Professional Documents
Culture Documents
Beyond Lab Research On White Rust and Passivation Nagle
Uploaded by
Max JunghannsOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Beyond Lab Research On White Rust and Passivation Nagle
Uploaded by
Max JunghannsCopyright:
Available Formats
Beyond Laboratory Research on
White Rust and Passivation
Christopher J. Nagle, EVAPCO, Inc.
10 the Analyst Volume 24 Number 3
Abstract
In support of customer requests for better outcomes
for new equipment commissioned with immediate
heat load, Evapco initiated an in-depth study to
investigate the formation and control of white rust.
This study, which began over three years ago, led to the
development of industry leading research equipment
specifically designed to evaluate white rust formation on
evaporative heat transfer surfaces. A small-scale closed-
circuit coolers was designed and built to replicate the
dynamic conditions associated with evaporative cooling
equipment commissioned with immediate heat load.
In addition to proprietary research, controlled
testing was performed using commercially available
products marketed as either white rust inhibitors
or passivation aids for evaporative cooling systems
containing galvanized materials of construction. The
controlled testing included routine wet chemistry, visual
inspections, and photography to determine how quickly
white rust formed and the relative percentage of surface
area impacted. Additional research was conducted to
focus on innovative pre-treatment technologies for the
galvanized coils utilized in closed-circuit coolers and
evaporative condensers. This research effort continued
with a goal of providing a factory applied pre-treatment
capable of minimizing the formation of white rust in
evaporative cooling equipment commissioned with
immediate heat load across a wide variety of makeup
water qualities and water treatment formulations.
A Brief History of White Rust in
Evaporative Cooling
For over 50 years the evaporative cooling industry
has relied on galvanized steel as a preferred material
of construction due to its cost-effective combination
of corrosion protection and long service life. The
galvanizing process provides the underlying steel with
a coating of superior hardness, ductility, and adherence
unmatched by any coating or painting process.
In the late 1980s and early 1990s, articles began to
surface noting an increased occurrence of premature
corrosion of new galvanized steel referred to as white
rust. A 1992 article in The Analyst noted, “The problem
with white rust corrosion of cooling water towers was
not widely observed until the mid-1980s.”1
The Cooling Tower Institute published Guidelines for
Treatment of Galvanized Cooling Towers to Prevent White
Rust (1994). The Association of Water Technologies
published White Rust: An Industry Update and Guide
Paper (2002 and 2012).
11 the Analyst Volume 24 Number 3
Beyond Laboratory Research on White Rust and Passivation continued
The 2002 version of AWT’s guide paper stated, “The Hot dipped galvanized coils did not receive a chrome
corrosion of galvanized steel cooling towers may be rinse but experienced an increase in observed white rust
referred to as white rust and the consequence of white along with mill galvanized components.
rust can be premature
2 failure of galvanized steel
components.” The chrome rinse “helps prevent initial corrosion and
staining until the steel is fabricated and placed into
Several of the early articles1-3 on the subject refer to service.”4 It is unlikely that a chromate rinse applied to
“cooling towers” generically as an apparent catchall for mill galvanized steel would provide long term protection
the broader array of evaporative cooling equipment. against white rust on its own as it would “wash away” or
When it comes to the topic of white rust and passivation, “wear off” as water is circulated across a tower, cooler, or
a distinction should be drawn between open cooling condenser.
towers constructed of mill-galvanized steel for basins
and casing as distinct and different from closed-circuit Not all mill galvanizers eliminated the use of hexavalent
coolers and evaporative condensers constructed of both or trivalent chrome rinses. In the early 1990s, Johnson
mill galvanized steel and hot-dipped galvanized steel and Mihelic noted “Traces of the chromate anion can be
coils. A chemical formulation used successfully to found on the surfaces of all 14 coupons utilized in this
pre-clean and passivate an open cooling tower started test, ..., and is still done by at least some manufacturers
without load may not be a good technical selection to of mill galvanized steel.”4 To this day, the majority
passivate a closed-circuit cooler or evaporative condenser of mill galvanized steel used in the manufacture of
commissioned with immediate heat load, as shown evaporative cooling equipment in North America
in Figure 1. receives a hexavalent chrome wash.
