Professional Documents
Culture Documents
Untitled 2
Uploaded by
Mentari ZikriOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Untitled 2
Uploaded by
Mentari ZikriCopyright:
Available Formats
Cyclic Voltammetry Experiment
James J. Van Benschoten . J a n e Y. Lewis, a n d William R. Heineman
University of Cincinnati. Cincinnati, OH 45221
Daryl A. Roston' and P e t e r T. Klsslnger
Purdue University. West Lafayene. IN 47907
Cyclic voltammetry (CV) is a very useful modern elec- recorder, and an electrochemical cell (I) .The potentiostat used is a
troanalytical technique; its principles an d theories have been Bioanalyticsl Systems, Inc. CV-1R. Alternatively, any number of
discussed by Kissinger an d Heineman (I) . Despite t h e ad - commercially available instruments, or a homebuilt model con-
vantages of th e technique a n d its widespread use, there is a lack structed from conventional operational amplifiers can be used ( 3 .
of CV experiments in t h e literature for students. Thus , this 4 ) . The current versus potential curves are recorded on a Hewlett-
experiment has been designed specifically for introducing CV Packard model 7015B XY recorder, As mentioned in reference ( 1
-
to students who are a t t h e advanced undergraduate or earlv
maduate level. There are three oarts to this exueriment, -- "
), the ootential .~scan varies linearlv with time. thus it is
feasible to use a rrrip chart recorder, even tho&h 11 is not ns
wnvenienr as an X ) ' . I f an <,rcill~mcopeia nvailahlr, i t can be used
each of which ma y be performed independently. in place of the rerorder. The p~trntialoutput is
P a r t I uses t h e Fel1I(CN)c3-/Fe11(CN)s4-couule a s a n fi~lhwedindependently wwlth n Keithley model 178 digital voltmeter.
ex-ample of a well-behaveb e ~ e i t r o c h e m i c areversible~~ A three-electrode system is used for the CV experiment (I) .The
system in aoueous solution. Thi s exoeriment demonstrates deter - electrodes reauired are a Pt warkinc electrode (area = 2.5 mm2),a Pt
minition of t h e following: t h e formal reduction potential (E"'): auxiliary electrode, and either a A ~ I A ~ C orI a saturated calomel
the number of electrons transferred in the redox process in);the electrode (SCEI reference electrode. All ootentials in this exoeriment
are referenced veraur. %'E The cell used la a H~mnalyticalSystems.
diffusion coefficient (D);electrochemical reveriibility; an d t h
In,. \'('-2ylaw wal wit h a t i t m l top. The u,p ha., iour hdes machmed to
e effects of varying concentration an d scan rate. nerommudate the three rlectn~desand a t u h for detrxygenating by
P a r t I1 investigates t h e electrochemical behavior of acet- bubbling with a stream of Ns.
aminophen, a n ingredient in many over-the-counter phar - Procedure. Pretreatment of the platinum working electrode surface
maceuticals (e.g., Tylenol*, Excedrin*, Nyquil*, etc.). T h i s may be required. Simply polishing the surface with powdered
organic system is more complex, involving chemical a s well a s alumina and rinsing thoroughly with distilled water should suffice.
electrochemical reactions. Principles t h a t are illustrated include: The elec-trode can be sonicated in an ultrasonic bath if available.
effect of a coupled chemical reaction o n t h e appear-ance of t h e The cell, as shown in reference ( I ) ,is assembled and filled
CV; effect of p H o n th e appearance of th e CV an d o n t h e with 1 M KNOJ just until the ends of the electrodes are immersed
(Note: avoid tilting the SCE; this may disturb the HgIHgnCln and
mechanism of reaction; use of scan rate for elucidating result in incorrect potentialsl. The system is deoxygenated by
mechanistic information; an d quantitative determinationof purging with Nz for approximately 10 mi". Following this, N2 is
acetaminophen in a l'ylenolm tnhlet. allowed to flow over the solution to prevent 0 2 from re-entering
P a r t 111-is designedto introduce t h e studen t t o practical the eell for the remainder of the experiment.
