Professional Documents
Culture Documents
Bronquiectasias DX y TX
Bronquiectasias DX y TX
Uploaded by
EderEmmanuelMartinezRuizOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Bronquiectasias DX y TX
Bronquiectasias DX y TX
Uploaded by
EderEmmanuelMartinezRuizCopyright:
Available Formats
MEDICINE
REVIEW ARTICLE
BronchiectasisDiagnosis and Treatment
Jessica Rademacher, Tobias Welte
SUMMARY
Background: Radiologically evident bronchiectasis is seen in 30% to 50% of
patients with advanced chronic obstructive pulmonary disease (COPD). As
COPD is now becoming more common around the world, bronchiectasis is as
well.
Methods: We review pertinent articles published before May 2011 that were
retrieved by a selective PubMed search.
Results: The principles of treatment of bronchiectasis in patients who do not
have cystic fibrosis (non-CF bronchiectasis) are derived from the treatment
of other diseases: secretolytic and anti-infectious treatment are given as in
cystic fibrosis, while anti-obstructive treatment is given as in COPD. The
few randomized trials of treatment for non-CF bronchiectasis that have been
completed to date do not permit the formulation of any evidence-based recommendations. Many potential treatments are now under evaluation. Hypertonic
saline is often used because of its demonstrated benefit in CF, even though no
benefit has yet been shown for non-CF bronchiectasis. Phase II trials of inhaled
mannitol have yielded promising results, leading to phase III trials that are now
underway. There may be a future role for inhaled antibiotics, particularly in
patients colonized with Gram-negative pathogens. Inhaled tobramycin and
colistin are well established in clinical practice, though not approved for nonCF bronchiectasis; clinical trials of aztreonam, ciprofloxacin, and gentamicin
are ongoing. Macrolides seem to bring an additional benefit, though the studies
that documented this involved only small numbers of patients. Long-term treatment with inhaled antibiotics and/or macrolides is indicated only if a benefit is
seen within three months of the start of treatment (less sputum, no exacerbations).
Conclusion: A national registry of patients with bronchiectasis should be established to help us gain better knowledge of its prognostic factors and treatment
options.
Cite this as:
Rademacher J, Welte T: Bronchiectasisdiagnosis and treatment.
Dtsch Arztebl Int 2011; 108(48): 80915. DOI: 10.3238/arztebl.2011.0809
Klinik fr Pneumologie, Medizinische Hochschule Hannover:
Prof. Dr. med. Welte, Dr. med. Rademacher
Deutsches rzteblatt International | Dtsch Arztebl Int 2011; 108(48): 80915
he term bronchiectasis describes a permanent
dilation of the bronchi and bronchioles as a result
of destruction of the muscles and elastic connective
tissues. The disorder mostly starts with a narrowing of
the bronchial tree triggered by an infection, which may
lead to destruction of the epithelium if it becomes
chronic. The disruption of the mucociliary clearance
results in retention of secretions and predestines the
patient for further infections.
In the past, bronchiectasis mostly had infectious
causes, such as epidemics of pertussis, measles, and
influenza. Todays most common cause in developing
countries is the postinfectious route. The development
of antibiotic treatments and vaccines has resulted in a
continuous decrease in the number of cases of bronchiectasis with postinfectious causes in industrial countries. Currently, congenital causes of bronchiectasis are
seen more observed than postinfectious causes.
In Europe, bronchiectasis is common in patients with
cystic fibrosis (CF) (1). However, in this review article
we focus on patients with bronchiectasis in whom
cystic fibrosis was excluded (non-CF bronchiectasis).
This article aims to provide an overview over what
is currently known from studies about the diagnostic
evaluation and therapy of this heterogeneous pathology.
Method
We conducted a selective literature search on PubMed.
Relevant articles published before May 2011 were
included in this review.
Incidence and prevalence
Because high-resolution computed tomography
(HRCT) scanning is more commonly used nowadays,
bronchiectasis is diagnosed earlier and at earlier stages.
This has resulted in a seeming increase in the prevalence of bronchiectasis. The question of whether the
increase in the numbers of cases is caused by the ageing
population and the increase in chronic lung disorders
will have to remain unanswered.
Very few data on prevalence are currently available.
In New Zealand, the prevalence was reported to be
3.7/100 000 population; in the United States, the rate
was reported to be as high as 52/100 000 (2). Different
diagnostic methods (clinical versus CT) and a different
selection of patients may be responsible for this
discrepancy.
