Professional Documents
Culture Documents
Materials Science LE4
Uploaded by
Jewls Hatud0 ratings0% found this document useful (0 votes)
13 views2 pagesMaterials Science
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentMaterials Science
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
13 views2 pagesMaterials Science LE4
Uploaded by
Jewls HatudMaterials Science
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 2
Materials Science LE #4 Equilibrium – best described in terms of
a thermodynamic quantity called free
Chapter 9: Phase Diagrams
energy.
Component – pure metals and/or Free energy – function of the internal
compounds of which an alloy is energy of a system, and also the
composed. randomness or disorder of the atoms or
System – a specific body of material molecules (entropy).
under consideration, or it may relate to A system is at equilibrium if its free
the series of possible alloys consisting energy is at a minimum under some
of the same components, but without specified combination of temperature,
regard to alloy composition. pressure, and composition.
Solubility limit – maximum A change in temperature, pressure,
concentration of solute atoms that may and/or composition from a system in
dissolve in the solvent to form a solid equilibrium will result in an increase in
solution (ex: sugar-water system). the free energy.
Phase – homogenous portion of a Phase Equilibrium – systems in which
system that has uniform physical and more than one phase may exist.
chemical characteristics. Metastable – the state of equilibrium is
If more than one phase is present in a never completely achieved because the
given system, each will have its own rate of approach to equilibrium is
distinct properties. extremely slow.
When two phases are present in a Phase diagram – also called as
system, it is not necessary that there be equilibrium diagram.
a difference in both physical and Three externally controllable
chemical properties (ex: ice and water parameters that will affect phase
system). structure: temperature, pressure, and
When a substance can exist in two or composition.
more polymorphic forms (having both Unary Phase Diagram – one-
FCC & BCC structures), each of these component phase diagram, pressure-
structures is a separate phase because temperature diagram; composition is
their physical characteristics differ. held constant and temperature and
Homogenous – single-phase system pressure are variables.
Heterogenous – two or more phases Binary Phase Diagram – temperature
(ex: ceramic, polymeric, and composite and composition are variables while
systems). the pressure is held constant (1 atm).
The physical properties and the Maps that represent the relationships
mechanical behavior of a material between temperature and the
depend on the microstructure. compositions and quantities of phases
Microstructure – subject to direct at equilibrium.
microscopic observation, using optical
or electron microscopes.
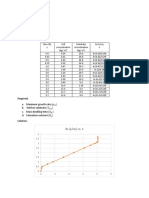
The copper-nickel system – possibly the Liquidus line – line separating the L
easiest type of binary phase diagram to and a + L phase fields; liquid phase is
understand and interpret. present at all temperatures above
this line.
Solidus line – located between the a
and a + L regions, below which only
the solid phase exists.
9.8 Interpretation of Phase Diagrams
1. Phases Present
Locate the temperature-
composition point on the diagram
and note the phase/s.
2. Determination of Phase
Compositions
3. Determination of Phase Amounts
Eutectic – melts and solidifies single
temperature
Eutectic structure –
Eutectic phase – phase that resides
in the eutectic structure
Primary phase – phase that was
formed prior to crossing the
isotherm
Microconstituent – an element of a
microstructure having an
identifiable and characteristic
structure
At temperatures below about 1080 Congruent transformation – no
degree Celcius, copper and nickel compositional alterations
are mutually soluble in each other in
the solid state for all compositions.
This complete solubility is explained
by the fact that both Cu and Ni have
the same crystal structure (FCC),
nearly identical atomic radii and
electronegativities, and similar
valences.
Isnomorphous – example is the
copper-nickel system; complete
liquid and solid solubility of the two
components.
