Professional Documents
Culture Documents
2 Ultraviolet-Visible Spectroscopy: 2.1 Revision
Uploaded by
زينبالجنابيOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
2 Ultraviolet-Visible Spectroscopy: 2.1 Revision
Uploaded by
زينبالجنابيCopyright:
Available Formats
2 Ultraviolet-Visible Spectroscopy
2.1 Revision
The following questions cover the important concepts that you should have understood in the first
year instrumentation subject.
1. What are the wavelength ranges for the ultraviolet and visible regions of the spectrum?
2. What molecular or structural features give rise to absorption of ultraviolet/visible (UV/VIS)
radiation in organic species? Give an example of an organic compound that would not absorb
UV/VIS radiation.
3. What molecular or structural features give rise to absorption of ultraviolet/visible (UV/VIS)
radiation in ionic species? Give an example of an ionic species that would not absorb UV/VIS
radiation.
4. What solvent and cell materials would be suitable for the following scan?
Analyte Region Solvent Cell
(a) copper sulfate visible
(b) copper sulfate UV/visible
(c) methylbenzene UV
(d) non-polar yellow dye visible
2. UV/VIS spectroscopy
2.2 Absorbing species
Organic compounds
As you are aware, double & triple bonds and non-bonded electrons on atoms such as N, O and the
halogens are the structural features that cause absorption in the UV/VIS region. Actually, this
statement isn’t strictly true!
In fact, all compounds absorb UV radiation, it’s just that the compounds without the multiple bonds
and non-bonded electrons absorb below the “dividing line” of 200 nm. Why that figure you might ask?
A purely practical one: measurements in the far UV (as < 200 nm is known) require a vacuum, since
the components of air absorb strongly. This makes for a less convenient, more complex device and
lots of interferences for quantitative analysis. So when we talk about UV spectra, we are actually
referring to near UV.
While we are on the topic of air absorbing UV, it raises a seeming contradiction to our general rule
about what absorbs in the (near) UV: after all, what are the structural features of nitrogen and oxygen
gases? Double (O=O) or triple (N≡N) bond and lots of non-bonded electrons (4 on each oxygen, 2 on
each nitrogen). So, really air should absorb well above 200 nm, but fortunately it doesn’t (and don’t
ask me why, because I don’t know).
For a compound to absorb strongly above 200 nm, it requires a number of multiple bonds in
conjugation (alternating single-multiple) and/or a number of non-bonded electron atoms.
EXAMPLE 2.1
The following compounds show the effect of the presence or absence of absorbing groups (number
given is the wavelength of maximum absorption).
< 200 210 256 282 312
CH3 Cl OH OH COOH
OH
OH
260 265 270 280 300
These are all obviously absorbing in the UV region only. For an organic compound to absorb in the
visible region (even if the peak isn’t past 400 nm), it will need numerous conjugated double bonds and
non-bonded electrons. Figure 2.1 shows the structure of methyl orange to illustrate this.
HO3S N N N(CH3)2
FIGURE 2.1 The structure of methyl orange (the yellow form in neutral solution)
Adv. Spectr/Chrom 2.2
2. UV/VIS spectroscopy
Inorganic compounds
Some simple metal ions absorb in the ultraviolet or visible region, because they have valence
electrons, eg Cu2+ and Ni2+, but usually what absorption there is, is not particularly strong, and
therefore, not useful for quantitative analysis.
EXERCISE 2.1
Why would weak absorption by a chemical species, eg Cu2+, make it not useful for
quantitative analysis?
Polyatomic ions, such as permanganate and dichromate, get their much stronger absorbance from a
combination of factors: multiple bonds and non-bonded electrons. In the majority of cases, complexes
of metal ions and ligands are needed for intense absorption, and the ligands are known as colour-
forming reagents.
2.3 Cells and solvents
The basic rule for the sample “container” – cell and solvent – is that it (they) should not absorb more
than 0.2 at wavelengths of interest.
Cells
The three standard cells for UV/VIS are, of course, quartz, glass and plastic. All three are suitable for
the visible region and into the UV down to 350 nm, but in practice, plastic is used because of its
cheapness, unless there is an organic solvent being used, in which case, glass becomes the choice.
Below 350 nm, quartz is the only choice.