809
MEDICINE
TABLE 1
Etiology of bronchiectasis (modified from [7])
Category
Cause
Postinfectious
Viral
Bacterial
Fungal
Atypical mycobacteria
ABPA
COPD
Idiopathic traction
Posttuberculous fibrosis
Radiation fibrosis
Pulmonary fibrosis
Aspiration
Foreign body aspiration
Obstruction
Benign tumors
Enlarged lymph nodes
Amyloidosis
Celiac disease
Yellow nail syndrome
Youngs syndrome
Immunological defects
Primary
CVID
Agammaglobulinemia
Hyper-IgE syndrome
Secondary
Chemotherapy
Immunosuppressant therapy
Tumor
Congenital defects
Anatomical
Scoliosis
Marfan syndrome
Tracheobronchomegaly
Others
Primary ciliary dyskinesia
Kartagener syndrome
Alpha-1-antitrypsin deficiency
defect ENaC protein
Diffuse panbronchiolitis
Rheumatoid arthritis
Systemic lupus erythematodes
Chronic bowel disease
ABPA, allergic bronchopulmonary aspergillosis; COPD, chronic obstructive pulmonary disease;
ENaC, epithelial sodium channel; CIVD, common variable immunodeficiency
histologically shows notable metaplasias of the squamous epithelium, although an increased incidence in
malignancies has not been observed.
Postinfectious causes
Different respiratory infections can cause bronchiectasis, including:
Pertussis
Gram negative bacteria (Pseudomonas aeruginosa, Haemophilus influenzae)
Viruses (HIV, paramyxovirus, adenovirus, and
flu)
Tuberculosis
Atypical mycobacteria.
Bronchiectasis subsequent to infection with Mycobacterium avium is a typical finding in Lady Windermere syndrome. Patients suffering from this syndrome
are often older, immunocompetent women without a
history of smoking or previous pulmonary pathologies
(4).
Congenital causes
The most common congenital cause for non-CF bronchiectasis is a primary ciliary dyskinesia (PCD). Insufficient ciliary movement results in reduced clearance of
secretions, triggering an increase in the rate of infections in turn. In combination with a situs inversus, the
resultant pathology is known as Kartagener syndrome.
Its prevalence is 1/20 000.
A more recently discovered congenital cause is a
mutation of the ENaC gene, which results in a defective
epithelial sodium channel. A hyperactive sodium channel triggers a disturbance to the salt and water homeostasis of the respiratory mucosa (5).
Chronic obstructive pulmonary disease
Patients with advanced chronic obstructive pulmonary
disease (COPD) may have bronchiectasis; the literature
reports rates between 30% and 50% (6). These patients
more often suffer from dyspnea and show poorer lung
function (6). CT-morphologically, bronchiectasis in
COPD differs from classic bronchiectasis, since the
ectasis is less pronounced but the peribronchial infiltration is more pronounced. With the rising global
prevalence of COPD, bronchiectasis is of increasing
importance.
Pathophysiology and etiology
Different mechanisms (Table 1) lead to the development of bronchiectasis, but the pathophysiological end
stage is similar. In the beginning, patients usually have
damaged bronchial epithelium as a result of inflammation; the surrounding parenchyma is infiltrated by
inflammatory cells (Figure 1). The destruction of the
neighboring tissues results in dilation in the form of
cylindrical, varicose, or cystic distensions with destruction of the surrounding structures. This will in turn lead
to deficient mucociliary clearance. The result is retention of secretions, which in turn attracts bacterial colonization with chronic inflammation (3). Furthermore, a
thickening of the bronchial mucosa will ensue, which
810
Clinical features
Patients with bronchiectasis complain of chronic
cough, sputum production, and lethargy. Hemoptysis,
chest pain, weight loss, bronchospasm, dyspnea, and
impaired physical performance have also been observed (7). The often mentioned three-layer sputum
consisting of a foamy upper layer, mucous middle
layer, and viscous purulent bottom layer is pathognomonic, but does not always occur. Some patients are
symptom free in everyday life and become clinically
conspicuous only during exacerbations.
Many patients have regular exacerbations, the average is 1.5 per year. Exacerbation is defined as the
Deutsches rzteblatt International | Dtsch Arztebl Int 2011; 108(48): 80915
MEDICINE
presence of four or more of the symptoms listed in Box
1 (8). The loss of lung function in non-smokers with
bronchiectasis has been reported to be about 50 mL/
year. Factors that would imply disease progression are
frequent exacerbations, chronic colonization with
Pseudomonas aeruginosa, and confirmed systemic
inflammation. In case of severe bronchiectasis, pulmonary hypertension and systolic and diastolic left
ventricular dysfunction may develop.
Diagnostic evaluation
Investigating the colonization
Microbiological sputum analysis is a standard diagnostic procedure. Risk factors for colonization include
varicose or cystic bronchiectasis, a forced expiratory
volume in one second (FEV1) <80%, and age <14
years at first diagnosis (7). The most common
pathogens are Haemophilus influenzae, Pseudomonas
spp, and Streptococcus pneumoniae. In progressive disease with recurring exacerbations and negative sputum
results, bronchoscopy is indicated for the purpose of
specimen sampling.
