Professional Documents
Culture Documents
Ichtiosis
Uploaded by
Anggi CalapiOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Ichtiosis
Uploaded by
Anggi CalapiCopyright:
Available Formats
109
CASE REPORT
▲
Progressive symmetrical erythrokeratodermia - Case report *
Eritroqueratodermia simétrica progressiva - Relato de caso
Bianca de Mello Guaraldi1 Thaís Jerez Jaime1
Rafael de Mello Guaraldi2 Daniel Fernandes Melo3
Osvania Maris Nogueira4 Nilton Rodrigues5
Resumo: Progressive symmetrical erythrokeratodermia is a rare autosomal dominant genodermatosis with
variable penetrance described by Darier in 1911. It is characterized by erythematous and keratotic plaques,
sharply defined and symmetrically distributed along the extremities, buttocks and, more rarely, on the face.
We report a case of a 55-year-old patient with lesions on the dorsum of the hands, interphalangeal pads,
wrists, groin and back feet. This case demonstrates a rare and late diagnosis, clinical profusion and presen-
ce of familiar involvement.
Keywords: Erythrokeratodermia variabilis; Keratodermia, palmoplantar; Keratosis
Abstract: Eritroqueratodermia simétrica progressiva é uma genodermatose rara de herança autossômica
dominante, com penetrância variável, descrita por Darier em 1911. É caracterizada por placas eritemato-
sas e queratósicas, bem delimitadas, simetricamente distribuídas ao longo das extremidades, nádegas e
mais raramente face. Os autores relatam o caso de uma paciente de 55 anos apresentando lesões no dorso
das mãos, coxins interfalangeanos, punhos, região inguinal e dorso dos pés. O caso demonstra raridade,
diagnóstico tardio, exuberância clínica e acometimento familial.
Palavras-chave: Ceratodermia Palmar e Plantar; Ceratose; Eritroceratodermia variável
INTRODUCTION
Erythrokeratodermias are classified as genoder- sis, moderate acanthosis with a prominent granular
matoses that share keratinization disorders. They are layer. Transitory well outlined erythematous plaques
represented by erythrokeratodermia variabilis (EKV) with a geographical appearance, are present in EKV
and progressive symmetric erythrokeratodermia and may affect any cutaneous surface.1,2,3 It is usually
(PSEK).1,2 An autosomal dominant inheritance is usual- seen in childhood, and the erythema tends to fade
ly seen in both, although there are reports of sporadic with time. Hyperkeratosis is clinically observed
cases as well as autosomal recessive inheritance in through the presence of thickened plaques and it is
PSEK. They are generally distinguished by their clini- usually more stable than the erythema. The variability
cal aspect, since the histopathology findings are in number, size, shape and duration of the erythema-
almost the same, showing orthokeratotic hyperkerato- tous plaques are defined by the given name of the ill-
Received on 16.12.2011.
Approved by the Advisory Board and accepted for publication on 11.02.2012.
* Study carried out at the Naval Hospital Marcílio Dias (HNMD) – Rio de Janeiro (RJ), Brazil.
Financial Support: none
Conflict of Interests: none
1
Graduate course in dermatology - Naval Hospital Marcílio Dias (Hospital Naval Marcílio Dias - HNMD) – Rio de Janeiro (RJ), Brazil.
2
MD, Fundação Técnico Educacional Souza Marques (FTESM) – Rio de Janeiro (RJ), Brazil.
3
MD, Federal University of the State of Rio de Janeiro (Universidade Federal do Estado do Rio de Janeiro - UNIRIO). Graduate degree in Dermatology from the
Federal University of the State of Rio de Janeiro (Universidade Federal do Estado do Rio de Janeiro - UNIRIO). Preceptor in Graduate Dermatology program
at the Naval Hospital Marcílio Dias (Hospital Naval Marcílio Dias - HNMD) – Rio de Janeiro (RJ), Brazil.