Figure 1: Evaporative Condenser With White Rust As early as 1990, papers discussing white rust corrosion
noted that “Because of the increasing restrictions on the
use of chromate-based corrosion inhibitors for use in
cooling water, a general trend toward alkaline cooling
water treatment has become an industry standard.”4
The precise timelines will need to be left to the water
treatment professionals who were practicing in the field
during the 1970s and 1980s. The generation of water
treatment professionals raised on alkaline treatment
programs could view the decrease in usage of pH control
associated with chromate and stabilized phosphate
treatment programs, the ascent of alkaline treatment
programs, and the increased incidence of white rust
on newly commissioned galvanized equipment as one
common timeline. In this transition from the dominance
of pH control to alkaline treatment programs, it was
As white rust became more prevalent in the mid 1980s, observed “Higher pH levels combined with newer
customers, engineers, and water treatment professionals deposit control agents may minimize corrosion, scaling,
searched for explanations. Some of the early articles and fouling in the coolers but can simultaneously cause
pondered what impact an increase in “aluminum content premature failure of a cooling tower made of galvanized
of the zinc alloy”1 or “the elimination of chrome rinse by steel.”5
the galvanizers”2 might have on the observed “increase Regardless of the possible or probable root causes for
in the incidence of white rust in cooling towers.”1 the uptick in observed white rust, which began in the
For decades, it was well understood that “The chromate mid 1980s, the problem persists for today’s customers
anion is extremely effective at minimizing the formation and their water treatment providers. In 2010, the
of white rust and at inhibiting corrosion at zinc International Institute of Ammonia Refrigeration held
surfaces.”4 Therefore, it would be logical to assume that their 32nd annual conference in San Diego, California.
the elimination of a chromate-based rinse from a mill Discussions with contractors and customers highlighted
galvanizing process might be a contributing factor to the challenges associated with passivating galvanized
the observed increase in white rust. There are likely at condensers for an industry facing shorter equipment lead
least three reasons why this explanation should not be times coupled with the near universal requirement for
considered the sole root cause: immediate heat load upon commissioning.
12 the Analyst Volume 24 Number 3
Beyond Laboratory Research on White Rust and Passivation continued
Investigation into Passivation Evapco designed two small-scale closed circuit
There have been continuing complaints from Evapco coolers named Tiny Cooler 1 and Tiny Cooler 2,
customers about galvanized steel developing white rust shown in Figure 3. These units provided the first test
corrosion, resulting in shorter equipment life. These platform for side-by-side analysis to study the impact of
complaints are particularly prevalent on coil products immediate heat load on new galvanized coils.
that are placed under immediate load. It is well Figure 3: Evapco’s “Tiny Coolers”
understood that the initial corrosion layer (passivation
layer) is very important to equipment life, so a research
program was started to evaluate the parameters that are
required to develop a good passivation layer.
The ongoing research includes the use of scanning
electron microscope-energy dispersive x-ray analysis
(SEM-EDXA) to determine the elemental composition
of white rusted areas and metal surfaces adjacent
to the location of the corrosion product shown in
Figure 2. Ongoing research and laboratory testing
suggest the following aluminum content range for
hot-dipped galvanized steel: 0.069 to 0.149%.
Figure 2: Galvanized Coil Samples From Tiny Coolers
(SEM-EDXA)
Beginning in 2011, these side-by-side coolers provided
an opportunity to study the effectiveness of pH control,
orthophosphate-based treatments, and other variables
on the potential for, and quantity of, white rust
formation on new galvanized coils started with
immediate heat load. The closed-loop water supplied to
the inlet of the coil bundle was heated to approximately
100°F, with the systems operating five days per week for
approximately 8 to 10 hours per day. Glutaraldehyde
nonoxidizing biocide was manually dosed three times
per week during operation, and sodium hypochlorite was
manually dosed at the end of each week just prior to the
idle weekend period.
The earliest series of tests provided makeup water
The research conducted to date has not identified a “produced” by blending approximately 70% reverse
correlation between aluminum content either within osmosis (RO) permeate with approximately 30%
white rust deposits or on non-white-rusted galvanized Taneytown, Maryland, municipal water. Table 1 lists the
areas near the corrosion product. water composition. Conductivity controllers automated
It should be noted that Evapco’s current research is the blowdown of the spray water to maintain cycles of
focused on hot-dipped galvanized coils as opposed to the concentration in the 2.0 to 2.5 range. Water temperature
“Galvanized Steel Surfaces”1 detailed in Table 1 of the and pH data were tracked automatically with daily
1992 Analyst article, which likely were mill galvanized. calibration, as required, based on handheld readings.
In support of customer requests for better commissioning
outcomes, Evapco, Inc., initiated a study of the
formation and control of white rust. This research
focused on the formation of a passive layer capable
of minimizing the formation of white rust in galvanized
cooling equipment commissioned with immediate
heat load.