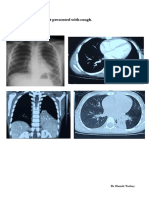
Imaging
The imaging method of choice is high-resolution computed tomography (Figure 2). Often the type and localization of the radiological changes can provide indications of the pathogenesis. Bronchiectasis in the
proximal airways is typical of allergic bronchopulmonary aspergillosis; multiple nodular bronchiectasis may
indicate infection with Mycobacterium avium complex.
Therapy
The treatment for bronchiectasis is mostly based on experiences gained from the treatment of COPD and CF
(Figure 3). Whether these concepts actually translate
has not been studied. Only few controlled studies exist,
so that for non-CF bronchiectasis, hardly any evidencebased recommendations can be made. The aims of
treatment for bronchiectasis are:
Treating the underlying disease
Improving mucociliary clearance or drainage
ofsecretions
Treating the infection
Treating airway obstruction
Treating the chronic inflammation that leads to
disease progression.
Treating the underlying disease
If possible, the underlying disease should be treated
first. This primarily applies to immunodeficiency
syndromes. In hypogammaglobulinemia, substitution
treatment with immunoglobulins can be given (0.4 g/kg
body weight every 46 weeks).
Draining secretions
Breathing therapy and physiotherapeutic measures are
the basic treatments for bronchiectasis, to improve
drainage of secretions and deal with dyspnea. The
mainstay of treatment is sufficient administration of
Deutsches rzteblatt International | Dtsch Arztebl Int 2011; 108(48): 80915
b
Figure 1: Bronchiectasis with chronically and acutely inflamed mucosa with accompanying
fibrosis
fluids for secretolytic purposes. This can be supported
by inhalation of hypertonic saline solution. Especially
inhaling hyperosmolar solutions has been found to be
beneficial. The raised salt concentration results in an
osmotic penetration of fluid into the secretions, thus
improving their rheological properties, which in turn
results in faster and more effective clearance. Studies
using 7% saline for inhalation by CF patients have
shown improved lung function and secretion clearance
(9). In non-CF bronchiectasis, reduced sputum viscosity was found in a small number of patients (n=24) during a non-exacerbation period (10).
In contrast to other hyperosmolar solutions, mannitol has the advantage of a longer half life within the
airways. In an open label, non-controlled study over 12
days, quality of life, lung function, and sputum viscosity were notably improved (11). A disadvantage of
mannitol is the fact that hyperresponsiveness increases
during inhalation. Currently, further studies are being
811
MEDICINE
BOX 1
Symptoms of exacerbation
Increase of sputum with cough
Increased dyspnea
Raised temperature >38 C
Increased wheezing
Lowered physical resilience
Fatigue
Deterioration in lung function
Radiological signs of infection
A minimum of 4 symptoms are the defining criteria for
an exacerbation
(modified from [8])
Figure 2: Bronchiectasis CT-morphology
conducted in order to gain licensing approval for the
substance.
Vaccinations
No randomized studies of vaccinations exist for this
group of patients. The effect of annual flu vaccination
has been proved for other chronic airway disorders,
such as COPD, and translates to patients with bronchiectasis. Data from smaller cohort have shown that
the pneumococcal vaccine is beneficial, although a
final conclusion cannot currently be drawn. In principle, the recommendation is to follow the vaccination
guidelines issued by Germanys Standing Vaccination
Committee (STIKO) for patients with chronic pulmonary disorders.
Antibiotics
In an acute exacerbation of bronchiectasis, antibiotics
should be given if an increase in dyspnea and sputum
volume is observed and the sputum has assumed a
yellow-green or green tinge.
If the patient is known to have chronic colonization
with respiratory pathogens, targeted treatment should
be started while taking into consideration the latest
antibiogram. If no microbiological result is available
then a broad spectrum antibiotic should be selected for
initial treatment. This should include treatment for
Pseudomonas strains, since these range among the
pathogens particularly in severely ill patients and determine the prognosis.
In the outpatient setting, the fluoroquinolones levofloxacin or ciprofloxacin are the only available options.
It needs to be borne in mind, however, that ciprofloxacin is not sufficiently effective against Pneumococci,
the most common pathogens in community acquired
pneumonia. Pletz et al. reported the case of a patient
with bronchiectasis in whom consecutive courses of
treatment failed in the presence of ciprofloxacin resistant Pneumococci (e1).
812
In hospital inpatients, the range of substances that
are effective against Pseudomonas spp is wider (carbapenem, cephalosporins with activity against Pseudomonas, ureidopenicillins). Whether combination therapy using a beta-lactam with an aminoglycoside or fluoroquinolone is superior to monotherapy with a substance that is active against Pseudomonas is the subject
of controversial debate. Pseudomonas infections
should be treated for 1014 days. In patients not at risk
from Pseudomonas aeruginosa, treatment with aminopenicillin/inhibitor or third-generation cephalosporins
is recommended. The duration of treatment is usually
seven days. Attempts to diagnose the pathogen should
be made before antibiotics are given, and the antibiotic
treatment should be tailored accordingly.