4
Biomedical scientist and Biologist, Federal University of the State of Rio de Janeiro (Universidade Federal do Estado do Rio de Janeiro - UNIRIO). - Preceptor
at the Graduate Course of Naval Hospital Marcílio Dias (Hospital Naval Marcílio Dias - HNMD). Collaborator at the Graduate course of the Teaching Hospital
Gaffrée e Guinle – Federal University of the State of Rio de Janeiro (Universidade Federal do Estado do Rio de Janeiro - HUGG-UNIRIO). Collaborator at the
Pediatric Dermatology Service of the Naval Hospital Marcílio Dias – Federal University of the State of Rio de Janeiro ((Hospital Universitário Gaffrée e Guinle
- Universidade Federal do Estado do Rio de Janeiro - HUGG-UNIRIO) – Rio de Janeiro (RJ), Brazil.
5
Military Head of the Dermatology Clinic of the Naval Hospital Marcílio Dias (HNMD) – Rio de Janeiro (RJ), Brazil.
©2013 by Anais Brasileiros de Dermatologia
An Bras Dermatol. 2013;88(1):109-12.
110 Guaraldi BM, Jaime TJ, Guaraldi RM, Melo DF, Nogueira OM, Rodrigues N
ness.1,3 Lesions may be modified depending on tempe-
rature, emotional stress and mechanical pressure.
They tend to worsen during pregnancy and around
30% of patients may suffer the onset of their symp-
toms at birth, which does not occur in PSEK cases. In
PSEK, keratotic plaques are on an erythematous base
and remain in a fixed area since the early stages of the
disease. Usually there is symmetric distribution
around joints and it attacks more specific skin surfa-
ces, such as extremities, buttocks and occasionally
face and torso. The lesions slowly become progressi-
ve in number and size, which increases throughout
the years. Both diseases share the fact that they stabi-
lize after puberty. The chosen treatment for erythroke-
ratodermias is oral retinoids. The limiting factor for
their use is the side effects, especially in children. FIGURE 1: Clinical aspect on the dorsum of the hands. Fixed and
There are reports of clinical improvement, however, well-defined symmetrically distributed erythematous-scaly plaques
recurrence is expected when the treatment is inter-
rupted. Formulas containing coaltar, salicylic acid,
corticosteroids and topic retinoids can be used, but
variation in results may occur. An asymptomatic clini-
cal case of classic PSEK is described below with fami-
ly involvement.
CASE REPORT
Female patient, 55 years old, phototype III,
referred the onset of skin lesions from the age of 5,
with progressive growth until the age of 18. She
reports that similar lesions in the same locations also
occurred on three of her siblings during their child-
hood. Their parents were not consanguineous.
Dermatological exams evidenced fixed, finely scaly,
symmetric erythematous keratotic plaques on the dor-
sum of the hands, interphalangeal pads, wrists, FIGURE 2: Clinical aspect of the elbows. Fixed and well-defined
elbows, groin and feet (Figures 1, 2, 3, 4). The pla- symmetrically distributed erythematous-scaly plaques
ques were thicker on the elbows and there was a well-
defined, brownish-colored hyperpigmentation halo
on the inguinal region. A biopsy of the dorsum of the
right foot revealed discrete papillomatosis and acant-
hosis, orthokeratotic hyperkeratosis in a basket-weave
pattern and maintenance of the granular layer, thus
eliminating the main hypothesis of psoriasis (Figure
5). For treatment, topical 0.025% tretinoin associated
with 20% urea and 3% salicylic acid was applied, as
well as moisturizing lotion in the morning. After 4
months’ treatment only mild clinical improvement
was noticed and 20mg/day acitretin was introduced,
with excellent response.
FIGURE 3:
DISCUSSION Clinical aspect
on dorsum of
Progressive symmetrical erythrokeratodermia, the feet. Fixed
also known as Gottron syndrome or Darier-Grotton and well-defi-
erythrokeratodermia, was described by Darier in ned symmetri-
cally distributed
1911. In 1923, Grotton was the one who defined the
erythematous-
name for this particular illness as it is known today.1,2 scaly plaques
An Bras Dermatol. 2013;88(1):109-12.