Solvents
The wider range of solvents available makes for more flexibility, but the starting requirement
for a solvent is that it dissolves the species of interest! All the solvents in Table 2.1 are
colourless, so any of them are suitable for the visible region. In the UV, most organic
compounds absorb to some extent, so it is a matter ensuring that the cutoff wavelength (see
Table 2.1) is lower than the wavelength(s) of interest.
TABLE 2.1 Minimum wavelengths for common solvents (common names in brackets)
Solvent nm Solvent nm
water 205 ethanenitrile (acetonitrile) 210
cyclohexane 210 ethoxyethane (ethyl ether) 210
ethanol 210 hexane 220
methanol 210 dioxane 220
dichloromethane 235 trichloromethane (chloroform) 245
methylbenzene (toluene) 280 propanone (acetone) 330
Adv. Spectr/Chrom 2.3
2. UV/VIS spectroscopy
EXERCISE 2.2
Choose a suitable solvent for the following analyses (wavelengths of interest in brackets).
(a) dimethylbenzene (250-300 nm)
(b) sodium benzoate (250-320 nm)
(c) aspirin (280-320 nm)
Some chemical companies sell a range of solvents known as Spectrograde, which are
specifically designed for use in UV spectroscopy. They are not necessarily more pure than AR
grade, but they are guaranteed not to have absorbing impurities. For example, AR grade
hexane might be 99.9% pure, but the impurity could be benzene, whereas spectrograde hexane
might only be 99% pure, but the impurity is heptane.
2.4 Radiation sources
Two radiation sources are required to cover the entire UV/VIS range:
• a deuterium discharge lamp for the UV
• a tungsten filament globe for the visible
Deuterium gas, when excited by low-voltage electrical energy produces an almost perfectly continuous
spectrum in the UV region between 160 and 375 nm. At longer wavelengths, the lamp still produces
some intensity, but the continuous spectrum is mixed with narrow emission lines, which cause
problems with quantitative measurements.
Tungsten filament lamps are simply the familiar light bulb of domestic use. They work by being heated
to incandescence, the temperature at which the solid emits visible radiation. Above this point the
continuous radiation produced (known as blackbody radiation) has an intensity that is dependent on
the temperature, rather than the chemical composition of the heated material. A tungsten filament
at 3000K produces radiation over a range of 350-2500 nm, with the peak around 1000 nm. The
intensity in the visible region is not uniform, and the lamp is not efficient, since only 15% of the
intensity is in the visible region.
The output of the tungsten filament is dependent on the applied voltage, and, therefore, it is crucial
that the power supply contains a voltage regulator to ensure a constant value. The significant levels
of infrared radiation (heat) are often removed by a filter.
For a full spectrum covering both UV and visible regions, the instrument blocks off the source not
required by use of a filter until the changeover point at 350 nm. There is inevitably a difference in
intensity between the two sources at this wavelength, but design, gain adjustment and the background
scan mean that there isn’t a bump in the spectrum.
2.5 Monochromators
Both prisms (in glass or quartz) or gratings can be used to disperse UV and visible radiation. Prisms
have the disadvantage of requiring very high quality calibration and optics, hence diffraction gratings
have become the most popular source of monochromation in UV/VIS spectrophotometers. Gratings
have the advantage of being considerably less expensive and optically more efficient.
Adv. Spectr/Chrom 2.4
2. UV/VIS spectroscopy
2.6 Detectors
These convert electromagnetic radiation into electrons and hence produce an output of current which
can be interpreted as a measure of radiant energy reaching the detector surface. The devices most
commonly employed in UV/VIS instruments are photomultiplier tubes (the most common) and
photodiodes.
Photomultiplier tubes employ a photoemissive cathode which ejects electrons when a light particle
strikes its surface. These then pass through a series of amplifying dynodes, which attract the electrons
because of increasingly higher applied potentials. Dynodes produce several electrons for every one
electron striking their surface. Finally the avalanche of electrons reach an anode. Hence, a small initial
electron flow from the cathode produces a much greater electron flow at the anode. A small current
known as the dark current flows even when no light is striking the detector. This arises from several
sources, including the natural radioactive emission of ß-particles (high energy electrons) from the glass
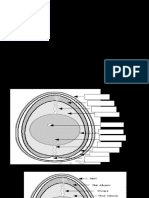
envelope surrounding the tube. Figure 2.2 shows the design of a typical photomultiplier tube.