15 the Analyst Volume 24 Number 3
Beyond Laboratory Research on White Rust and Passivation continued
Table 1: Early Tiny Cooler Test-City and Blended In an effort to increase the pace of discovery, benchtop
Water Data tests were initiated and designed to simulate equipment
Taneytown Blended makeup started with heat load. Previous Analyst articles reference
Analyte Ave. Min. Max. Ave. Min. Max. the use of galvanized panels in conjunction with small-
pH 7.2 7.35 7.83 7.7 7.6 7.9 scale testing.6
Conductivity (mhos) 515 468 575 184 171 195
A test matrix was established to evaluate hot dip
Calcium (CaCO3) 152 119 178 45 36 50
galvanized test strips, mill galvanized test strips, and
Alkalinity 161 155 182 56 49 59
both varieties exposed to proprietary pretreatment
Chloride (mg/L) 47 155 182 17 15 19
processes. The test schedule incorporated six to eight
Sulfate (mg/L) 22 19 27 7 6 8
weeks of simulated passivation followed by a two to
Later testing reduced the percentage of RO permeate to three-week transition to an alkaline treatment program.
study the impact of using a variety of different acids to All test panels were submerged by day and allowed to air
trim alkalinity/pH. dry overnight.
For the earliest tests, the passivation phase of the Immersion solutions were “manufactured” by blending
trial lasted between four and six weeks with manual Taneytown city water and RO permeate to achieve
chemical feed and testing of inhibitor residuals, pH, and calcium hardness of 50 mg/L and alkalinity at 60 mg/L
conductivity twice per day. Every Wednesday, makeup (both as CaCO3). Solutions were heated to 90°F, aerated,
and recirculating water samples were collected for a and changed out daily during the test. pH ranged
complete laboratory analysis using atomic absorption between 7.2 and 7.8, reaching a maximum of 8.0 over
spectroscopy and ion chromatography. At the end of the course of a day.
the prescribed passivation period, a transition phase of Twenty-eight separate jars, shown in Figure 4, each
approximately two to three weeks was initiated to move containing two galvanized test strips (Figure 5), were
to an alkaline treatment program. utilized during the benchtop testing. Six distinct
The Tiny Cooler testing in 2011 and early 2012 inhibitor chemistries were tested across the 28 jars
suggested the following for new galvanized equipment during the initial screening.
commissioned with immediate heat load: Figure 4: Benchtop Testing Setup
Simply maintaining spray water pH below 8.3 during
the passivation phase does not preclude white rust
formation.
Commercially available orthophosphate based
treatments suggested to control white rust in
combination with pH control do not ensure a uniformly
passivated surface.
Applying factory pre-treatment to the galvanized coil
prior to shipment and startup can reduce the potential
for subsequent white rust formation.
Although is was informative, research using the Tiny
Coolers alone was time consuming. The original testing Figure 5: Hot-Dipped Galvanized Test Strip
took four to six weeks, but due to the lack of satisfactory
results, the passivation and transition times were
increased, resulting in a significantly longer test cycle.
Each test then took between 12 and 16 weeks, limiting
the speed of innovation. As research unfolded, more
hypotheses and variables were uncovered that were
worthy of additional study.
By 2012, AWT’s updated guide on the subject
maintained its earlier reference to a survey amongst their
membership, which found “white rust corrosion was
identified as a serious and prevalent problem.”2
16 the Analyst Volume 24 Number 3
Beyond Laboratory Research on White Rust and Passivation continued
The benchtop testing conducted between March and Figure 7: Evapco’s Nano Coolers
July 2012 demonstrated the following:
• Comparing jars with the same temperature, aeration,
water quality, inhibitor type, and concentrations,
the factory-applied pretreated test strips exhibited
less white rust potential than test strips that had not
received pretreatment, as shown in Figure 6.
• Benchtop galvanized strips tested with inhibitors
used in previous Tiny Cooler testing exhibited less
white rust formation compared to the Tiny Cooler
coil results.