Progressive symmetrical erythrokeratodermia - Case report 111
FIGURE 4: Clinical aspect of the inguinal area. Erythematous FIGURE 5: Histopathology stained by Hematoxilin Eosin. HE, 100x.
plaques, with a brownish-colored halo delimiting the An orthokeratotic hyperkeratosis in a basket-weave pattern.
affected region Presence of moderate acanthosis and focus of thickened
granular layer
Darier described it as a rare keratinization disorder, the cohesion of the stratum corneum cells.6,8
characterized by non-migratory keratotic plaques on Transmission in PSEK is hereditary and it results in an
an erythematous base, appearing insidiously and autosomal dominant pattern, with sporadic cases
slowly on circinate areas, surrounded by a delicate occurring in up to 50% of the total diagnosed. It
hyperpigmented projection, with symmetric distribu- affects both sexes equally during the first months or
tion on knees, elbows, hands and feet. There may years of their lives.9 Genetic penetration, however, can
occur palmoplantar hyperkeratosis in approximately be incomplete which generates a variable clinical
50% of patients. The pathogenesis of erythrokerato- expressivity. Spontaneous remission does not usually
dermia requires further studies.4 Analysis has shown occur, although cases have been described in the lite-
that mitotic activity increased on the affected skin of rature. Diagnosis is established on clinical bases and
patients with PSEK and was normal in EKV patients. histopathology is most important to exclude the main
Physiopathology of both conditions reveal different differential diagnosis, psoriasis. The therapy will be
aspects; molecular genetic alteration in PSEK still directed according to clinical manifestations. In clini-
remains unknown. Reports show that a mutation cally moderated non-malignant cases, the use of reti-
occurs in the loricrin protein, which is the major cons- noids, keratolytics and even steroids would be an
tituent of the cornified cell, with gene-coding region option. In more extensive cutaneous involvement,
located on 1q21.3.5,6,7 It is believed that the mutant systemic therapy is indicated, with retinoids being the
loricrin accumulates in the nucleus, interfering in the main choice. The most commonly used drugs are aci-
keratinocyte apoptosis process, leading to a pathologi- tretin and etretinate in doses varying between 0.5 to
cally thickened stratum corneum; differently from the 1.0mg/kg. Phototherapy has shown good results. In
EKV, which shows a genetic mutation recently located the case reported there was favorable clinical respon-
in the lower arm of chromosome 1, responsible for the se to systemic therapy with acitretin, but without total
expression of connexin 31, generating anomalies in remission of the lesions. ❑
An Bras Dermatol. 2013;88(1):109-12.
112 Guaraldi BM, Jaime TJ, Guaraldi RM, Melo DF, Nogueira OM, Rodrigues N
REFERENCES
1. Nico MMS, Neto CF, Oliveira ZNP. Eritroqueratodermia simétrica progressiva. An 8. Ishida-Yamamoto A, Kato H, Kiyama H, Armstrong DK, Munro CS, Eady RA, et al.
Bras Dermatol. 1995;70:551-3. Mutant loricrin is not crosslinked into the cornified cell envelope but is transloca-
2. Zanini M, Bertizo D, Corrêa e Silva K, Paschoal LHC, Landaman G, Freitas E. ted into the nucleus in loricrin keratoderma. J Invest Dermatol. 2000;115:1088-94.
Eritroqueratodermia simétrica progressiva: relato de um caso esporádico e de sur- 9. Yan HB, Zhang J, Liang W, Zhang HY, Liu JY. Progressive symmetric erythrokera-
gimento tardio. Med Cutan Iber Lat Am. 2003;31:192-4. toderma: report of a Chinese family . Indian J Dermatol Venerol. 2011;77:597-600.
3. Nazarro V, Blanchet-Bardon C. Progressive symmetric erythrokeratodermia.
Histological and ultrastructural study of patient before and after treatment with
etretinate. Arch Dermatol 1986;122:434-40.
4. Rodriguez-Pichardo A, Garcia-Bravo B, Sanchez-Pedreno P,Camacho-Martinez F. MAILING ADDRESS:
Progressive symmetric erythrokeratodermia. J Am Acad Dermatol 1998;19:129-30.