Photomultiplier tubes have a limited
operating life due to breakdown of the
output photocathode, and cannot be exposed to the
+ve sunlight or bright room lighting. If this
electrod
happens, a large semi-permanent dark
current results, reducing the efficiency of the
tube at low light intensities when in
operation.
Photodiodes are semiconductor-based
devices which conduct a current when
electrons strike their surface. These devices
have excellent stability (give good baselines),
electrons dynode a good signal-to-noise ratio and are extremely
reliable.
Photodiodes are becoming popular, joined in
banks (or arrays), in multi-channel
instruments known as diode array
spectrophotometers. The advantages of such
instruments have been discussed in Chapter
light
sensitive 1. A typical UV/VIS diode array instrument
can produce a complete 200-800 nm
photon spectrum in 0.1 seconds. This is achieved by
using an array of diodes as a detector, instead
FIGURE 2.2 A typical photomultiplier tube design of conventional photomultiplier tubes. Each
(from www.physics.ubc.ca diode is responsible for detecting a small portion of the
spectrum (1-2 nm per diode) and all diodes operate at the one
time. Besides the speed advantages, these instruments do not drift in wavelength (their calibration is
always perfect). They have a disadvantage, however, in that they cannot resolve with any greater detail
than the width of the portion of the spectrum examined by each diode (1 or 2 nm). For most analysis
this is not a problem as resolution greater than this level is rarely required.
Adv. Spectr/Chrom 2.5
2. UV/VIS spectroscopy
What You Need To Be Able To Do
• define important terminology
• describe characteristics of organic and inorganic species that allow them to absorb UV/VIS
radiation
• choose appropriate cells and solvents for UV/VIS work
• list and describe common radiation sources used in UV/VIS spectrophotometers
• list and describe common detectors used in UV/VIS spectrophotometers
Revision Questions
1. Look up the structure of aspirin. It absorbs at around 275 nm. What structural components
cause it to absorb radiation?
2. What solvent would be appropriate to record as much of the UV/VIS spectrum for (a) caffeine
and (b) naphthalene. You should look up the structures of each of these to help with your
answer.
3. Why are the emission lines of deuterium above 375 nm a limitation on the lamp’s use in the
visible region?
4. Why is it important that the voltage to the tungsten filament is constant?
5. What is the meaning of “dark current” for detectors? What significance does it have?
6. What limits the resolution of a diode-array spectrophotometer? Why doesn’t it normally matter
that the resolution is typically 2 nm?
Answers to these questions on following page.
Answers to class exercises can be found in the Powerpoint file provided on the website.
Adv. Spectr/Chrom 2.6
2. UV/VIS spectroscopy
Answers to Revision Questions
To answer questions 1 & 2 requires you to find the structures for the compounds.
1. Absorption comes from 4 conjugated double bonds and multiple unbonded e's on oxygen
atoms.
2. Naphthalene is obviously non-polar, so hexane would be appropriate
Caffeine is fairly polar, so water or ethanol would be OK.
3. See deuterium spectrum at Wikipedia. The sharp peaks at 500-700 would be very difficult to
deal because very small changes in wavelength result in large differences in intensity. The gain
would be a problem.
4. Intensity is directly related to voltage.
5. Output from detector when there is no radiation hitting it. It reduces the sensitivity of the
detector, because it is more difficult to tell what is due to radiation and what is dark current.
6. The number of detectors. UV/VIS peaks are generally 30-50 nm wide, so a 2 nm bandpass is
more than adequate.