The importance of antibiotics treatment outside
exacerbations is the subject of controversy. Thus far,
attempts to reduce the amount of pathogens by means
of long-term oral antibiotic treatment, and to lower the
rate of exacerbations, have remained unsuccessful. The
development of resistance patterns in patients receiving
long-term therapy may be important in this context.
Inhaled antibiotics are standard treatment for
patients with CF who have been colonized with Pseudomonas aeruginosa (12). Since 25% of patients with
non-CF bronchiectasis are colonized with Pseudomonas aeruginosa, this therapeutic principle may offer an
advantage in this setting. In the meantime, smaller
studies have shown the importance of inhaled antibiotics in non-CF bronchiectasis. Significant clinical
improvement has been shown, with a reduced density
of pathogens and eradication of Pseudomonas
aeruginosa in up to 35% of cases (13). Patients receiving treatment with inhaled tobramycin had fewer symptoms and an improved quality of life (14). Inhaled
colistin led to improvements in lung function and
quality of life (15); furthermore, a reduction in
inpatient admissions and exacerbations has been
reported (16). Inhaled aztreonam lowered the rate of
Deutsches rzteblatt International | Dtsch Arztebl Int 2011; 108(48): 80915
MEDICINE
FIGURE 3
TABLE 2
Selection of the most researched inhaled antibiotics
Substance
Results
Source
Tobramycin
Eradication in 1335%, reduction of pathogenic
load, improved lung function
(13)
(14)
Colistin
Rise in FEV1, eradication in 3 of 18 cases, fewer
exacerbations, reduction of pathogenic load
(15)
(16)
Aztreonam
CF: improved lung function, longer interval to
exacerbation
(17)
Liposomal ciprofloxacin
Ciprofloxacin
Reduction of pathogenic load
Reduction of pathogenic load
(e3)
(e4)
Gentamycin
Fewer exacerbations, improved quality of life
Eradication of P.a. in 30.8 % improved quality of
life
(19)
(18)
CF, cystic fibrosis; P. a., Pseudomonas aeruginosa;
FEV, forced expiratory volume in one second
Germany) compared with conventional treatment with
metered-dose aerosol inhalers or powder inhalers.
Therapeutic scheme for bronchiectasis (modified from [e2])
exacerbations, reduced symptoms, and improved lung
function in patients with CF (17). A study of inhaled
aztreonam in non-CF bronchiectasis is in the planning
stages.
A liposomal preparation of inhaled ciprofloxacin
confirmed the reduced pathogenic load of Pseudomonas aeruginosa (e3); the same was confirmed for
powder inhalation of ciprofloxacin (e4). Further studies
of inhaled amikacin and intratracheal instillation of
fosfomycin/tobramycin are expected.
In a randomized controlled study, inhaled gentamycin led to eradication of Pseudomonas aeruginosa in
30.8% of cases and prolonged the interval to the next
exacerbation (120 days versus 61.5 days) (18). Another
study of inhaled gentamycin found a lowered rate of
exacerbations and improved quality of life (19). Table 2
provides an overview of inhaled antibiotics.
Anti-obstructive therapy
If a patients airways are obstructed, anti-obstructive
treatment similar to COPD should be considered. Parasympatholytics and beta-sympathicomimetics constitute the treatment of choice. Long-acting substances
(tiotropium bromide or salmeterol/formoterol) seem
superior to short-acting substances. Proof of superiority
is lacking for inhalation therapy with compression or
ultrasound nebulizers (both of which are popular in
Deutsches rzteblatt International | Dtsch Arztebl Int 2011; 108(48): 80915
Inhibiting inflammation
Oral corticosteroids are often administered in acute
exacerbations of bronchiectasis, in analogy to acute exacerbations of COPD. For inhaled steroids, long-term
usage seems to confer benefits. Tsang et al. showed in a
study of 73 patients with non-CF bronchiectasis a reduction in the exacerbation rate and sputum production
when using inhaled steroids (20). Randomized studies
are, however, lacking.
Macrolide antibiotics such as azithromycin have a
potent anti-inflammatory effect in addition to their antibacterial effects. They reduce the production of proinflammatory cytokines. The cytokines act as chemokines for neutrophils and effect an expression of
adhesion molecules, which neutrophils require for their
migration from the bloodstream to the interstitium. Furthermore, in addition to their regular bacteriostatic
effects, macrolides inhibit the production of biofilms
by Pseudomonas aeruginosa (independently of their
antibiotic effectiveness) (21).
