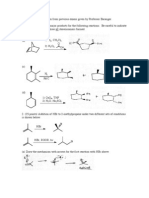
1. (21 points) Draw the products for the following reactions.
Be careful to indicate stereochemistry and to draw all stereoisomers formed. a)
1) O3 2) Me2S
b)
1) BH3-THF 2) H2O2, NaOH, H2O
c)
1) OsO4 2) Na2SO3, H2O
2. (16 points) The following reaction would not occur as pictured. Draw the products that would form and explain your answer either with words or with a mechanism with arrows.
H2SO4, H2O
HO
3. (27 points) We have discussed a number of electrophilic addition reactions involving alkenes. a) The addition of H2C:, Br2, and CH3COOOH proceed by similar mechanisms. Draw the mechanism with arrows for the addition of these reagents to the alkene shown below. (For Br2 show the formation of the first intermediate).
b) In all of these reactions, the bonding orbital of the alkene acts as the Lewis base and HOMO. Draw and label the Lewis acid or LUMO on two of the three reagents above (H2C:, Br2, and CH3COOOH) that reacts with the bonding orbital of the alkene.
�c) The addition of H2 to an alkene does not proceed by a similar mechanism. Why does H2 react by a different mechanism? 4. (22 points) a) Consider the reaction below. Draw a reaction coordinate energy diagram that illustrates the two competing pathways and the formation of the major product. Label the axes, starting materials, intermediates, products, G, G, and transition states.
+
Br major product + HBr
Br
b) Explain how the Hammond postulate justifies using the relative stabilities of the 2 and 3 carbocations to predict that the rate of formation of the major product is faster than the rate of formation of the minor product. 5. (15 points) Outline a synthetic route for the following molecule from the starting material given. Make sure to design the synthesis that will give the highest yield of the correct product.
HO H H OH
from
6. (32 points) Draw the products for the following reactions. Be careful to indicate stereochemistry and to draw all stereoisomers formed.
(a) 1. OsO 4, THF 2. Na2SO 3, H2O
(b) HBr ROOR
�(c)
1) O3 ,CH2Cl 2 2) H2O2, CH3COOH
7. (35 points) 3-methyl-1-butene can be used as a starting material for the three alcohols shown below.
i
OH
ii
HO
iii
HO
a) Supply the reagents in the boxes above the arrows. b) Choose either reaction ii or iii from part (a) and explain why the reaction is regioselective. Although a mechanism with arrows may help you answer this question, it is not a sufficient answer, nor is it required. You must explain why the regioselective step is regioselective. 8. (18 points) Outline a synthetic route for the following molecule from the starting material given. Make sure to design the synthesis that will give the highest yield of the correct product.
H3C H make Br H CH3 Br from
9. (24 points) You have learned two methods for incorporating halogens into molecules. Write the reagents required to perform each of the following transformations:
�(b)
Br (c)
OH
Br
10. (49 points) Write the major product of each of the following reactions. Write all stereoisomers that are formed.
(a) (c) CH3I + Na+NH 2
H 1) BH 3, THF 2) H2O2, NaOH, H2O NH3
(e)
H2O Br
11. (36 points) Which of the following reaction proceeds at a faster rate? Explain your choice in a few sentences. Keep in mind that the reactions may proceed at the same rate.
(c)
OH + Na+OCH 3 CH3 OH OCH3 + Na+OH
or
I + Na+OCH 3 CH3 OH OCH3 + Na+I
�(d)
I + H2O OH + HI
or
I + H2S SH + HI
12. (30 points) Consider the reaction of the alkene shown below with OsO4.
1) OsO4 2) H2O, Na2SO 3
(a) Draw the products of this reaction. (b) Draw the mechanism of this reaction with arrows. Only draw the mechanism of the first step, not the mechanism of the step involving the Na2SO3. (c) Are the products of this reaction enantiomers, diastereomers, or constitutional isomers? (d) Are the products of this reaction chiral? Would the product mixture be optically active? 13. (35 points) (a) Label the stereocenters in the bromoalkane below as R or S. (D is heavier than H, but lighter than C.)
D H
H Br
(b) Draw Newman projections of the staggered conformations of the bromoalkane shown above and indicate which is most stable and which is least stable. (CH3 is approximately three times larger than Br; D is the same size as H.)
�(c)When this bromoalkane is allowed to react with K+OC(CH3)3 two alkenes are formed. Assume that D and H do not differ in their reactivity. Draw the mechanism with arrows for this reaction showing how both alkenes are formed. (d) Based on the Newman projections you drew in part b and the mechanism you drew in part c, which alkene would you expect to be the major product and why? Once again, assume that D and H do not differ in their reactivity. 14. (30 points) Draw both chair conformations of the following molecules and indicate which is more stable.
(a)
(b)
15. (40 points) Most of the reactions covered this semester involve the reaction of the HOMO of a Lewis base with the LUMO of a Lewis acid. (a) In the reactions below indicate which substrate is the Lewis base and which is the Lewis acid.
(a)
+ Br2 CCl 4 H Br Br H + H Br Br H
(b)
+ I Na+SH SH
�(b) Choose one of the reactions above and draw the interaction between the HOMO of the Lewis base and the LUMO of the Lewis acid. Identify which orbital of each substrate is the HOMO and LUMO (i.e., the lone pair on atom A, the * orbital of the A-B bond etc.) 16. (30 points) Synthesize the following molecules from the indicated starting materials.
(a)
from CN