5. Suga Y, Jarnik M, Attar OS, Longley MA, Bundman D, Steven AC, et al. Transgenic Bianca de Mello Guaraldi
mice expressing a mutant form of loricrin reveal the molecular basis of the skin Rua Cezar Zama Nº 185
diseases, Vohwinkel syndrome and progressive symmetric erythrokeratoderma. J Lins de Vasconcelos
Cell Biol. 200;151:401-12.
6. Akman A, Masse M, Mihci E, Richard G, Christiano AM, Balle BJ, et al. Progressive 20725-090 Rio de Janeiro, RJ.
symmetrical erythrokeratoderma: report of a Turkish family and evaluation for lori- Tel.: 2599-5599
crin and conexin gene mutations. Clin Exp Dermatol. 2008;33: 582-4. E-mail: biaguaraldi@hotmail.com
7. Chu DH, Arroyo MP. Progressive and symmetric erythrokeratoderma. Dermatol
Online J. 2003;9:21.
How to cite this article: Guaraldi BM, Jaime TJ, Guaraldi RM, Melo DF, Nogueira OM, Rodrigues N. Progressive
symmetrical erythrokeratoderma: a case report. An Bras Dermatol. 2013;88(1):109-12.
An Bras Dermatol. 2013;88(1):109-12.
You might also like
- Estudo aberto prospectivo de 30 pacientes com onicomicose por Scytalidium spp. tratados com terbinafina oral, ciclopiroxolamina esmalte e onicoabrasãoFrom EverandEstudo aberto prospectivo de 30 pacientes com onicomicose por Scytalidium spp. tratados com terbinafina oral, ciclopiroxolamina esmalte e onicoabrasãoNo ratings yet
- Sindrome de Reiter - Relato de CasoDocument8 pagesSindrome de Reiter - Relato de CasoDavi LeocadioNo ratings yet
- DownloadDocument7 pagesDownloadIsablla LopesNo ratings yet
- Hiperqueratose Palmo-Plantar Epidemolítica (Vöner)Document11 pagesHiperqueratose Palmo-Plantar Epidemolítica (Vöner)vanderson1980No ratings yet
- 1627-8463-1-PB - Hemangioma RubiDocument3 pages1627-8463-1-PB - Hemangioma RubicbetterNo ratings yet
- DescargaDocument3 pagesDescargaJAVIER FAJARDONo ratings yet
- The Genodermatoses: An Overview - UpToDateDocument85 pagesThe Genodermatoses: An Overview - UpToDatedrajaninehorsthNo ratings yet
- Caso Clínico - Larva MigransDocument4 pagesCaso Clínico - Larva MigransIvanildo VieiraNo ratings yet
- Sindrome de ProteusDocument8 pagesSindrome de ProteusAdriano SilvaNo ratings yet
- Dermatofibroma AneurismáticoDocument3 pagesDermatofibroma Aneurismáticogust96No ratings yet
- Examedapeleefneros 220625 180821 1656461142Document24 pagesExamedapeleefneros 220625 180821 1656461142Stephanny VictóriaNo ratings yet
- PsoríaseDocument6 pagesPsoríaseNatália Stephan PadilhaNo ratings yet
- v10 Lesoes em Dedos Na Pratica DermatologicaDocument8 pagesv10 Lesoes em Dedos Na Pratica DermatologicaEnfermaria - ConfhicNo ratings yet
- Dermatites: Diagnóstico E Terapêutica: Dermatitis: Diagnosis and TherapyDocument5 pagesDermatites: Diagnóstico E Terapêutica: Dermatitis: Diagnosis and TherapyOrnede ParielaNo ratings yet
- V 86 N 5 A 23Document4 pagesV 86 N 5 A 23ruggeri33No ratings yet
- Vasculites-Anais de DermatologiaDocument15 pagesVasculites-Anais de DermatologiaAlessa Arruda PintoNo ratings yet
- Juares Et Al - Síndrome de Rendu-Osler-WeberDocument6 pagesJuares Et Al - Síndrome de Rendu-Osler-WeberRodolfo VenturaNo ratings yet