In the therapy of neutrophil-dominated, chronic
inflammatory pulmonary disorders, such as diffuse
panbronchiolitis (DPB) or CF, macrolides are already
in use, successfully and without notable side effects.
The treatment with macrolide antibiotics has led to a
reduction in the amount of sputum and improved 5-year
survival in patients with non-CF bronchiectasis too
(22). At this point in time, however, neither inhaled
antibiotics nor macrolides are licensed for the treatment
of patients with non-CF bronchiectasis.
Allergic bronchopulmonary aspergillosis
Allergic bronchopulmonary aspergillosis (ABPA) is a
rare but typical complication in bronchiectasis. The
813
MEDICINE
BOX 2
Diagnostic criteria for allergic
bronchopulmnaory aspergillosis
1. Asthma
2. Skin reactions*1
3. Raised total IgE in serum (> 1 000 ng/L)*1
4. Raised specific IgE and/or IgE to Aspergillus fumigatus
in serum*1
5. Bronchiectasis*1 *2
6. Radiologically confirmed infiltrate
7. Eosinophilia in blood (> 500/L)
8. Precitpitating antibodies to Aspergillus fumigatus
(modifed from Agarwal [23])
*1 Obligatory criterion for defining the diagnosis.
*2 If criteria 14 are met but no bronchiectasis is found, the finding is
taken to be seropositive allergic bronchopulmonary aspergillosise.
underlying pathophysiology is a sensitization to Aspergillus fumigatus, which leads to a CD4+/TH2 mediated
inflammatory reaction. Typical symptoms include a
raised body temperature, weight loss, a drop in FEV1,
and pulmonary infiltrates on the radiograph.
Bronchiectasis can be a sequela of ABPA, but it can
also predispose to ABPA. The clinical diagnosis is difficult and is based on criteria set out by Greenberger
(overview in Agarwal [23]) (Box 2).
Acute exacerbation of ABPA usually requires treatment with systemic steroids for a long period of time
(23). In order to prevent recurrence, long-term oral
therapy with itraconazole is indicated in patients with
pulmonary colonization; several studies have shown
KEY MESSAGES
Thanks to improved antibiotic treatments and vaccination programs, more congenital than postinfectious
causes of bronchiectasis have been observed.
Different mechanisms lead to bronchiectasis, but the
pathophysiological end stage of inflammation and destruction is the same.
For diagnostic purposes, a sputum specimen should be
sampled. If imaging is used then high-resolution computed tomography is the method of choice.
The treatment of the underlying disease should always
be considered. The basics of treatment include breathing measures and physiotherapeutic measures. Treatment of exacerbations should be based on the diagnostic test results for the pathogen and the antibiogram.
Addititional administration of inhaled antibiotics or
macrolides should be considered in the individual case
scenario.
Surgical measures are available for circumscribed
bronchiectasis.
814
the effectiveness of this treatment in CF. Inhaled
amphotericin B is the subject of studies. Individual reports have documented successful treatment with a
monoclonal antibody against IgE (omaluzimab).
Surgical therapy
Surgery is the method of choice in unilateral and localized bronchiectasis. Several studies have shown that
resection of the bronchiectasis improved symptoms
(24). In the different studies, mortality varied from 1%
to 8.6% (e5, e6), the rate of surgical complications was
8.825% (e7). Complications included pneumonias,
postoperative hemorrhage, atelectasis, bronchopulmonary fistula, and wound infection. In particular cases,
the resection of bilateral bronchiectasis may be the aim,
but the lesions should be limited and completely resectable (24). In severe complications, such as life threatening hemorrhage or fungal infection, surgical therapy
can be used as the method of last resort.
Hemoptysis, which is mostly caused by bleeds from
hypertrophied vessels of the inflamed mucosa, can be
controlled by bronchial artery embolization (coiling) if
required. This should only be done in specialized centers.
Lung transplantation in advanced disease
Lung transplantion can be a useful intervention in very
advanced non-CF bronchiectasis. It is of vital importance to identify the right time for putting the patient on
the transplant list. In accordance to the guidelines, the
following criteria should be met:
FEV1 < 30 % and an exacerbation with inpatient
admission to intensive care, or
More than three exacerbations per year, or
Recurrent pneumothorax, or
Hemoptysis requiringand receivingintervention (25).
A double lung transplant is the method of choice in
more than 90% of cases. In case only one lung is transplanted, there is a risk that pathogens are transferred
from the native lung into the transplanted lung. Experiences from large centers have shown that he long-term
prognosis does not differ much from that of other indications, with 5-year survival rates between 55% and
60% (e8).
Conflict of interest statement
Dr Rademacher has received honoraria for speaking from Forest and MSD.
Professor Welte has received honoraria for acting as an adviser from Novartis,
Bayer Pharma, and Gilead.
