Professional Documents
Culture Documents
Polystyrene Methods 2520of 2520production
Polystyrene Methods 2520of 2520production
Uploaded by
Amal BudimanOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Polystyrene Methods 2520of 2520production
Polystyrene Methods 2520of 2520production
Uploaded by
Amal BudimanCopyright:
Available Formats
Production of Polystyrene
PROCESS DESCRIPTION
Manufacturing process:
Polystyrene is manufactured by the addition polymerization of styrene monomer
unit. Dow Chemical is the world's largest producer with a total capacity of 1.8 million
metric tones in the USA, Canada, and Europe (1996 figures). The main manufacturing
route to styrene is the direct catalytic dehydrogenation of ethyl benzene:
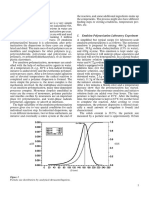
The reaction shown above has a heat of reaction of -121 KJ/mol (endothermic).
Nearly 65% of all styrene is used to produce polystyrene.
The overall reaction describing the styrene polymerization is:
This reaction is carried out in an inert organic solvent environment, which
provides the reaction medium for this cationic polymerization reaction
Production of Polystyrene
The different methods available for styrene polymerization are:
1. Solution (bulk) polymerization.
2. Emulsion polymerization.
3. Suspension polymerization.
Solution (bulk) polymerization:
Solution (bulk) polymerization is commonly referred to as mass polymerization in
the industry. The vast majority of all polystyrene produced today is produced via this
technology. The common solvents used in this process are the styrene monomer itself and
ethyl benzene. The two types of mass polymerization are batch and continuous, of which
continuous mass is by far the most popular.
Batch mass polymerization consists of a polymerization section containing
agitated vessels polymerizing up to 80% conversion in a batch method. The polymerized
solution is then pumped to a batch finishing section for either devolatisation or plate and
frame final polymerization and grinding.
The most widely used process for polymerization of polystyrene today is the
continuous mass process. This solution is continuously prepared in a holding vessel and
will then be injected into the reactor system.
Production of Polystyrene
Typical feed to the first reactor would consist of 50 weight percent styrene
monomer, 100 ppm water (based on styrene weight), 2000 ppm boron trifluoride (based
on styrene weight), with the balance being organic solvent. The polymerization reaction
gives off heat that is carried away from the reactors by jacketing them with a heat transfer
fluid. The temperature of the reactants should not vary by more than 15 0C throughout
the reactor series. Temperature control is very important in this reaction because as the
reaction temperature increases, the average molecular weight of the polystyrene
decreases.
The reaction temperature range is 40-70 0C. Temperature can also be
controlled by intermediate shell and tube heat exchangers. Monomer conversions of up to
85wt% polystyrene are obtainable in these reactors.
Emulsion polymerization: Emulsion polymerization is generally used for
polymerization of styrene with other monomers or polymers. It is not a generally
commercially accepted method of producing crystal polystyrene or high impact
polystyrene.
Emulsion
polymerization
is
carried
out
similarly
to
suspension
polymerization except that the monomer droplets are microscopic in size.
Suspension polymerization:
This is also called pearl polymerization. It has proved highly efficient for largescale production of polymers of high average molecular weight. By variation of the
polymerization condition it is possible to produce a range of polymers with different
properties and processing characteristics so that a number of grades are offered by the
manufacturers to meet the differing requirements of the conversion process and the final
product.
There are many different ways of making polystyrene using suspension process.
Most producers use a batch process, although there is no technical reasons why a
continuous process could not work. In the suspension process a number of small styrene
drops 0.15-0.50mm in diameter are suspended in water. The reaction occurs within these
Production of Polystyrene
drops. To aid in the formation of proper size drops a suspending agent is used, and to
keep them at that size a stabilizing agent is added. A catalyst is used to control the
reactionrate.
Suspension polymerization offers considerable advantages over the single phase
techniques in so far that heat removal control is no longer a problem but there are
disadvantages
such
as
the
need
to
use
dispersing
agent.