Adv. Spectr/Chrom 2.7
You might also like
- 2 - UV-VIS - Take Home Exam, Num5Document6 pages2 - UV-VIS - Take Home Exam, Num5Mheira VillahermosaNo ratings yet
- 2 Uv-VisDocument7 pages2 Uv-VisAnonymous xpYo0nR0No ratings yet
- Instrumentation: Sources of UV RadiationDocument16 pagesInstrumentation: Sources of UV Radiationexample2000No ratings yet
- Lecture 2Document54 pagesLecture 2bemnnashatNo ratings yet
- 2-UV-Vis Molecular SpectrometryDocument27 pages2-UV-Vis Molecular SpectrometryWahyuni EkaNo ratings yet
- Effects of Ultraviolet Radiation: Important Informations About UVC-LampsDocument3 pagesEffects of Ultraviolet Radiation: Important Informations About UVC-LampsHugo SantanaNo ratings yet
- UV Vis LectureDocument33 pagesUV Vis LectureJulie AlexanderNo ratings yet
- UV VIS Phenol LabDocument6 pagesUV VIS Phenol LabJoão Paulo FioriNo ratings yet
- Uv Visible SpectrosDocument6 pagesUv Visible Spectrospankajbhattb_pharmaNo ratings yet
- Analysis of Samples Absorbance Susing UV-VisDocument17 pagesAnalysis of Samples Absorbance Susing UV-VisRaiwata Mertanjaya100% (2)
- Ultraviolet-Visible Spectroscopy Refers To Absorption Spectroscopy in TheDocument7 pagesUltraviolet-Visible Spectroscopy Refers To Absorption Spectroscopy in Thekhubu88No ratings yet
- KK-CHP 2.4 (IR) Part 4 of 4Document33 pagesKK-CHP 2.4 (IR) Part 4 of 4ShafiqahFazyaziqahNo ratings yet
- E2297 22942 PDFDocument5 pagesE2297 22942 PDFvelu100% (1)
- DG-Lecture 1 - UV-VISDocument35 pagesDG-Lecture 1 - UV-VISArjun MaharajNo ratings yet
- UV Effect On PEDocument9 pagesUV Effect On PEDan Anghelea100% (4)
- Raman Spectroscopy Studies of Er3-Doped Zinc TelluDocument6 pagesRaman Spectroscopy Studies of Er3-Doped Zinc TelluYosej ParthNo ratings yet
- 621 Unit 6Document35 pages621 Unit 6AyeshaNo ratings yet
- Unit 12Document14 pagesUnit 12Fahra Aqilla AzzurahNo ratings yet
- Experience With Optical Partial Discharge DetectionDocument4 pagesExperience With Optical Partial Discharge DetectionDaniel Tamata SolorioNo ratings yet
- StrumentationDocument3 pagesStrumentationGirish Chandra MazumderNo ratings yet
- SP3321 Version Notes June 18Document7 pagesSP3321 Version Notes June 18Jose MezaNo ratings yet
- Flow Photochemistry PDFDocument28 pagesFlow Photochemistry PDFAli TunaNo ratings yet
- Chapter 6 Principle and Application of UV and Visible SpectrosDocument23 pagesChapter 6 Principle and Application of UV and Visible SpectrosEsuendalew DebebeNo ratings yet
- Atomic Spectroscopy - II (Solutions)Document3 pagesAtomic Spectroscopy - II (Solutions)LakshayNo ratings yet
- UV Visible SpectrosDocument13 pagesUV Visible SpectrosMumtahina EshaNo ratings yet
- 247 JMES 3877 AyekokeDocument7 pages247 JMES 3877 AyekokeArpit Kumar MishraNo ratings yet
- Chapter 26/27: Molecular Absorption SpectrometryDocument14 pagesChapter 26/27: Molecular Absorption SpectrometryS. MartinezNo ratings yet
- Chapter 5 Principle and Application of UV and Visible SpectrosDocument24 pagesChapter 5 Principle and Application of UV and Visible SpectrosEsuendalew DebebeNo ratings yet
- Electronic Spectroscopy UV-VisibleDocument28 pagesElectronic Spectroscopy UV-VisibleKarthikeyan G100% (1)
- An Introduction To Ultraviolet/Visible Absorption SpectrosDocument26 pagesAn Introduction To Ultraviolet/Visible Absorption SpectrosAndy Mejia ThanthawiNo ratings yet