Manuscript received on 20 May 2011, revised version accepted on 27 July
2011.
Translated from the original German by Dr Birte Twisselmann.
REFERENCES
1. Bilton D: Update on non-cystic fibrosis bronchiectasis. Curr Opin
Pulm Med 2008; 14: 5959.
2. Twiss J, Metcalfe R, Edwards E, et al.: New Zealand national incidence of bronchiectasis too high for a developed country. Arch
Dis Child 2005; 90: 73740.
Deutsches rzteblatt International | Dtsch Arztebl Int 2011; 108(48): 80915
MEDICINE
3. Fuschillo S, De Felice A, Balzano G: Mucosal inflammation in
idiopathic bronchiectasis: cellular and molecular mechanisms. Eur
Respir J 2008; 31: 396406.
16. Dhar R, Anwar GA, Bourke SC, et al.: Efficacy of nebulised colomycin
in patients with non-cystic fibrosis bronchiectasis colonised with
Pseudomonas aeruginosa. Thorax 2010; 65: 553.
4. Dhillon SS, Watanakunakorn C: Lady Windermere syndrome: middle
lobe bronchiectasis and Mycobacterium avium complex infection
due to voluntary cough suppression. Clin Infect Dis 2000; 30:
5725.
17. McCoy KS, Quittner AL, Oermann CM, et al.: Inhaled aztreonam lysine for chronic airway Pseudomonas aeruginosa in cystic fibrosis.
Am J Respir Crit Care 2008; 178: 9218.
5. Fajac I, Viel M, Sublemontier S, et al.: Could a defective epithelial
sodium channel lead to bronchiectasis. Respir Res. 2008; 9: 46.
18. Murray MP, Govan JR, Doherty CJ, et al.: A randomized controlled
trial of nebulized gentamicin in non-cystic fibrosis bronchiectasis.
Am J Respir Crit Care Med 2011; 183: 4919.
6. Patel IS, Vlahos I, Wilkinson TM, et al.: Bronchiectasis, exacerbation
indices, and inflammation in chronic obstructive pulmonary disease.
Am J Respir Crit Care Med 2004; 15; 170: 4007.
19. Antoniu SA, Trofor AC: Inhaled gentamicin in non-cystic
fibrosis bronchiectasis: effects of long-term therapy. Expert Opin
Pharmacother 2011; 12: 11914.
7. Goeminne P, Dupont L: Non-cystic fibrosis bronchiectasis: diagnosis
and management in 21st century. Postgrad Med J 2010; 86:
493501.
20. Tsang KW, Tan KC, Ho PL, et al.: Inhaled fluticasone in bronchiectasis: a 12 month study. Thorax. 2005; 60: 23943.
8. ODonnell AE, Barker AF, Ilowite JS, et al.: Treatment of idiopathic
bronchiectasis with aerosolized recombinant human DNase I.
rhDNase Study Group.Chest 1998; 113: 132934.
9. Donaldson SH, Bennett WD, Zeman KL, et al.: Mucus clearance and
lung function in cystic fibrosis with hypertonic saline. N Engl J Med
2006; 354: 24150.
10. Kellett F, Redfern J, Niven RM: Evaluation of nebulised hypertonic
saline (7%) as an adjunct to physiotherapy in patients with stable
bronchiectasis. Resp Med 2005; 99: 2731.
11. Daviskas E, Anderson SD, Gomes K, et al.: Inhaled mannitol for the
treatment of mucociliary dysfunction in patients with bronchiectasis: effect on lung function, health status and sputum. Respirology
2005; 10: 4656.
12. Ratjen F, Munck A, Kho P, et al.: Treatment of early Pseudomonas
aeruginosa infection in patients with cystic fibrosis: the ELITE Trial.
Thorax 2010; 65: 28691.
13. Drobnic ME, Su P, Montoro JB, et al.: Inhaled tobramycin in
non-cystic fibrosis patients with bronchiectasis and chronic bronchial
infection with Pseudomonas aeruginosa. Ann Pharmacother 2005;
39: 3944.
14. Scheinberg P, Shore E: A pilot study of the safety and efficacy of
tobramycin solution for inhalation in patients with severe bronchiectasis. Chest 2005; 127: 14206.
15. Steinfort DP, Steinfort C: Effect of long-term nebulised colistin of
lung function and quality of life in patients with chronic bronchial
sepsis. Intern Med J 2007; 37: 4958.
Deutsches rzteblatt International | Dtsch Arztebl Int 2011; 108(48): 80915
21. Shinkai M, Henke MO, Rubin BK: Macrolide antibiotics as immunomodulatory medications: proposed mechanisms of action. Pharmacol Ther 2008; 117: 393405.
22. Anwar GA, Bourke SC, Afolabi G, et al.: Effects of long-term lowdose azithromycin in patients with non-CF bronchiectasis. Respir
Med 2008; 102: 14946.