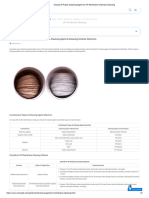
Figure 6: Test Strip With Factory Pretreatment (L)
Compared to Untreated Galvanized Strip (R)
In conjunction with the two Tiny Coolers, the addition
of the Nano Coolers provides an opportunity to
study more variables side by side. One example of the
increased flexibility available from the Nano Coolers
was a test that studied the impact of varying calcium,
chloride, and sulfate concentrations in the spray water on
the galvanized coil sections shown in Figure 8.
Figure 8: Elevated Chloride (Nano Cooler 2) vs.
Elevated Sulfate (Nano Cooler 3) Coil Sections
Galvanized test strips immersed in heated solutions do
not appear to correlate well with reactions that occur
when a heated coil is cooled by spray water. This suggests
that heating the liquid (immersion bath) does not create
the same increase in corrosion potential as heating the
coil metallurgy (increasing skin temperature).
Because of the poor correlation between heated solution
benchtested samples and heated coils from the Tiny
Coolers, the use of benchtesting was minimized to
enable refocusing on tests utilizing heated coil sections.
Following benchtop screening, additional research
equipment was built to evaluate white rust formation
on evaporative heat transfer surfaces. Four very small
coolers were designed and built to provide faster testing
of hypotheses and variables. Each new “Nano Cooler,”
shown in Figure 7, holds up to four small coil sections,
which are exposed to identical operating conditions
(e.g., temperature, water quality, inhibitor dosage).
18 the Analyst Volume 24 Number 3
Beyond Laboratory Research on White Rust and Passivation continued
This study demonstrated that elevated sulfate (540 mg/L) Proprietary passivation formulations and commercially
showed less potential to increase white rust compared to available treatment products have been tested. Testing to
elevated chloride (200 mg/L), as shown in Table 2. date suggests that vendors and their customers should be
Further, comparing two coil sections exposed to skeptical of products that claim to passivate equipment
elevated chloride (200 mg/l), the coil section exposed to starting with immediate heat load in as little as 24 hours,
spray water containing an average of 66 mg/L calcium 48 hours, one week, or two weeks. A commercially
developed more white rust versus one exposed to an available product advertised as a poly/orthophosphate
average of 195 mg/L calcium, as shown in Figure 9. inhibitor that will “prevent white rust” on galvanized
surfaces in as little as three or four days or up to two
Table 2: Water source weeks was tested in a Nano Cooler. Visually, the test
showed promise during the first three weeks of feeding
NC1 will use water from the blended MU tank without any additions
the product (see Figure 10). As the test transitioned to
MU Rec mg/L mg Per
Target Target
mg/L By Adding
in MU gallon
alkaline treatment with pHs above 8.5, however,
white rust quickly appeared, as shown in Figure 11.
CaCl2 (Cal-Plus)
NC2 33 100 Ca (Sol. 745 g/L @ CaCl2 70 265
A subsequent Nano Cooler test sequence suggests that
68 °F) more than four weeks may be required to sufficiently
Na2SO4, 99% passivate new galvanized surfaces before a successful
NC3 180 540 SO4 (Sol. 200 g/L @ Na2SO4 264 999 transition to an alkaline treatment program is possible.
68 °F)
With this insight, the test of a commercially available
NaCl
NC4 67 200 Cl (Sol. 360 g/L @ NaCl 76 288 product, shown in Figures 10 and 11, is scheduled to be
68 °F) repeated with a longer passivation timeframe.
Figure 9: Higher Calcium (Nano Cooler 2) vs. Lower Figure 10: Nano Cooler 1 Following Third Week of
Calcium (Nano Cooler 4) Coil Sections Commercial Product Testing
Figure 11: Nano Cooler 1 Coil Section After One Day of
Operation at pH 8.8
The impact of variables such as coil inlet temperature
(85–100 °F), duration of passivation (2–10 weeks),
transition from passivation to alkaline treatment
(0–3 weeks), passivation chemistry, pretreatment
techniques, wet operation (8–22 hours/day), and a variety
of makeup water characteristics continue to be explored
using both the Nano and Tiny Cooler systems.
19 the Analyst Volume 24 Number 3
Beyond Laboratory Research on White Rust and Passivation continued
The investigation continued with a goal of optimizing a
cost-effective, factory-applied pretreatment capable of
minimizing the formation of white rust in evaporative
cooling equipment commissioned with immediate heat
load. Several years of bench, Nano Cooler, and Tiny
Cooler testing have consistently shown that factory
pretreated galvanizing develops less white rust than
untreated hot-dip galvanization in side-by-side testing
under identical operating conditions, as shown in
Figures 5 and 11.