Detailed process of suspension polymerization:
Suspension polymerization is a batch system popular for speciality grades of
polystyrene. It can be used to produce either crystal or high impact grades. In impact
production, the styrene and rubber solution is bulk polymerized beyond phase inversion
and is then suspended in water to create oil in water suspension utilizing soaps and
suspending agents. The suspended droplets are then polymerized to completion, utilizing
initiator and a staged heating profile. The water phase is used as a heat sink and heat
transfer medium to a temperature-controlled jacket. For the production of crystal
polystyrene the styrene monomer itself is suspended and polymerized via the same
mechanism.
The requirements of polymerization are:
a] Initiator. b] Suspending agent. c] Stabilizing agent. d] Catalyst
Initiators: The initiators generally used are benzoyl peroxide and t-butylhydroperoxide.
Suspending agent: To aid in the formation of the proper size drops a suspending agent is
added. Some typical suspending agents are methylcellulose, ethyl cellulose and
polyacrylic acids. Their concentration in the suspension is between 0.01-0.5% of
monomer
charged.
Stabilizing agent: To keep the drops at proper size, a stabilizing agent is added. The
Production of Polystyrene
stabilizing agents are often insoluble inorganic such as calcium carbonate, calcium
phosphates or bentonite clay. They are present in small amount than the suspending
agents.
Catalyst: A catalyst is used to control the reaction rate. The catalysts are usually
peroxides. The most common ones are benzoyl, diacetyl, lauroyl, caproyl and tert-butyl.
Their
The
concentration
ratio
of
varies
monomer
from
to
0.1-0.5%
dispersing
of
medium
the
is
monomer
between
10
charged.
and
40%.
Polymerization temperature: Polymerization of styrene occurs at temperature range of 90950C.
Process description: The suspension method is carried out in large reactors equipped
with agitators, the styrene monomer being maintained in the aqueous phase as droplets
with a diameter varying between 0.4-1mm by use of a dispersing agent such as partially
hydrolyzed polyvinyl acetate, inorganic phosphates or magnesium silicates. To reduce the
cycle time of the reactors, the entering water and styrene will be preheated. The
temperatures of the input streams will be sent so as to obtain the desired reaction
temperature. The water entering the reactor will be heated to 950C. The bulk of the
styrene is to be heated to 850C before being charged. This is done in a vertical double
pipe heat exchanger, which is directly above the reactor. To prevent the polymerization
from occurring in the heat exchanger or piping system, there are to be no obstructions
between
this
heat
exchanger
and
the
reactor.
The catalyst, rubber stabilizer, and suspending agent are premixed in styrene and
discharged by gravity into the reactor. This mixture will not be preheated, since it might
polymerize. Typical water to monomer ratios is 1:1 to 3:1. A combination of two or more
initiators is used with a programmed reaction temperature to reduce the polymerization
time
to
minimum
for
given
amount
of
residual
styrene.
Purification steps and Extrusion: If the water can be removed using physical separation
processes, then the styrene and the other impurities dissolved in it will also be discharged.
Production of Polystyrene
A centrifuge with a washing step will be used to do this. The material leaving the
centrifuge has 1-5% water.
The final purification step is drying. The polystyrene leaving this unit must meet
the specifications set. (0.03% water). Then it is passed through a devolatisation extruder
to
remove the volatile residues and to
convert
the polymer into
pellets.
It was assumed that 3% of polystyrene would be removed from the process in
airveying, drying, centrifuging, transferring, or as bad as bad product. At least 95% of
that which is lost in processing must be intercepted before it leaves the plant. Most of it
can be removed and sold as off-grade material. This waste is split among the various
streams leaving the processing area.