- Spektroskopi UV-Vis (Prespektif Kimia)Document18 pagesSpektroskopi UV-Vis (Prespektif Kimia)Diah Ayu KinasihNo ratings yet
- Spectrophotometry: Spectra-Structure RelationshipsDocument4 pagesSpectrophotometry: Spectra-Structure RelationshipsAngel Rumiris GultomNo ratings yet
- Performance of UV-Vis SpectrophotometersDocument6 pagesPerformance of UV-Vis SpectrophotometersHuong ZamNo ratings yet
- Null 6Document10 pagesNull 6abrish fatimaNo ratings yet
- An Electromagnetic WaveDocument54 pagesAn Electromagnetic WaveKomal DahiyaNo ratings yet
- E2297-04 (2010) Standard Guide For Use of UV-A and Visible Light Sources and Meters Used in The Liquid Penetrant and Magnetic Particle Methods PDFDocument5 pagesE2297-04 (2010) Standard Guide For Use of UV-A and Visible Light Sources and Meters Used in The Liquid Penetrant and Magnetic Particle Methods PDFAnonymous PEHDuKRjoNo ratings yet
- Chemistrynotes Uv - VisDocument7 pagesChemistrynotes Uv - Visjavaunie wrightNo ratings yet
- 5 UV Visible Spectroscopy 2000Document27 pages5 UV Visible Spectroscopy 2000girma245selaleNo ratings yet
- Uv Analysis Method Development For Diclofenac and Paracetamol in CombinationDocument28 pagesUv Analysis Method Development For Diclofenac and Paracetamol in Combination0921py100% (5)
- 16527Document22 pages16527Jaikrishna SukumarNo ratings yet
- Chm622 Uv VisDocument50 pagesChm622 Uv VisAnonymous oSuBJMNo ratings yet
- CAPE CHEMISTRY Module 2 SpectrosDocument6 pagesCAPE CHEMISTRY Module 2 Spectrosasjawolverine100% (5)
- Hand Out Instrumen UV VisDocument4 pagesHand Out Instrumen UV VisArwa AsscNo ratings yet
- RD009EN00 - UV Technologies in Water Purification SystemsDocument12 pagesRD009EN00 - UV Technologies in Water Purification SystemsRonald SalasNo ratings yet
- Dmk3023 Chapter 3Document49 pagesDmk3023 Chapter 3NityantiniNo ratings yet
- PC 20166Document10 pagesPC 20166chopin.wlive.cnNo ratings yet
- Expt - UV-Vis Spectroscopy - ManualDocument5 pagesExpt - UV-Vis Spectroscopy - ManualSneha SNo ratings yet
- Rurini Retnowati - Molecule Organic SpectrosDocument84 pagesRurini Retnowati - Molecule Organic SpectrosAh-Maad AeNo ratings yet
- UV-absorbance Detector For HPLC Based On A Light-Emitting DiodeDocument5 pagesUV-absorbance Detector For HPLC Based On A Light-Emitting DiodeyulliarperezNo ratings yet
- UV-Vis InstrumentDocument7 pagesUV-Vis InstrumentNorizzatul Akmal100% (1)
- Laser Mass Spectrometry For Environmental and Industrial Chemical Trace AnalysisDocument16 pagesLaser Mass Spectrometry For Environmental and Industrial Chemical Trace AnalysisTamara WahidNo ratings yet
- Uv Visible SpectrosDocument48 pagesUv Visible SpectrosDhanvanth100% (7)
- UvDocument78 pagesUvebooksufiNo ratings yet
- Unit 2Document59 pagesUnit 2NTGNo ratings yet
- MFSN 802 PresentationDocument27 pagesMFSN 802 PresentationGibsonNo ratings yet
- ND: YAG Laser (Yttrium Aluminium Garnet) : (J. E. Geusic Et Al. at Bell Laboratories in 1964)Document32 pagesND: YAG Laser (Yttrium Aluminium Garnet) : (J. E. Geusic Et Al. at Bell Laboratories in 1964)KARTHIKEYA REDDY B SNo ratings yet
- Semester 4 Job Cards 1Document5 pagesSemester 4 Job Cards 1kulwantNo ratings yet
- Fuel Processing Technology: Maciej Sienkiewicz, Kaja Borz Ędowska-Labuda, Artur Wojtkiewicz, Helena JanikDocument8 pagesFuel Processing Technology: Maciej Sienkiewicz, Kaja Borz Ędowska-Labuda, Artur Wojtkiewicz, Helena JanikMEHDI FARROKHINo ratings yet