Significant investment in the construction and
ongoing operation of the small-scale test equipment
(see Figures 3 and 6) provides a platform to move
beyond laboratory research relying on
coupons or test strips. The ability to
operate controlled tests to evaluate the
impact of heat load, water chemistry,
inhibitor formulations, and factory-
applied pretreatments on galvanized
surfaces will help to provide the data
necessary for a better understanding
of the control parameters necessary for
successful passivation in the presence
of heat load. These insights will make it
easier to provide better commissioning
outcomes with reduced occurrence of
white rust, for customers and water
treatment professionals alike.
References WATER IS LIFE –
1. The Associated Labs. Technical Committee
(The Analyst, Spring 1992).“An Evaluation of
White Rust and Cooling Tower Metallurgy”
THAT’S WHY WE
2. Association of Water Technologies, White Rust: CARE
An Industry Update and Guide Paper 2002
3. Rachels, G. K. (The Analyst, Spring 1991). What components do you need to
“White Rust” (The Water Treaters’ Achilles Heel) boost your business success?
4. Johnson, K. M. and Mihelic, J. B. Cooling Tower
Institute Technical Paper Number TP-91-14. Visit Brenntag in Grand Rapids Source from the experts!
Update on White Rust Corrosion and Control AWT Convention and Expo
September 13–16, Booth #207 & 209 We understand chemistry
5. Kunz, R. G. and Hines, D. W. (NACE Corrosion and applications.
90 Paper 348). Corrosion of Zinc in Cooling
Water Experienced, trusted, regional
6. Busch, B. D. and Oldsburg, M. T. (The Analyst, technical water experts to
Summer 2000). “Advances in the Inhibition of answer specific questions.
White Rust Corrosion”
The largest global product portfolio.
Christopher Nagle is vice president of 176 stocking locations in
Water Systems at Evapco. He can be the US and Canada.
reached at (410) 756-2600 or
cnagle@evapco.com.
Contact us today
Phone: (800) 890-0355
wateradditives@brenntag.com
www.brenntagnorthamerica.com
21 the Analyst Volume 24 Number 3
6488_BNA_WaterAdditives_4.5x7inch_En.indd 1 06.04.17 13:56
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5796)
- 1600878255cooling Towers Legionellosis and Water Management Programs 2020 Awt1 Copy Pdf1600878255Document45 pages1600878255cooling Towers Legionellosis and Water Management Programs 2020 Awt1 Copy Pdf1600878255Max JunghannsNo ratings yet
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- 1601401358chris Nagle Paper Cooling Towers Legionellosis and Water Management Programs Awt pdf1601401358Document21 pages1601401358chris Nagle Paper Cooling Towers Legionellosis and Water Management Programs Awt pdf1601401358Max JunghannsNo ratings yet
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- A Breakthrough in Chlorine Dioxide Generation Without Precursors: Bluesense™ OxibluDocument9 pagesA Breakthrough in Chlorine Dioxide Generation Without Precursors: Bluesense™ OxibluMax JunghannsNo ratings yet
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (400)
- Chlorine Dioxide Generation Bluesense Oxiblu: A Breakthrough in Without PrecursorsDocument29 pagesChlorine Dioxide Generation Bluesense Oxiblu: A Breakthrough in Without PrecursorsMax JunghannsNo ratings yet
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- Choose A Proper Cleaning Agent For UF Membrane Chemical CleaningDocument2 pagesChoose A Proper Cleaning Agent For UF Membrane Chemical CleaningMax JunghannsNo ratings yet
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Evaporative Condenser Passivation: Cameron Klein Strand Associates, IncDocument27 pagesEvaporative Condenser Passivation: Cameron Klein Strand Associates, IncMax JunghannsNo ratings yet
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- AWT Corrosion Coupon Guidance Form07142019Document1 pageAWT Corrosion Coupon Guidance Form07142019Max JunghannsNo ratings yet
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- C3121810 Dra STDocument1 pageC3121810 Dra STMax JunghannsNo ratings yet
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (589)