considerations in a cyclic voltammetry experiment. Instead of
-
While the system is beingdeoxygenated, the scan parameters can be
followine recommended conditions t h e studen t now is set. The working electrode should be disconnected or switched off
persuaded t o investigate factors ~ u c has: t h e effect of 02o n during this procedure. The initial potential is set at 0.80 V, and the
scan limits at 0.80 V and -0.12 V using the recorder as a monitor. All
th e C'Y: choosinr the aoorooriate... electrode: ~srablishin aPO- scans are initiated in the negative direction with a scan rate of 20
tential $can lim& an d studying Pt electrode surface waves. It is mV/s. These settings will be used unless otherwise specified.
honed that . uoon comoletion of this section, t h e studen t can When deoxygenation is complete, the working electrade is
effktively design a n eiectrochemical experiment for a n y switched on. After allowing the current to attainaeonstant value
particular chemical system of interest. (in about 10 s), the potential sean is initiated and a background
CV of the sup-porting electrolyte solution is obtained.
Part I-Fundamentals of Cyclic Voltammetry After turning off the working electrode, the eell is cleaned and
T h e Fe111(CN)6"-/Fe"(CN)$L- couple is known t o b e well- re-filled with 4 mM KaFe(CNI6 in 1M KNOs. Following the
behaved both chemically an d electrochemically (2). As such, i t same pro-cedure as above, a CV of the Fe'1L(CN)i"/Fe1'(CN)8-
is often used a s a model system in electrochemical experi-ments. couple is ob-tained.
The effect of the sweep rate ( u ) on the voltammoprams is
I t ca n b e used t o determine electrode areas a n d to diagnose observed by using this same soiutionand recording CV's at the
problems associated with new electrochemical cell designs. In following rates: 20,50,75,100,125,150,115,and 200mV/s.
this experiment, t h e couple is used t o clearly demonstrate s o m Retweeneach scan, initial conditions at the electrode surface are
e ~ i m p o r t a n tprinciples of cyclic voltam-metry. restored by moving the working electrode gently up and down
without actually removing it from so-lution. Care should be taken
that no buhbles remain on the elec-trodes.
Experimental ('wwenrration likewiseaffrcts the magnrtudr of thr penkcurrent.
This i seen by uhtainmg +cansof 2.6.8. and I d m A f K
Reagents. All chemicals are reagent grade and are used assuch. A
10O-mlstock solution of apprnximateiy 10 mM K3Fe(CNk is prepared ,l.'t.tC'Fi~fiuang nswwo .~
rate uf 20 mV,s A \r,ltarnmoeram of tne
in 1 M KN03. From this, 25 ml each of approximately 2,4,6, and 8 mM unkn<,wnK8nCN)a solution should be ru'n as well.
solutions in 1 M KN03 are made. In addition, a 25-ml K3Fe(CN)6 The affect of the supporting electrolyte on the appearance of the
solution of approximately 4 mM is prepared in 1 M NazS04.All so- CV isdemonstrated by recordingvoltammogramsof (1) 4 mM ferri-
lution concentrations must be accurately known. An unknown cyanide in 1 M KNOs and (2) 4 mM ferricyanide in 1M Na2S01.
KsFe(CN)fisolution should be provided by the instructor.
Apparatus. The cyclic voltammetry apparatus consists of a po- Results and Discussion
tentiastat with potential sean capability, a voltmeter (optional), a Using th e dat a obtained, a number of simple calculations can
he done tha t will yield considerable information about th e ferri-
Current address: Department of Chemistry. Northern Illinois ferrocyanide system. Anodic peak current (i,) cathodic peak
Uni-versity. DeKalb. IL 60115. current (iw), E"', and n can be determined as described
772 Journal of Chemical Education
" ' I
0.8 04 00 ;-loL
WTENTIAL V vr SCE 2.04.06.0 6.0 100
CONCENTRATION mM
POTMTIAL. V rr 5CE F i a r e 2. IAl.. Cvcllc. voltammmam of KIF~CN)-.... In 1 MKNO*. Scan rate =
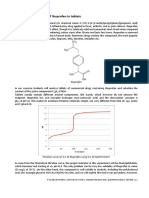
Figure 1. (A) Cyclic voltammograms of 4 mMKsFe(CN)sin 1 MKNOJ. Pi 20 mVls. PL elechade. Concentration = 2. 4, 6. 8, and 10 mM. (8)Plot of i, versus
elec-bade. Scan rate ( 4 = 20. 50. 75. 100. 125. 150. 175. end 200 mVls. (8) concentration and i, vsrsus concentration horn voltammograms in (A).