- VIEIRA 2012 - Sindrome de GorlinDocument5 pagesVIEIRA 2012 - Sindrome de GorlinJohnatanNo ratings yet
- Cicatrizes Hipertroficas e QueloidesDocument9 pagesCicatrizes Hipertroficas e QueloidesAna Babel SánchezNo ratings yet
- 374-Texto Do Artigo-1786-1-10-20061031Document11 pages374-Texto Do Artigo-1786-1-10-20061031Jota CarlosNo ratings yet
- Dermatite SeborreicaDocument4 pagesDermatite SeborreicaInRolsy SlveiarNo ratings yet
- Lipomatose Simétrica e Pelagra Associadas Ao AlcoolismoDocument4 pagesLipomatose Simétrica e Pelagra Associadas Ao AlcoolismoKatia BarcellosNo ratings yet
- Será Fungo?: DossierDocument13 pagesSerá Fungo?: DossierAlberto Tolotti LeiteNo ratings yet
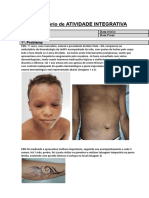
- Problema 3Document12 pagesProblema 3ERICA LUANA SILVA VELOSONo ratings yet
- Afecções Podais Cirúrgicas em Grandes Animais - AulaDocument19 pagesAfecções Podais Cirúrgicas em Grandes Animais - AulaThais Manzoni100% (1)
- Dermatologia Na ABDocument44 pagesDermatologia Na ABAllyson SilvaNo ratings yet
- 32 Artigo3Document4 pages32 Artigo3Japah MeloNo ratings yet
- Semiologia DermatológicaDocument18 pagesSemiologia DermatológicaPaulo Albuquerque100% (1)
- HEMANGIOMA CAVERNOSO Grupo 4Document4 pagesHEMANGIOMA CAVERNOSO Grupo 4Julia RamosNo ratings yet
- Aula 08 - Doença Eritemato-Escamosas (Revisada)Document9 pagesAula 08 - Doença Eritemato-Escamosas (Revisada)leticyacasteloNo ratings yet
- v95 Atualizacao em Vasculites Visao Geral e Aspectos Dermatologicos Relevantes para o Diagnostico Clinico e Histopatologico - Parte IIDocument15 pagesv95 Atualizacao em Vasculites Visao Geral e Aspectos Dermatologicos Relevantes para o Diagnostico Clinico e Histopatologico - Parte IIPaulo Victor Amorim MarquesNo ratings yet
- Semiologia 10 - Dermatologia - Semiologia Dermatológica PDFDocument18 pagesSemiologia 10 - Dermatologia - Semiologia Dermatológica PDFMedicinaNo ratings yet
- Tegumento Comum e Seus AnexosDocument13 pagesTegumento Comum e Seus Anexosbento uaneNo ratings yet
- Seminario de Dermato FuncionalDocument34 pagesSeminario de Dermato FuncionalArodo da SilvaNo ratings yet
- Resumo Super Material Semiologia Dermatologica e Lesoes Elementares 5070844 1701318177Document21 pagesResumo Super Material Semiologia Dermatologica e Lesoes Elementares 5070844 1701318177CAMILO SANTIAGO ALVES DA SILVANo ratings yet
- 601-Article Text-1118-1-10-20161015Document7 pages601-Article Text-1118-1-10-20161015Lucas ModestoNo ratings yet
- v5 OnicomatricomaDocument5 pagesv5 OnicomatricomaBruno Guedes Rec50% (2)
- Adenoma Pleomorfico em Palato MoleDocument4 pagesAdenoma Pleomorfico em Palato MoleEdgar Ferreira SantosNo ratings yet
- Prurido No IdosoDocument7 pagesPrurido No IdosoRiviane LucenaNo ratings yet
- Complexo Granuloma Eosinofílico em Felinos DomésticosDocument6 pagesComplexo Granuloma Eosinofílico em Felinos DomésticosLarissa AlonsoNo ratings yet
- Dermatite Herpetiforme Caso ClinicoDocument5 pagesDermatite Herpetiforme Caso ClinicojanafaNo ratings yet
- Classificação Das QueimadurasDocument8 pagesClassificação Das QueimadurasAvanúziaNo ratings yet