23. Agarwal R: Allergic bronchopulmonary aspergillosis. Chest 2009;
135: 80526.
24. Bagheri R, Haghi SZ, Fattahi Masoum SH, et al.: Surgical management of bronchiectasis: analysis of 277 patients. Thorac Cardiov
Surg 2009; 58: 2914.
25. Orens JB, Estenne M, Arcasoy S, et al.: Pulmonary Scientific Council of the International Society for Heart and Lung Transplantation.
International guidelines for the selection of lung transplant candidates: 2006 updatea consensus report from the Pulmonary
Scientific Council of the International Society for Heart and Lung
Transplantation. J Heart Lung Transplant 2006; 25: 74555.
Corresponding author:
Prof. Dr. med. Tobias Welte
Abteilung Pneumologie
Carl-Neuberg-Str. 1
30625 Hannover, Germany
welte.tobias@mh-hannover.de
For eReferences please refer to:
www.aerzteblatt-international.de/ref4811
815
MEDICINE
REVIEW ARTICLE
BronchiectasisDiagnosis and Treatment
Jessica Rademacher, Tobias Welte
eReferences
e1. Pletz MW, McGee L, Burkhardt O, et al.: Ciprofloxacin treatment
failure in a patient with resistant Streptococcus pneumoniae
infection following prior ciprofloxacin therapy. Eur J Clin Microbiol
Infect Dis. 2005; 24: 5860.
e2. Rademacher J, Pletz MW, Welte T: Behandlung nicht mit
zystischer Fibrose assoziierter Bronchiektasen (Non-CFBronchiektasen. Internist (Berl) 2010; 51: 15105.
e3. Bilton D, Bruinenberg P, Otulana B, et al.: Inhaled liposomal ciprofloxacin hydrochloride significantly reduces sputum pseudomonas
aeruginosa density in CF and Non-CF bronchiectasis. Am J Respir
Crit Care Med 179; 2009: A3214.
e4. Wilson R, Welte T, Polverino E, et al.: Randomized, placebocontrolled, double-blind, multi-center study to evaluate the safety
and efficacy of Ciprofloxacin dry powder for inhalation (Ciprofloxacin DPI) compared with placebo in patients with non-cystic fibrosis bronchiectasis. Am J Respir Crit Care Med 183; 2011: A6407.
e5. Agasthian T, Deschamps C, Trastek VF, et al.: Surgical management of bronchiectasis. Ann Thorac Surg 1996; 62: 9768.
e6. Kutlay H, Cangir AK, Enn S, et al.: Surgical treatment in bronchiectasis: analysis of 166 patients. Eur J Cardiothorac Surg
2002; 21: 6347.
e7. Fujimoto T, Hillejan L, Stamatis G.: Current strategy for surgical
management of bronchiectasis. Ann Thorac Surg 2001; 72:
17115.
e8. Weiss ES, Allen JG, Meguid RA, et al.:The impact of center volume on survival in lung transplantation: an analysis of more than
10,000 cases. Ann Thorac Surg 2009; 88: 106270.
Deutsches rzteblatt International | Dtsch Arztebl Int 2011; 108(48): 80515
MEDICINE
BOX 2
Diagnostic criteria for allergic
bronchopulmnaory aspergillosis
1. Asthma
2. Skin reactions*1
3. Raised total IgE in serum (> 1 000 ng/L)*1
4. Raised specific IgE and/or IgE to Aspergillus fumigatus
in serum*1
5. Bronchiectasis*1 *2
6. Radiologically confirmed infiltrate
7. Eosinophilia in blood (> 500/L)
8. Precitpitating antibodies to Aspergillus fumigatus
(modifed from Agarwal [23])
*1 Obligatory criterion for defining the diagnosis.
*2 If criteria 14 are met but no bronchiectasis is found, the finding is
taken to be seropositive allergic bronchopulmonary aspergillosise.
KEY MESSAGES
Thanks to improved antibiotic treatments and vaccination programs, more congenital than postinfectious
causes of bronchiectasis have been observed.
Different mechanisms lead to bronchiectasis, but the
pathophysiological end stage of inflammation and destruction is the same.
For diagnostic purposes, a sputum specimen should be
sampled. If imaging is used then high-resolution computed tomography is the method of choice.
The treatment of the underlying disease should always
be considered. The basics of treatment include breathing measures and physiotherapeutic measures. Treatment of exacerbations should be based on the diagnostic test results for the pathogen and the antibiogram.
Addititional administration of inhaled antibiotics or
macrolides should be considered in the individual case
scenario.
Surgical measures are available for circumscribed
bronchiectasis.