Plot 01 iw YBTSUI v ' ' ~and ,1 versus uU2 horn voltammogams in (A).
in reference ( I ) using the voltammogram of 4 m M tions should span the range of 0.10 to 5.0 m M ; 10-25 ml of each
K3Fe"'(CN)6 in 1M KN03. are needed.
An unknown solution is prepared by dissolving a Tylenolm
The effect of the scan rate, v, on the appearance of the CV tablet in 250 ml of pH 2.2 buffer. A workable concentration is
can be seen in Figure 1A. As described by the Randles- obtained by diluting a 5-ml aliquot of this solution to 50 ml.
Sevcik equation (I),,i and ip, i n c r e m as u"~. A plot of this All APAP solution concentrations need to be accurately known.
equation (Fig. 1B) yields a straight line, the slope of which can be Apparatus. The apparatus used in this experiment is identical to
that in Part I with the exception of the carbon paste workii
used to determine the diffusion coefficient (D in cm2/s) of the electrode (area = 9.1 mm2).
system. T h e area of the working electrode will either have to be Procedure. The carbon oaste electrode is ~renared ..as described
provided by the instructor or experimentally deter-mined. in the Hiuanalytirnl Sysrems. Inc. manual. Thw elrrtrc,dt must he
Evidence indicating that the system is electrochemically carefully parked tu avoid erroneous resulu and rare ;lhould c,e
reversible (discussed in reference ( I ) ) can he seen in Figure 1A mken not to gauge it once it is ready for use.
as well. T h e separation of the peak potentials (AEJ is i n d e ~ Using the 3 m M APAP solution in pH 2.2 buffer, scan limits are
e n d e noft v. This can he discussed bv the student. established at 1.0 V and -0.2 V. Scans are initiated in the positive direction
at 0.0 V. Cvclic voltammomams of the 3 mM APAP solution in each of
The Kandlrs-Sevcik equation ( 1 )also indicates that i, and the 3 buffers are then obcined at a scan rate of 40 mV/s and 250 mV1s.
...i are d i r e d v..oro~urtional.to concentration. The effect of The solution should be stirred between each run.
increasing concentration on the appearance of the CV can be seen As was demonstrated in Part I, CV's can be used for
in Figure 2A. A calibration curve (Fig. 2B) can be con-structed quantitative analysis. A standard curve can be constructed using
and used to determine the concentration of the un-known the series of 5 APAP solutions in pH 2.2 buffer. From this, the
ferricvanide solution. concentration of APAP in the Tylenol solution is determined.
A formal potential as reported in the literature is a working Results and Discussion
number that is dependent on the conditions of the experiment
(5). T h e effect ofthe particular supporting electrolyte used is The oxidation mechanism of APAP is as follows
demonstrated by comparing CV's of K3Fen1(CN)6in KN03 and
Na2SOa.
Part ll-Effect of Coupled Chemlcal Reactions
Acetaminophen (N-acetyl-p-aminophenol, APAP), the active
-
ineredient in Tvlenol". is commonlv used as an a s ~ i r i n OH
I
11 111
substitute. However, unlike aspirin, it is known to cause liver and NAPQI
APAP
kidnev damaee when administered in laree amounts. I t is suspectld
that :metabolite of APAP is the actual hepato-toxic agent, thus
APAP and its metabolites have been ex-tensively investigated (6).
Voltammetric studies in aqueous solution have revealed
chemical as well as electrochemical steps (7). The APAP system
therefore is useful in demonstrating the mechanistic information
that can he obtained from CV's.
Experimental
Reagents. All chemicals are reagent grade and are used as such. APAP is electrochemically oxidized in a pH-dependent, 2-electron.
The fallowing supporting electrolyte solutions are needed: 500 ml 2-proton Drocess to N-acetvl.-.D-.~uinoneimine (NAPQI') (step
of pH 2.2 and 200 mlof DH6 Mcllvaine buffer with an 1). ~ i occurrence of follow-up chemical re-
ionicstrenethof 0.6 M (8);200 ml of i.8 M HSSO~. actions involving NAPQl is pH. dependent. Bv varving the pH
. . . - .