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5796)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1091)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (589)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (345)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (122)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (400)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- Org Chem Lab ReportDocument5 pagesOrg Chem Lab ReportAyshee CapuchinoNo ratings yet
- Libro of GeologyDocument188 pagesLibro of GeologyEvelyn Ebenezer100% (1)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- Topics Engg Econ ProblemsDocument2 pagesTopics Engg Econ ProblemsJewls HatudNo ratings yet
- Reflected Ceiling PlanDocument1 pageReflected Ceiling PlanRonielle MercadoNo ratings yet
- Kinetics Probset (LE4)Document4 pagesKinetics Probset (LE4)Jewls HatudNo ratings yet
- Biochem Final RequirementDocument5 pagesBiochem Final RequirementJewls HatudNo ratings yet
- Kinetics Probset (LE3)Document1 pageKinetics Probset (LE3)Jewls HatudNo ratings yet
- EXPT1 - Lab ReadinessDocument2 pagesEXPT1 - Lab ReadinessJewls HatudNo ratings yet
- Synthetic Polymers: Fetalvero, Mico S. - Hatud, Jewls MDocument15 pagesSynthetic Polymers: Fetalvero, Mico S. - Hatud, Jewls MJewls HatudNo ratings yet
- Surface Imperfections: Vijeesh P Department of Physics The Cochin College, Kochi-2Document10 pagesSurface Imperfections: Vijeesh P Department of Physics The Cochin College, Kochi-2VIJEESH PNo ratings yet
- Applied ThermodynanmicsDocument93 pagesApplied ThermodynanmicsDiana Catalina Gallo100% (1)
- Dry Gas Seal Failure and Trouble ShootingDocument22 pagesDry Gas Seal Failure and Trouble ShootingDonNo ratings yet
- Mechanochemical SynthesisDocument46 pagesMechanochemical SynthesisYuppie RajNo ratings yet
- Performance Testing For Hot Mix AsphaltDocument51 pagesPerformance Testing For Hot Mix AsphaltProf. Prithvi Singh Kandhal100% (1)
- Petrophysics in Shale GasDocument26 pagesPetrophysics in Shale GasAtul Bhoil100% (1)
- Certifcato Di Collaudo/Test Certificate: Info@lame-Srl.191.itDocument2 pagesCertifcato Di Collaudo/Test Certificate: Info@lame-Srl.191.itMohamed Raafat100% (1)
- Beer BrewingDocument4 pagesBeer BrewingeupheemaNo ratings yet
- E 347GFDocument2 pagesE 347GFJohnston Carvalho100% (1)
- Chapter 6-Introduction To NanoelectronicsDocument83 pagesChapter 6-Introduction To NanoelectronicsshobiiiscNo ratings yet
- Guideline WHO - Guidance On Test Method Validation of in Vitro Diagnostic Medical Devices PDFDocument21 pagesGuideline WHO - Guidance On Test Method Validation of in Vitro Diagnostic Medical Devices PDFKelly CampenNo ratings yet
- Mech 5sem DMEDocument114 pagesMech 5sem DMEGanesh DongreNo ratings yet
- Quadring GB enDocument32 pagesQuadring GB en_Autarch_No ratings yet
- Carbotrap Carbon Black: An Excellent Adsorbent For Solvent or Thermal Desorption/GC Analyses of Airborne Organic CompoundsDocument4 pagesCarbotrap Carbon Black: An Excellent Adsorbent For Solvent or Thermal Desorption/GC Analyses of Airborne Organic CompoundsAndrew LiebermannNo ratings yet
- Fusion Splicing: Standard Sleeves: Dimensions & Applicable FiberDocument2 pagesFusion Splicing: Standard Sleeves: Dimensions & Applicable Fiberdict engineeringNo ratings yet
- Waz Vs AVAZDocument5 pagesWaz Vs AVAZblasuaNo ratings yet
- Taj Scientific Educational Equipment Plasticware Price ListDocument13 pagesTaj Scientific Educational Equipment Plasticware Price Listgeorge tacheNo ratings yet
- Image Contrast Enhancement For Deep-Sea Observation SystemsDocument6 pagesImage Contrast Enhancement For Deep-Sea Observation SystemsMohammed NaseerNo ratings yet
- Measurement of Earth Resistance and ResistivityDocument9 pagesMeasurement of Earth Resistance and ResistivityIsuru Kasthurirathne100% (4)
- Miscellaneous Treatment Methodselec3Document4 pagesMiscellaneous Treatment Methodselec3Edgelle Ejercito100% (2)
- SpiraxSarco B7 Self Acting Temperature ControlsDocument36 pagesSpiraxSarco B7 Self Acting Temperature Controlsdanenic100% (1)
- Review of Related Literature: Performance Evaluation of Corn Cob-Derived Activated Carbon On The Removal of Mercury and Lead On Contaminated WaterDocument10 pagesReview of Related Literature: Performance Evaluation of Corn Cob-Derived Activated Carbon On The Removal of Mercury and Lead On Contaminated WaterKenneth John Anthony TabagNo ratings yet
- PerkinElmer Lambda35 Manual EN PDFDocument114 pagesPerkinElmer Lambda35 Manual EN PDFLeonardo HerreraNo ratings yet
- 49.NCCI Torsion PDFDocument11 pages49.NCCI Torsion PDFJibbs OyawoyeNo ratings yet
- Topic 8 - Coordination CompoundDocument40 pagesTopic 8 - Coordination Compoundizz isalahNo ratings yet
- Introduction To Aerodynamics MITDocument505 pagesIntroduction To Aerodynamics MITalbertoroco100% (2)
- Test of Seal CoatDocument2 pagesTest of Seal CoatKUNAL PAULNo ratings yet