Deutsches rzteblatt International | Dtsch Arztebl Int 2011; 108(48): 80915
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5814)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1092)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (845)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (590)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (897)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (540)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (348)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (822)
- Crack The Core Exam - Volume 1 4th PDFDocument551 pagesCrack The Core Exam - Volume 1 4th PDFJavier Palma Rojas67% (6)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (122)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (401)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- PASSMEDICINE MCQs-RESPIRATORY MEDICINEDocument111 pagesPASSMEDICINE MCQs-RESPIRATORY MEDICINEHashim Ahmad86% (7)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- Nelson Textbook of Pediatrics - 20th Edition Table of ContentsDocument27 pagesNelson Textbook of Pediatrics - 20th Edition Table of ContentsJesse Bonne Cataylo Garillos43% (7)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- 100 Concepts - MCBDocument316 pages100 Concepts - MCBNisreen SalameNo ratings yet
- Bronchiectasis - Monograph 2011Document265 pagesBronchiectasis - Monograph 2011Misael ClintonNo ratings yet
- Respiratory Questions by DR - OkashaDocument96 pagesRespiratory Questions by DR - Okashamohammed okashaNo ratings yet
- Acute Sinusitis: Practice EssentialsDocument22 pagesAcute Sinusitis: Practice EssentialsAna-Maria DuMiNo ratings yet
- Lung Pathology BOARDDocument315 pagesLung Pathology BOARDShazaan NadeemNo ratings yet
- Case Studies in Veterinary ImmuneologyDocument209 pagesCase Studies in Veterinary ImmuneologyNathalia gallegoNo ratings yet
- Kartagener SyndromeDocument4 pagesKartagener Syndromemilkah mwauraNo ratings yet
- DextrocardiaDocument11 pagesDextrocardiajuwitavalenNo ratings yet
- MRCP 2Document26 pagesMRCP 2riyaz414No ratings yet
- AnswersDocument401 pagesAnswersmedstudentNo ratings yet
- Validation of A Health-Related Quality of LifeDocument8 pagesValidation of A Health-Related Quality of LifeMarcellaNo ratings yet
- Kartagener 'S Syndrome: Review of A Case SeriesDocument8 pagesKartagener 'S Syndrome: Review of A Case SeriesNabila RizkikaNo ratings yet
- Respiratory PhysiologyDocument38 pagesRespiratory PhysiologyskNo ratings yet
- Aiims - Pediatrics Q&ADocument207 pagesAiims - Pediatrics Q&AqqypprcyxyNo ratings yet
- 2 - Respiratory Notes MRCP-I Passmedicine 2017Document92 pages2 - Respiratory Notes MRCP-I Passmedicine 2017kathi raja sekhar100% (1)
- A Case Report On Kartagener's SyndromeDocument3 pagesA Case Report On Kartagener's SyndromeBaru Chandrasekhar RaoNo ratings yet
- Symptoms: Kartagener Syndrome Is A Type ofDocument5 pagesSymptoms: Kartagener Syndrome Is A Type ofalexandreopatoNo ratings yet
- Asthma: Differential Diagnosis and ComorbiditiesDocument9 pagesAsthma: Differential Diagnosis and ComorbiditiesQuel PaivaNo ratings yet
- Chest Injury and Its TypesDocument14 pagesChest Injury and Its TypesKoochi PoojithaNo ratings yet
- PDF Case Studies in Veterinary Immunology 1St Edition Laurel Gershwin Author Ebook Full ChapterDocument53 pagesPDF Case Studies in Veterinary Immunology 1St Edition Laurel Gershwin Author Ebook Full Chapterjaime.currie230100% (3)
- 11.multisystem Process Disorders 5 Blocks @eduwaves360Document1,060 pages11.multisystem Process Disorders 5 Blocks @eduwaves360AbigailNo ratings yet
- Primary Ciliary DyskinesiaDocument23 pagesPrimary Ciliary DyskinesiaSarit LevinskyNo ratings yet
- DR Hamdi's Cases 52.compressedDocument159 pagesDR Hamdi's Cases 52.compressedSlm BngshNo ratings yet
- Test Bank For Color Textbook of Histology 3rd Edition Leslie P GartnerDocument12 pagesTest Bank For Color Textbook of Histology 3rd Edition Leslie P Gartnerbrennaquani2mNo ratings yet
- Chapter 15 Lung Pathology ErnieDocument20 pagesChapter 15 Lung Pathology ErnieZandra Lyn AlundayNo ratings yet
- Clinical Manifestations and Diagnosis of Bronchiectasis in AdultsDocument16 pagesClinical Manifestations and Diagnosis of Bronchiectasis in AdultsDAT TRANNo ratings yet
- 'COMPREHENSIVE SOLUTIONS' Combi - Dr. Afsar Khan PDFDocument587 pages'COMPREHENSIVE SOLUTIONS' Combi - Dr. Afsar Khan PDFwattuser KassoNo ratings yet