A stock solution of approximately 0.070MAPAP is prepared in0.05 of the media and the scan rate ofthe cyclic voltammetry ex-
M perehlaric acid. This should be kept in the refrigerator when not periment, chemical reactions involving NAPQI can be "mapped-
in use. Using the stock solution, 10-25 ml of an APAP solution
is prepared in each of the three supporting electrolyte solutions. out." At p H values 26, NAPQI exists in the stable unprotonated
The concentration of these APAP solutions should be form (11). Cyclic voltammograms recorded for
approximately 3 mM. N A P a t pH 6 are shown in Figure 3. Reasonably well-defined
For the purpose of establishing a calibration curve, four additional anodic and cathodic waves are evident. The anodic current
APAP solutions in pH 2.2 buffer are needed. These four concentra- represents step 1in the mechanism detailed above while the
Volume 60 Number 9 September 1983 773
E,VOLTS vr Wepcl
//
1
roe 70.4r 0 . z
Figure 3. Cyclic voltammogramr of 3.6 mM APAP in pH 6 Mcllvsine buffer.
Carbon paste electrode.
cathodic current represents the reverse of this step. The
similarity in appearance of the p H 6 cyclic voltamm~grams
observed with 40 and 250 mV/s scan rates indicates that the Figure 4. Cyclic voltammcqrams of 3.6 mMAPAP in pH 2.2 Mcllvaine buffer.
involved species are stable in the time domain of the cyclic Carbon paste electrode.
voltammetrv experiment. The large separation between the
anodic andcathodic peak currents &the pH 6 cyclic vol-
tammograms is a manifestation of sluggish electron transfer
kinetics.
Under more acidic conditions, NAPQI is immediately
protonated (step 21, yieldmg a less itablebut elrctn,cherni-calls
artivr species (1111 ahirh rapidlv yields (step ;I) a hy-clrnted
firm I IYr that is elertroch~~micallvinactiw at the ex-
-, ,
amined potentials. Cyclic voltammograms shown in Figure 4
are consistent with this mechanism. The DH of the media was
2.2. A small cathodic wave due to the reduction of pro-
tonated NAPQI " (111). is evident when the scan rate of 250 mV/s
.
is employed. This wave is even more pronounced when faster
scan rates are emnloved:." .however. faster scan rates reauire the
use of an oscilloscope to record thk current-potential rksponse.
With a slower scan rate of 40 mV/s, a cathodic wave for the
reduction of protonated NAPQI is not observed. All of the
protonated NAPQI (111) is converted to the inactive hydrated
form (IV) before negative enough potentials are reached durinz
the reverse scan of the cvclic voltammetrv exoeri-
...-.. .
Hvdrated NAPQI (IV) converts (step 4) to benzoquinone;
however, the media has to he extreme& acidic for thk rate of the
process to be sienificant enough that reduction of benzo-qui&ne
is observed during the cyclic voltammetry experi-ment. The
media for the cyclic voltammograms detailed in Figure 5 was 1.8
M HzS04. A poorly defined cathodic wave for the reduction of
benzoquinone (V) was observed when a scan rate of 250 mV/s Figwe 5. Cyclic wliamwmms of 3.6 mM APAP in 1.8 M W . . Cmbm psste
electrode.
was employed. The reduction wave is broad because the
formation of benzoquinone (V) from hydrated NAPQI (IV)
occurs during the reverse scan. When the scan rate is 40 mV/s, the
increased length of time required to reach negative enough determining whether 02 is an interference. Generally, this is
potentials during the reverse scan allows for the accumulation of accomplished by some amount of trial and error. This effort is
benzoquinone (V). Consequently, a well-defined reduction wave justified, however, because as shown in parts I and 11, con-
was observed for henzoouinone ( V ) when the slower scan raw siderable information can be obtained from a relatively
wnsernployrd.The sermd scan in thr nositive direction virld3 i u simple experiment. This section demonstrates how these
~anodic wave in addition to that oE APAP which coriesponds to
selections are made, thus giving the student insight on how
the oxidation of hydro-quinone, the reduction product of todesign an effirirnt C V experimrnt for almost anv chemical
benzoquinone. systtm of interest.