You might also like
- Suspension Polymerization of Methyl MethacrylateDocument4 pagesSuspension Polymerization of Methyl MethacrylateZeenat RanaNo ratings yet
- Polybutadiene Rubber Project ReviewDocument20 pagesPolybutadiene Rubber Project ReviewJaveed Ali100% (1)
- Solution Polymerisation of StyreneDocument4 pagesSolution Polymerisation of StyreneMatthew Ong75% (4)
- Experiment 1Document11 pagesExperiment 1Kelvin LimNo ratings yet
- Styrene Production From EthylbenzeneDocument10 pagesStyrene Production From EthylbenzeneChegg ChemNo ratings yet
- Polymer Next PDFDocument31 pagesPolymer Next PDFali100% (2)
- Advanced Pharmaceutical analysisFrom EverandAdvanced Pharmaceutical analysisRating: 4.5 out of 5 stars4.5/5 (2)
- UPTU New Transcript 161015Document2 pagesUPTU New Transcript 161015Anish Kumar0% (1)
- Styrene BulkPolymerizationDocument2 pagesStyrene BulkPolymerizationNitin HansaliaNo ratings yet
- Styrene MonomerDocument13 pagesStyrene MonomerSerkan Gecim100% (1)
- Basics of Polystyrene ProductionDocument3 pagesBasics of Polystyrene ProductionRaj Sunil KandregulaNo ratings yet
- c06s06 3Document15 pagesc06s06 3unghoangduyNo ratings yet
- Unit 1 Techniques of PolymerizationDocument11 pagesUnit 1 Techniques of PolymerizationShevaniga SridharNo ratings yet
- Plastic Industries: An OverviewDocument10 pagesPlastic Industries: An OverviewKubra ĖdrisNo ratings yet
- StyreneDocument35 pagesStyreneDavid Wong67% (3)
- M.F PolymerizationDocument14 pagesM.F PolymerizationM. MoizNo ratings yet
- 3 s2.0 B0080431526004939 MainDocument6 pages3 s2.0 B0080431526004939 MainMohammed JamaliNo ratings yet
- Pva Emulsion PolymerisationDocument15 pagesPva Emulsion Polymerisationrajesh kothariNo ratings yet
- 2 Methods of PolymerizationDocument13 pages2 Methods of Polymerizationbt21102047 Vishwajeet YadavNo ratings yet
- Module 1Document19 pagesModule 1ABIGAIL OLAJUMOKE JOSEPHNo ratings yet
- Polyurethane Chemistry1Document7 pagesPolyurethane Chemistry1Ahmed AwadNo ratings yet
- M.F PolymerizationDocument14 pagesM.F Polymerizationsaqib sulmanNo ratings yet
- Acrylics: PolymerisationDocument3 pagesAcrylics: PolymerisationSdkmega Hh100% (1)
- Experiment 1 Bulk PolymerizationDocument7 pagesExperiment 1 Bulk Polymerizationsoran najebNo ratings yet
- Aa PDFDocument7 pagesAa PDFnguyen duyNo ratings yet
- Suspension Polymerization of Methyl Methacrylate PDFDocument3 pagesSuspension Polymerization of Methyl Methacrylate PDFWalid AdnanNo ratings yet
- Chapter IIaDocument8 pagesChapter IIaSyed Naveed Abbas NaqviNo ratings yet
- Article: Separation of The Glycerol-Biodiesel Phases in An Ethyl Transesterification Synthetic Route Using WaterDocument6 pagesArticle: Separation of The Glycerol-Biodiesel Phases in An Ethyl Transesterification Synthetic Route Using WaterMáximo Décimo MeridioNo ratings yet
- 3 Rekayasa Reaksi PolimerisasiDocument86 pages3 Rekayasa Reaksi Polimerisasirudy_423522658No ratings yet
- Polymer Class 2Document17 pagesPolymer Class 2Ñojib Ëasar ProttoyNo ratings yet
- Chapter 2 Process and Material SelectionDocument12 pagesChapter 2 Process and Material SelectionAli AhsanNo ratings yet
- Polymerization ProcessDocument38 pagesPolymerization ProcessAlina Aziz100% (1)
- Bioethanol Manufacturing From Lignocellulosic BiomassDocument7 pagesBioethanol Manufacturing From Lignocellulosic BiomassRifqiNo ratings yet
- Synthesis of Methylamine - OrgSynDocument5 pagesSynthesis of Methylamine - OrgSynzodd01No ratings yet
- Chem3020: Polymer Chemistry: Polymerization TechniquesDocument13 pagesChem3020: Polymer Chemistry: Polymerization TechniquesLuan GabrielNo ratings yet
- Silicon Wax Emulsion For Polyurethane Water Release AgentDocument3 pagesSilicon Wax Emulsion For Polyurethane Water Release Agentmyosi laboratoriumNo ratings yet