- v10 Foliculite Supurativa Cronica de Couro Cabeludo Desafio TerapeuticoDocument4 pagesv10 Foliculite Supurativa Cronica de Couro Cabeludo Desafio TerapeuticoJúlia Oliveira de CarvalhoNo ratings yet
- Aula MalasseziaDocument90 pagesAula MalasseziaTatiane RodriguesNo ratings yet
- 01 Semiologia Da PeleDocument20 pages01 Semiologia Da PeleRita Félix TavaresNo ratings yet
- HemangiomaDocument32 pagesHemangiomalivroslaanatoNo ratings yet
- ICTIOSESDocument8 pagesICTIOSESJosé Augusto CeronNo ratings yet
- Poster Esporitricose 1Document1 pagePoster Esporitricose 1Ticiana CavalcanteNo ratings yet
- Artigo - 2023.2Document4 pagesArtigo - 2023.2savio.c.salesNo ratings yet
- Tutoria 2 - Caso 1Document25 pagesTutoria 2 - Caso 1Rhafaella Rocha Rosa de LimaNo ratings yet
- Eritrocitose em GatoDocument28 pagesEritrocitose em GatoFabiana SoaresNo ratings yet
- Esporotricose TraduzidoDocument9 pagesEsporotricose TraduzidolucasNo ratings yet
- Pitiriase Versicolor 2 PDFDocument2 pagesPitiriase Versicolor 2 PDFLucas Da SilvaNo ratings yet
- Cirurgia Dos Cistos e TumoresDocument10 pagesCirurgia Dos Cistos e Tumoreswil_morett76No ratings yet
- 330-Article Text-604-1-10-20150413Document4 pages330-Article Text-604-1-10-20150413Maria Jorgia PleuriesNo ratings yet
- Revisão Ted 2011 - Casos Relevantes para EstudoDocument142 pagesRevisão Ted 2011 - Casos Relevantes para EstudoAndré Cartell100% (1)
- Glosário DermatopatologiaDocument18 pagesGlosário DermatopatologiaTiago Resende TellesNo ratings yet
- DERMATOFITOSEDocument2 pagesDERMATOFITOSEVinicius Gonçalves de SouzaNo ratings yet
- Dysplastic Nevus Atypical NevusDocument10 pagesDysplastic Nevus Atypical NevusHeather Marie MathisNo ratings yet
- Relação de Prestadores Montes ClarosDocument5 pagesRelação de Prestadores Montes ClarosCris OliveiraNo ratings yet
- Quadro Comparativo. VitaminasDocument5 pagesQuadro Comparativo. VitaminasLion DoslionNo ratings yet
- ABV Aula 3 - 2 - Vias de Administração e EliminaçãoDocument3 pagesABV Aula 3 - 2 - Vias de Administração e EliminaçãoLidianeHelenaNo ratings yet
- Pontos MD Codes - Pesquisa GoogleDocument1 pagePontos MD Codes - Pesquisa GoogleLorena Silveira CardosoNo ratings yet
- Prova Emergência Suseme 2014 - RJDocument5 pagesProva Emergência Suseme 2014 - RJRafael Teixeira MorseNo ratings yet
- Doses PediatriaDocument4 pagesDoses PediatriaRicardo RosyNo ratings yet
- Avaliação Nutricional Gestante PDFDocument34 pagesAvaliação Nutricional Gestante PDFAlcione Frankline Oliveira AlvesNo ratings yet
- Patologias Da Coluna VertebralDocument34 pagesPatologias Da Coluna VertebralCesar Odilon PereiraNo ratings yet
- Escore de AdmissãoDocument1 pageEscore de AdmissãoFrederico RodriguesNo ratings yet
- Diagnósticos e Prescrições de Enfermagem Nas Síndromes Gripais e SRAGDocument23 pagesDiagnósticos e Prescrições de Enfermagem Nas Síndromes Gripais e SRAGInara PaixaoNo ratings yet
- Imagenologia o Corpo Humano Sob Outro Ponto de Vista CRBM-5 - CONSELHO REGIONAL DE BIOMEDICINA - 5 REGIÃO - Rio Grande Do SuDocument1 pageImagenologia o Corpo Humano Sob Outro Ponto de Vista CRBM-5 - CONSELHO REGIONAL DE BIOMEDICINA - 5 REGIÃO - Rio Grande Do SuAndré luisNo ratings yet