Part Ill-Dissolved Oxygen, Potential Limlts, Surface Experimental
Elfects Reagents. All chemicals are reagent grade and require no
When investigating a chemical system for the first time with further purification. Solutions of approximately 25 ml1.0 M
H2SO&and 25 ml 1.0 M KNOu are required.
cyclic voltammetry, there are several experimental conditions that A ~ o a r o t u sThe. electrochemical aooaratus is described in Dart I
need to be established. Among these are choosing the appropriate
electrode, solvent, and supporting electrolyte; and
774 Journal of Chemical Education
O1 REMOVED
0.4 0.0 -0.4 -0.8 -1.2
POTENTIAL, V vs SCE
Figure 6. Cyclic volternmqlrams ol 1.0 MKWsshowing effect of OZ.Oxygen
rernovd by deaxygenating wilh N2 fn 10 min. Au electrode. Scan rate = 50 v' ,
rnV/s.
1.2 0.9 0.6 0.3 00 -0.3
POTENTIAL, V vs SCE
Figure 8. Cyclic valtarnmqlram showing R eisctrDMt surface waves
in 1 M H2S0,. Scan rate = 50 rnV/s.
Plntinum makw a very interwing elcctrde i n that several surface
wnves npprar in a 1,ackgmund rean. Thmr wavra ran he
investigated in a dwrygmated solution of 1.0 M HJOI wing a
pdirhed electnde. A voltammogram is obtained by initiating the
scan in the negative direction at 0.30 V with potential limits of
1.425V and -0.225 V. The positive potential limit is progressively
stepped in to 0.825 V (in 0.15 V increments) obtainine a CV at eaeh
new limit. Leavine ~-this limit at 0.825 V, the negativepotential
limit is then likewisestepped in to 0.075 V (in 0.075 V increments)
obtaining a CV at each new set-ting.
Results and Discussion
Though a compound is not chemically altered by 0 2 (i.e.,
oxidized, degraded, etc.), the voltammogram of that com-pound
may nonetheless be subject to 0 2 interference. This results from
the fact that 0. is electrochemicallv active (i.e..
reducible to Hz02 and HZO~These. two process"es should h i
amarent from the CV's of oxveenated 1.0 M KNO* (see Fie.
f3j.'1f the compound of i n t e r e z e d u c e s in the samepktential
-.
region.-. deoxveenation is a necessarv ~rocedure .
Choosing the appropriate solvent, supporting electrolyte, and
I . working electrode for a CV e x ~ e r i m e nreauirest several
rtmsiderati(;n.s.The major requirements of the xivent are that the
1.6 00 0.0 -08 -1.6
POTPITIAL, Y vr SCE iveries of interest he soluble and stahle in it. The SUI>-porting
electrolyte must provide a relatively high ionic strength (>0.01 M
F r g ~ e7 Cyclic voltarnmDgrams of 1 0 M <NOs bs.ng d tlerenl wommg ) and be compatible with the svstem. A
slsc-bmes (Alglassycarbon. IBicaman pase. ICIAU.ID1 Fi Cycilc further consideration is the window that is available. In aqueous
vohmmogsm 01 1 0 MH,SO. mng IE) Au electrode Scan rate = 50 mVls
solution, this is often limited on the negative end by the reduction
9.1 mm2),and an Au electrode (area = 2.5 mm2)are used. Any or all of
of HzO to H2and on the positive end by the oxidation of Hz0 to
these electrodes may he investigated depending on availability. 0 2 . It is influenced by the choice of working electrode, solvent,
Procedure. The effect andlor interference of dissolved 01in the
"
and supporting electrolyte. Elec-trolysis of the species of interest
supporting electrolyte is readily determined. Using the Au working must occur within this range for a CV experiment. Figure 7 (A-D)
electrode (polished as was the Pt electrode in part I) acyclicvoltam- shows the potential windows provided by several working
mogram of 1.0 M KN03 with scan limits of 0.7 V and -1.0 V is run. electrodes in 1M KNOs. Figure 7C and 7E demonstrate the
This is initiated at 0.40 V in the negative direction with a scan rate of
50 mV1s. The solution is then deaxygenated for 15 min as described in influence that two different supporting electrolytes, 1 M KN03
part I, and a CV of the oxygen-free solution is recorded. and 1.0 M H2S04 re-spectively, can have. These illustrate the
The potential window for variouselectrodes can be just as readily
determined. At the low current settines .. tvoicallv. used in CV. this is pH dependence of the Hz0 electrolysis waves.
