Professional Documents
Culture Documents
Answersbrownchap3 p111
Uploaded by
Parham ElahiOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Answersbrownchap3 p111
Uploaded by
Parham ElahiCopyright:
Available Formats
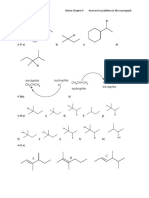
CHEM 221
Brown Chapter 3
Answers to problems in the coursepack
3.12 a) CHCl3 only one isomer; CH2Cl2 only one isomer; CHClBrF two isomers
b) CHCl3 only one isomer; CH2Cl2 two isomers cis and trans
CHFClBr three isomers
3.13 a) yes C-2 is a stereogenic center b) no, it is symmetrical c) yes C-3 is a stereogenic center d) yes C-2
is a stereogenic center
3.14 a) CH3CH2CH(CH3)CH2CH2CH3 this is the smallest acyclic alkane with a chiral center on C-3. There is a
smaller cycloalkane with a chiral center, trans-1,2-dimethylcyclobutane
b) CH3CH(OH)CH2CH3 c) CH3CH2CH(CH3)C=0 d) CH3CH2CH(CH3)C=0CH3 e) CH3CH2CH(CH3)CO2H f)
CH3CH2CH(CH3)CO2CH3
3.15 a)
e)
b)
f)
c)
g)
3.16 They are all identical stereorepresentations
d)
h)
CHEM 221
3.17 a) no stereocenter b)
Brown Chapter 3
Answers to problems in the coursepack
it has two stereoisomers c)
it has two stereoisomers d) no stereocenter e) no
stereocenter f)
two stereocenters
g) no stereocenter
3.19 a) OH (1), -CH2OH (2), -CH3 (3) and H (4) b) CH=CH2 (1), -CH2CO2H (2), CH2CH=CH2 (3) and CH3 (4)
c) NH3+(1), CO2- (2), CH3(3) and H (4) d) NH3+(1), -CH2SH (2), CHO (3) and CH3 (4)
3.20 (-)-carvone is R and (=)-carvone is S
CHEM 221
Brown Chapter 3
3.21a) S-2-butanol b)
Answers to problems in the coursepack
c)
The last one is more stable. It is similar in stability compared with the
conformer shown in b). Both have only one gauche interaction whereas the first one drawn in c) has two
gauche interactions.
3.22
3.24
are identical and they are called meso
3.23 S OH (1), -14CH2(2), -COOH (3) and CH2 (4)
These two stereorepresentations
CHEM 221
Brown Chapter 3
Answers to problems in the coursepack
These two stereorepresentations are
enantiomers one of another.
3.25 a)
b)
c)
d)
4 stereoisomers possible
4 stereoisomers possible
4 stereoisomers possible
2 stereoisomers possible
CHEM 221
e)
f)
g)
h)
Brown Chapter 3
Answers to problems in the coursepack
8 stereoisomers possible
2 stereoisomers possible
4 stereoisomers possible
4 stereoisomers possible
CHEM 221
Brown Chapter 3
3.26
stereoisomers, but some are geometrically impossible!
Answers to problems in the coursepack
Theoretically, 64
3.27
3.29 a) enantiomers b) diastereoisomers c) diastereoisomers
3.30 a), c), d), f) and h) are meso compounds because they have an internal plane of reflection.
You might also like
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Lecture 23 - Integration of MetabolismDocument32 pagesLecture 23 - Integration of MetabolismParham ElahiNo ratings yet
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- 3-221-2016F Hybridization and ConformationDocument110 pages3-221-2016F Hybridization and ConformationParham ElahiNo ratings yet
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (399)
- Lecture 3 - Protein StructureDocument59 pagesLecture 3 - Protein StructureParham ElahiNo ratings yet
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- 27 Clicker QuestionsDocument9 pages27 Clicker QuestionsParham ElahiNo ratings yet
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- Chap 22Document42 pagesChap 22Parham ElahiNo ratings yet
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- 28 Clicker QuestionsDocument8 pages28 Clicker QuestionsParham ElahiNo ratings yet
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Chem 221 Bruice Chapter 4 Answers To Problems in The CoursepackDocument7 pagesChem 221 Bruice Chapter 4 Answers To Problems in The CoursepackParham ElahiNo ratings yet
- Chap 40Document60 pagesChap 40Parham ElahiNo ratings yet
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (344)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (73)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (120)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)