- Tratamento Fisioterapico Asma Pmca MeiryDocument41 pagesTratamento Fisioterapico Asma Pmca MeiryEmmanuel DouradoNo ratings yet
- 11 - Paralisia Facial - 2Document15 pages11 - Paralisia Facial - 2brunosmelo100% (1)
- Refluxo Gastroesofágico Diagnóstico e TratamentoDocument9 pagesRefluxo Gastroesofágico Diagnóstico e TratamentoRoberta AndradeNo ratings yet
- DFL Cartilha de Anestesia LocalDocument12 pagesDFL Cartilha de Anestesia LocalElcijane AlvesNo ratings yet
- Portaria SES-PE #660 de 17 de Outubro de 2022Document4 pagesPortaria SES-PE #660 de 17 de Outubro de 2022Paula JácomeNo ratings yet
- Tpmed2017 GabaritoDocument31 pagesTpmed2017 GabaritoGuilhermeNo ratings yet
- PROTOCOLO MUNICIPIAL TEA BelémDocument90 pagesPROTOCOLO MUNICIPIAL TEA BelémBrenda Nascismento100% (1)
- Relatório Aula Prática CitopatologiaDocument11 pagesRelatório Aula Prática CitopatologiaMariana GonçalvesNo ratings yet
- Preparo Colonoscopia - ManhãDocument3 pagesPreparo Colonoscopia - ManhãRafaela MendesNo ratings yet
- M3 - Aula - Crises Convulsivas PDFDocument41 pagesM3 - Aula - Crises Convulsivas PDFBabi S.No ratings yet
- Etiologia Da Doença PeriodontalDocument61 pagesEtiologia Da Doença PeriodontalIkaro MenezesNo ratings yet
- 9 Dicas de Dietas para Hidradenite SupurativaDocument9 pages9 Dicas de Dietas para Hidradenite SupurativaTookmed Doctor ́sNo ratings yet
- TCC OBESIDADE EM FELINOS - REVISÃO de Literatura .Document23 pagesTCC OBESIDADE EM FELINOS - REVISÃO de Literatura .vetdomeupetNo ratings yet
- Apostila 14Document15 pagesApostila 14Karla BarataNo ratings yet
- Slide MagnetoterapiaDocument43 pagesSlide MagnetoterapiaGlaydesNo ratings yet
- Nota Informativa COVID-19 009 - 2020 - 10112023Document1 pageNota Informativa COVID-19 009 - 2020 - 10112023luizcmarquesNo ratings yet
- Manual - Tratamento - Malária - PNCM (v29.07.21) - CompactadoDocument47 pagesManual - Tratamento - Malária - PNCM (v29.07.21) - CompactadoEufrásio António100% (1)
- Tratamento Da Gonorreia PDFDocument23 pagesTratamento Da Gonorreia PDFAdalberto BatistaNo ratings yet
- Slide - Avaliação de AudioDocument8 pagesSlide - Avaliação de AudioAna Luiza PereiraNo ratings yet
- Desbloqueie o poder da sua mente: Programe o seu subconsciente para se libertar das dores e inseguranças e transforme a sua vidaFrom EverandDesbloqueie o poder da sua mente: Programe o seu subconsciente para se libertar das dores e inseguranças e transforme a sua vidaRating: 4.5 out of 5 stars4.5/5 (227)
- Corpo como Palavra: Uma visão bíblica sobre saúde integralFrom EverandCorpo como Palavra: Uma visão bíblica sobre saúde integralRating: 5 out of 5 stars5/5 (1)
- Deu a louca nos signos: Um guia divertido do zodíaco por AstroloucamenteFrom EverandDeu a louca nos signos: Um guia divertido do zodíaco por AstroloucamenteRating: 5 out of 5 stars5/5 (1)
- Treinamento cerebral: Compreendendo inteligência emocional, atenção e muito maisFrom EverandTreinamento cerebral: Compreendendo inteligência emocional, atenção e muito maisRating: 4.5 out of 5 stars4.5/5 (169)
- Descolonizando afetos: Experimentações sobre outras formas de amarFrom EverandDescolonizando afetos: Experimentações sobre outras formas de amarRating: 5 out of 5 stars5/5 (6)