, Metals such as Au and Pt are not totallv inert as once thought (
generally l~m~red either end hg large offrcnlr wnwz ['ring n 1.0
M 9 , l O ) .When used as electrodes, ~ t a n d exhibit~u surface
K N h sdutim, n scan rntr oi 50 my s, and a current range of ap- waves which limit the usable votential ranee. At an Au electnde
proxmately hll pA in.. the worknhlr pute~,tialrange can be (as shown in Fig. 7 E ) these waves are atrributed to adiorhed
deter-mined for eaeh electrode. oxide furmation and redurtion at low pH. Figure H illustrates the
Not only is the potential range dependent on the particular elec- numerous l't surface waves. At negative po-tentials, fmnation
trode used, but alsoon the suppurtingelectrolyteused. This isdem- and oxidation ol adsurhed Hv UCCUW: in the positive region,
onstrated by comparing the potential limits available on an Au formation and reduction of adsorbed oxide
working electrode in 1.0 M KN03 and 1.0 M H2S04.
Volume 60 Number 9 September 1983 775
14) Kimingcr, P. T., in "Labcrstory T ~ h n i q u e ain E l e c t r 4 y f i c a l ChemhV/."KYsbger.
is observed. These waves have been studied and can be used to P. T,and Heineman. W. K. IEditomI Marrel Dakkar, New York, in pma. Chapt.
determine accurately electrode area (9,10) . As demon- E.
strated in Figure 8, if the potential range is sufficiently (51 ~ d t h o f fI.. ~ . . ~ n m a i e cWk,. J . J ~ h y chems. 39.945 (1935).
(fil Miner. I). .I.. Rice. J. H..Rimin. R. M.. Kiasin~or.P. T.. Aml. Chsm.. 53. 2258
limited, a very clean workable background can be obtained. 119811.
171 Preddy,.....-C. A . Miner. D. .I., Meinsma. D. A . Kiasinpr, P. T., Anal. Chem., sub-
-;* -A ".
Literature Cited (8) Riving. P. J., Markoulitz. J. M.. Rorenthal. I .. Ami . Chm~.28.1179(1956).
(91 sayer, n. T.. ~nlxrtaJX...I. ~ . . - ~ ~ ~~ ~l ~i t ~m ~h ~t ~forchemistaeis tl ~
11) Kissinwr. P. T.. Heineman, W. R.. 3. CHEM.EDUC..EO, 702 119831. ." widey.
(21 DeAn~e1is.TP. ., Heineman, W. R., J. CHBM. BDVC..53.94 11976l. Now York. 1914. pg. -9.
(3) Schwan. W. M.,Shain, I., A n d Chem..35.177011963). I101 Bsuer. H. H.. "Electrdie : Modern Ideas Concerninr E l r d r d o Reactions."
Gwrg Thismr l'uhl~sherr,StutOa~.Gsrmany1972.. ~ pl W. 1 0 3 .