- Polymer Class 3Document9 pagesPolymer Class 3Ñojib Ëasar ProttoyNo ratings yet
- Obtainment of Polystyrene On An Industrial Level: Reaction MechanismDocument2 pagesObtainment of Polystyrene On An Industrial Level: Reaction MechanismYanin CyrusNo ratings yet
- Introduction of Pet: Solid State Polymerization (SSP)Document3 pagesIntroduction of Pet: Solid State Polymerization (SSP)Furqan SaeedNo ratings yet
- Bio Production Sdn. BHD.: Biodiesel Production by TransesterificationDocument47 pagesBio Production Sdn. BHD.: Biodiesel Production by Transesterificationhakim amirNo ratings yet
- Chapter 3 Polymerization TechDocument38 pagesChapter 3 Polymerization Techfyza8790No ratings yet
- Experiment 1: Emulsion Polymerization of StyreneDocument11 pagesExperiment 1: Emulsion Polymerization of StyreneNurshahanimNo ratings yet
- DEIPA - CN102924301A - Preparation Method of N, N-Bis (2-Hydroxuethyl) IsopropanolamineDocument5 pagesDEIPA - CN102924301A - Preparation Method of N, N-Bis (2-Hydroxuethyl) IsopropanolamineListya Eka AnggrainiNo ratings yet
- Polystyrene and CopolymersDocument7 pagesPolystyrene and Copolymerskeerthi srijithNo ratings yet
- Udex PlantDocument4 pagesUdex PlantMiko Yee Teng100% (1)
- 3 Polimerisasi HomogenDocument38 pages3 Polimerisasi Homogenutami10No ratings yet
- BTPDocument15 pagesBTPPARTH NAGARNo ratings yet
- Process Analytics in PE PlantsDocument9 pagesProcess Analytics in PE PlantsdavidNo ratings yet
- en Polyolefins AppDocument12 pagesen Polyolefins AppJoshiMBNo ratings yet
- Katch UmbelliferoneffrDocument9 pagesKatch Umbelliferoneffrapi-456902531No ratings yet
- Polymer: Neslihan Alemdar, A. Tuncer Erciyes, Niyazi BicakDocument7 pagesPolymer: Neslihan Alemdar, A. Tuncer Erciyes, Niyazi Bicaklivius12No ratings yet
- ICH404-Lecture Note 1Document2 pagesICH404-Lecture Note 1Lukman Bola Abdulra'ufNo ratings yet
- PRPC CH 7 - Part 2Document15 pagesPRPC CH 7 - Part 2lakshya chovatiaNo ratings yet
- Separation Processes-20150725-155901297Document21 pagesSeparation Processes-20150725-155901297Lohith LoliNo ratings yet
- Lab 3 HysysDocument8 pagesLab 3 HysysZulhafizi Kamaruddin100% (2)
- Experiment 2 Solution PolymerizationDocument36 pagesExperiment 2 Solution PolymerizationBeyar Shewket0% (1)
- Sustainable synthesis of ciclopentene derivatives through multicomponent reactions in continuous flow regimeFrom EverandSustainable synthesis of ciclopentene derivatives through multicomponent reactions in continuous flow regimeNo ratings yet
- TQM PlanningDocument113 pagesTQM PlanningAnish KumarNo ratings yet
- APM 2017 Poster 1Document1 pageAPM 2017 Poster 1Anish KumarNo ratings yet
- Polymer Journal ListDocument4 pagesPolymer Journal ListAnish KumarNo ratings yet
- Literature ReviewDocument6 pagesLiterature ReviewAnish KumarNo ratings yet
- MatStudio Castep - 2Document63 pagesMatStudio Castep - 2Anish Kumar100% (1)
- Thermoplastic Carbonate Groups ToughDocument3 pagesThermoplastic Carbonate Groups ToughAnish KumarNo ratings yet
- Refractive Index of PolymersDocument6 pagesRefractive Index of PolymersAnish KumarNo ratings yet
- MatStudio CASTEP GuideDocument219 pagesMatStudio CASTEP GuideAnish KumarNo ratings yet
- APM Additive Manufacturing FinalDocument4 pagesAPM Additive Manufacturing FinalAnish KumarNo ratings yet
- Anna University Annexure IDocument2 pagesAnna University Annexure IAnish KumarNo ratings yet
- Thermoplastic Carbonate Groups ToughDocument3 pagesThermoplastic Carbonate Groups ToughAnish KumarNo ratings yet
- Anish KumarDocument2 pagesAnish KumarAnish KumarNo ratings yet
- Nanofiller Reinforced NanocompositeDocument247 pagesNanofiller Reinforced NanocompositeAnish KumarNo ratings yet