- Fogo no parquinho: Namoro à luz da Palavra de DeusFrom EverandFogo no parquinho: Namoro à luz da Palavra de DeusRating: 5 out of 5 stars5/5 (9)
- 21 dias para curar sua vida: Amando a si mesmo trabalhando com o espelhoFrom Everand21 dias para curar sua vida: Amando a si mesmo trabalhando com o espelhoRating: 4.5 out of 5 stars4.5/5 (49)
- O fim da ansiedade: O segredo bíblico para livrar-se das preocupaçõesFrom EverandO fim da ansiedade: O segredo bíblico para livrar-se das preocupaçõesRating: 5 out of 5 stars5/5 (16)
- Herbologia Mágica: A cura pela Natureza com base na Fitoterapia & Botânica OcultaFrom EverandHerbologia Mágica: A cura pela Natureza com base na Fitoterapia & Botânica OcultaRating: 4.5 out of 5 stars4.5/5 (7)
- Expansão da Consciência: A Verdadeira História da Humanidade e a Transição PlanetáriaFrom EverandExpansão da Consciência: A Verdadeira História da Humanidade e a Transição PlanetáriaRating: 5 out of 5 stars5/5 (1)
- Crianças desafiadoras: Aprenda como identificar, tratar e contribuir de maneira positiva com crianças que têm Transtorno Opositivo-DesafiadorFrom EverandCrianças desafiadoras: Aprenda como identificar, tratar e contribuir de maneira positiva com crianças que têm Transtorno Opositivo-DesafiadorNo ratings yet
- Eu te amo, mas não te quero mais: Histórias de fins e recomeçosFrom EverandEu te amo, mas não te quero mais: Histórias de fins e recomeçosRating: 5 out of 5 stars5/5 (6)
- Magias, Feitiços, Rituais E Simpatias De São Cipriano Em PdfFrom EverandMagias, Feitiços, Rituais E Simpatias De São Cipriano Em PdfRating: 5 out of 5 stars5/5 (3)
- Almanaque do Pensamento 2024: Guia astrológico para 2024From EverandAlmanaque do Pensamento 2024: Guia astrológico para 2024Rating: 5 out of 5 stars5/5 (2)
- Encantamentos, Rezas E BenzedurasFrom EverandEncantamentos, Rezas E BenzedurasRating: 4.5 out of 5 stars4.5/5 (11)
- Nana Nenê: Como cuidar de seu bebê para que durma a noite toda de forma naturalFrom EverandNana Nenê: Como cuidar de seu bebê para que durma a noite toda de forma naturalRating: 4.5 out of 5 stars4.5/5 (5)
- O Verdadeiro Ho'oponopono: Como restaurar sua harmonia, limpar memórias e manifestar milagresFrom EverandO Verdadeiro Ho'oponopono: Como restaurar sua harmonia, limpar memórias e manifestar milagresRating: 5 out of 5 stars5/5 (5)
- Banhos, Defumações, Encantamentos, Feitiços, Magias, Oferendas, Rituais E Simpatias De Maria PadilhaFrom EverandBanhos, Defumações, Encantamentos, Feitiços, Magias, Oferendas, Rituais E Simpatias De Maria PadilhaRating: 5 out of 5 stars5/5 (1)
- Chakras: O guia definitivo para irradiar energia positiva, equilíbrio, chakras e despertar seu terceiro olhoFrom EverandChakras: O guia definitivo para irradiar energia positiva, equilíbrio, chakras e despertar seu terceiro olhoRating: 5 out of 5 stars5/5 (10)
- As 5 linguagens do amor - 3ª edição: Como expressar um compromisso de amor a seu cônjugeFrom EverandAs 5 linguagens do amor - 3ª edição: Como expressar um compromisso de amor a seu cônjugeRating: 5 out of 5 stars5/5 (220)