776 Journal of Chemical Education
You might also like
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (122)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (589)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (401)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (842)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (897)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5806)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (345)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1091)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- International Standard: Hexagon Regular Nuts (Style 1) - Product Grades A and BDocument14 pagesInternational Standard: Hexagon Regular Nuts (Style 1) - Product Grades A and BUdit Kumar SarkarNo ratings yet
- Microbiology With Diseases by Body System 5th Edition Bauman Test BankDocument25 pagesMicrobiology With Diseases by Body System 5th Edition Bauman Test BankRhondaHogancank100% (50)
- Experiment No. 6 SpectrosDocument6 pagesExperiment No. 6 SpectrosArianne Balaoing80% (5)
- Quiz 1 InorgDocument5 pagesQuiz 1 InorgDanielle Lois AbagNo ratings yet
- High Performance Liquid Chromatographic Technique and Validation For Determination of Favipiravir in Bulk and Tablet FormulationDocument7 pagesHigh Performance Liquid Chromatographic Technique and Validation For Determination of Favipiravir in Bulk and Tablet FormulationEditor IJTSRDNo ratings yet
- Electrochemical Energy Systems: IIT KanpurDocument12 pagesElectrochemical Energy Systems: IIT KanpurAjeet kumarNo ratings yet
- Redox Reaction _ Practice Sheet __ JEE ChallengersDocument5 pagesRedox Reaction _ Practice Sheet __ JEE ChallengersCalming MusicNo ratings yet
- Module 6 Lecture Notes: 6.1 An Introduction To LogarithmsDocument19 pagesModule 6 Lecture Notes: 6.1 An Introduction To LogarithmsAlain LeeNo ratings yet
- Chất hấp phụDocument19 pagesChất hấp phụbachtrantrung08111999No ratings yet
- 14 Addition of Enolates To Enones The Michael ReactionDocument8 pages14 Addition of Enolates To Enones The Michael ReactionNikola NinkovNo ratings yet
- Biology Paper 5 PracticalDocument5 pagesBiology Paper 5 Practicalpro.is.only.me.cause.i.am.vrindaNo ratings yet
- Bio Data-Srinu Naik-UpdatedDocument8 pagesBio Data-Srinu Naik-Updatedharsha bathineniNo ratings yet
- Rubber Gloves Manufacturing Process Application Note B212055ENDocument3 pagesRubber Gloves Manufacturing Process Application Note B212055ENMuhammad ZikryNo ratings yet
- Chem Notes 1Document20 pagesChem Notes 1ElMicoNo ratings yet
- Chapter 4-Chemical EquilibriaDocument103 pagesChapter 4-Chemical Equilibriahulk alanNo ratings yet
- Cen 09803 Acsnews4 PDFDocument2 pagesCen 09803 Acsnews4 PDFNurafindaNo ratings yet
- Unit 4: Defect Chemistry and Defect Equilibria: 1) Concentration of Intrinsic DefectsDocument7 pagesUnit 4: Defect Chemistry and Defect Equilibria: 1) Concentration of Intrinsic DefectsTejinder SinghNo ratings yet
- Acrodur DS 3530 February 2018 R3 EDFDocument3 pagesAcrodur DS 3530 February 2018 R3 EDFAllen de RichardNo ratings yet
- 232 693 6 PB PDFDocument11 pages232 693 6 PB PDFFadillah RamadaniaNo ratings yet
- Starch and GlycogenDocument20 pagesStarch and GlycogenLaksilu Viduraga Peiris100% (1)
- Difference Between Polycarbonate and ABSDocument5 pagesDifference Between Polycarbonate and ABSNeelakandan DNo ratings yet
- Ams 4911LDocument7 pagesAms 4911Ljamesdong1120No ratings yet
- Experiment-3 Metals and Their Properties UpdatedDocument4 pagesExperiment-3 Metals and Their Properties UpdatedSnehaNo ratings yet
- EVALUASI OUTLINE SPEK & DISAIN WTP (10 MARET 2023) - 1bi-Op PDFDocument4 pagesEVALUASI OUTLINE SPEK & DISAIN WTP (10 MARET 2023) - 1bi-Op PDFMartindarma TugasNo ratings yet
- Penelitian Pengembangan Minyak Atsiri Sebagai Aromaterapi Dan Potensinya Sebagai Produk Sediaan Farmasi MuchtaridiDocument9 pagesPenelitian Pengembangan Minyak Atsiri Sebagai Aromaterapi Dan Potensinya Sebagai Produk Sediaan Farmasi MuchtaridiFitria Dewi 'uthie'No ratings yet
- Roquette Cosmetics Brochure Formulation Guide A6Document11 pagesRoquette Cosmetics Brochure Formulation Guide A6Sathyapriya SubramaniNo ratings yet
- Chemistry EavporationDocument20 pagesChemistry EavporationSouvik JEE 2024No ratings yet
- Technical Data: Plastic Steel 5-Minute Putty (SF)Document4 pagesTechnical Data: Plastic Steel 5-Minute Putty (SF)Hoàng DũngNo ratings yet
- 8B. Acid-Base Titration of Ibuprofen in TabletsDocument2 pages8B. Acid-Base Titration of Ibuprofen in TabletsAhmed Salih100% (1)
- Be You TifulDocument51 pagesBe You TifulnelsapurwaningsihNo ratings yet