- H H H H: TOPIC: HybridisationDocument2 pagesH H H H: TOPIC: HybridisationssdsjknNo ratings yet
- Submitted by Submitted To: Adit Dubhashe MR - Rajesh JainDocument17 pagesSubmitted by Submitted To: Adit Dubhashe MR - Rajesh JainalpanaNo ratings yet
- INVENTORY CHECK For HIGG LASTDocument21 pagesINVENTORY CHECK For HIGG LASTDyeing Dyeing100% (1)
- Worksheet - 1 - Metals and Non MetalsDocument2 pagesWorksheet - 1 - Metals and Non MetalsSOULSNIPER 15No ratings yet
- Brain Targeting Drug Delivery SystemDocument35 pagesBrain Targeting Drug Delivery SystemOmotoso KayodeNo ratings yet
- Synthesis of ZnCu Metal Ion Modified Natural Palygorskite Clay TiO2Document8 pagesSynthesis of ZnCu Metal Ion Modified Natural Palygorskite Clay TiO2MihaiNo ratings yet
- Equation of StateDocument39 pagesEquation of StateOssama BohamdNo ratings yet
- API en - Atm.co2e.pc Ds2 en Excel v2 2764338Document28 pagesAPI en - Atm.co2e.pc Ds2 en Excel v2 2764338Lion HunterNo ratings yet
- Wesley Methodist School Klang MONTHLY TEST 1 (2019) Chemistry Form 4 Total: 50 MarksDocument8 pagesWesley Methodist School Klang MONTHLY TEST 1 (2019) Chemistry Form 4 Total: 50 MarksRenu SekaranNo ratings yet
- Aflas EngDocument11 pagesAflas EngVictor Flores ResendizNo ratings yet
- Fluid Mechanics Ii (Meng 3306) : Worksheet 1 Chapter1. Potential Flow TheoryDocument4 pagesFluid Mechanics Ii (Meng 3306) : Worksheet 1 Chapter1. Potential Flow TheoryAddisu DagneNo ratings yet
- 21cfr352 10Document2 pages21cfr352 10K.c. VerHageNo ratings yet
- Msds Theophylline AngDocument5 pagesMsds Theophylline AngTyaga MahardikaNo ratings yet
- 04 Combustion ChamberDocument6 pages04 Combustion ChamberprasannabalajiNo ratings yet
- Asme Material SpecsDocument4 pagesAsme Material SpecsRajwinder Singh100% (1)
- URINARY CRYSTALS NotesDocument5 pagesURINARY CRYSTALS NotesAlarice Cn100% (1)
- Chemistry Midterm Study GuideDocument7 pagesChemistry Midterm Study Guideapi-313911292No ratings yet
- Astm D 4972 - R (2007)Document3 pagesAstm D 4972 - R (2007)Bertina Salcedo RamosNo ratings yet
- 02a LASERs Session 2 (ND YAG Laser, He-Ne Laser and Semiconductor Laser)Document7 pages02a LASERs Session 2 (ND YAG Laser, He-Ne Laser and Semiconductor Laser)Bibaswan ChakmaNo ratings yet
- Structure of An EggDocument29 pagesStructure of An EggsheilaNo ratings yet
- Various Factor Affecting Stability of FormulationDocument46 pagesVarious Factor Affecting Stability of FormulationSandip Prajapati100% (1)
- Flash TanksDocument3 pagesFlash TanksnincitoNo ratings yet
- Bic Sodio Shandong MSDSDocument7 pagesBic Sodio Shandong MSDSFIORELLA VANESSA MAITA MUCHANo ratings yet
- Reaction CalorimetryDocument10 pagesReaction CalorimetryGrennioNo ratings yet
- Long-Term Aerobic Granular Sludge Stability Through Anaerobic Slow FeedingDocument12 pagesLong-Term Aerobic Granular Sludge Stability Through Anaerobic Slow Feedingsamuel campos carvalhoNo ratings yet
- Discovering and Developing Molecules With Optimal Drug-Like Properties (20 PDFDocument510 pagesDiscovering and Developing Molecules With Optimal Drug-Like Properties (20 PDFPaqui Miranda GualdaNo ratings yet
- Derakane - Tech SupportDocument32 pagesDerakane - Tech SupportJainam ShahNo ratings yet
- Journal Pre-Proofs: Forensic ChemistryDocument18 pagesJournal Pre-Proofs: Forensic ChemistryrichierieeNo ratings yet