- PMC E Bulletin 106D Final 9.2018 PDFDocument24 pagesPMC E Bulletin 106D Final 9.2018 PDFMax JunghannsNo ratings yet
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- ATC E Bulletin 153E Final LR PDFDocument44 pagesATC E Bulletin 153E Final LR PDFMax JunghannsNo ratings yet
- CaF2 Pretorius PDFDocument21 pagesCaF2 Pretorius PDFAlexis YañezNo ratings yet
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- Dezurik Eccentric Plug Valves Pec Pef 12 3 Pec Eccentric Plug Valves Technical 12-00-1bDocument12 pagesDezurik Eccentric Plug Valves Pec Pef 12 3 Pec Eccentric Plug Valves Technical 12-00-1bOleg ShkolnikNo ratings yet
- Turck SenzoriDocument791 pagesTurck Senzorimb1314No ratings yet
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1091)
- Cathodic Protection SystemDocument27 pagesCathodic Protection SystemSandeep GuptaNo ratings yet
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- Hollow Structural Sections Hss Steel TubeDocument15 pagesHollow Structural Sections Hss Steel TubeCesar BarraganNo ratings yet
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- Process of Press Tool Design and Its Manufacturing For Blanking OperationDocument8 pagesProcess of Press Tool Design and Its Manufacturing For Blanking OperationamdevaNo ratings yet
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (345)
- DDocument36 pagesDFuad EngineNo ratings yet
- SheetCam TNG Manual - A4Document159 pagesSheetCam TNG Manual - A4Fabio Fernando FigaroNo ratings yet
- Maxi KoolDocument17 pagesMaxi KoolEric E' Sandoval ANo ratings yet
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- Surface Protective Systems: Oxydur K 425Document2 pagesSurface Protective Systems: Oxydur K 425SHAIK ASIMUDDINNo ratings yet
- Here's How ItDocument12 pagesHere's How ItKongu RameshNo ratings yet
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- 20 Kitchen Tools and Its UsesDocument4 pages20 Kitchen Tools and Its UsesJay Rondina92% (13)
- Types of Heat Treating Equipment For FastenersDocument4 pagesTypes of Heat Treating Equipment For FastenersevalenciaNo ratings yet
- NFEM Ch02Document17 pagesNFEM Ch02SandipNo ratings yet
- Corrosion Principles: Engr. Dr. AD Chandio Associate Professor Myd & Mmy Ned Uet, KhiDocument52 pagesCorrosion Principles: Engr. Dr. AD Chandio Associate Professor Myd & Mmy Ned Uet, KhialiNo ratings yet
- Tank Cleaning MachineDocument9 pagesTank Cleaning Machinevidyadhar G100% (2)
- KSB Omega PumpsDocument68 pagesKSB Omega PumpsNataliaBostan67% (3)
- Time and Temperature Effects On Tensile, Bond, and Compressive StrengthsDocument5 pagesTime and Temperature Effects On Tensile, Bond, and Compressive StrengthsEman SalehNo ratings yet
- DrillingDocument27 pagesDrillingTapas BanerjeeNo ratings yet
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (122)
- Ercom Brief ProfileDocument57 pagesErcom Brief ProfileKarthik DhayalanNo ratings yet
- MESSER Gase For Life On-Site - Gas - Production PDFDocument12 pagesMESSER Gase For Life On-Site - Gas - Production PDFnefoussiNo ratings yet
- Watchdog Installation Guide PDFDocument34 pagesWatchdog Installation Guide PDFbulentbulutNo ratings yet
- Effect of Water On Quality and Preservation of FoodDocument10 pagesEffect of Water On Quality and Preservation of FoodrupinisinnanNo ratings yet
- Magnetic Flux LeakageDocument9 pagesMagnetic Flux LeakageCheeragNo ratings yet
- 08 SeismicDesignOfStructuresWithViscousDampers - DesbloqueadoDocument15 pages08 SeismicDesignOfStructuresWithViscousDampers - DesbloqueadoFrancisco Héctor BañuelosNo ratings yet
- Bert Rowe's-Mercedes-Benz 'A'-Class Info. Starter Motor Location & InformationDocument29 pagesBert Rowe's-Mercedes-Benz 'A'-Class Info. Starter Motor Location & Informationeliah100% (1)
- As TronicDocument56 pagesAs Tronicabeid khamis100% (1)
- Maintenance Interval Schedule GENSET 250KVADocument2 pagesMaintenance Interval Schedule GENSET 250KVARegi HermawanNo ratings yet
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- Dispersion EsDocument49 pagesDispersion EsJose Fernando Solanilla DuqueNo ratings yet
- ABS & Royalex Canoe Maintenance GuideDocument1 pageABS & Royalex Canoe Maintenance GuideSwift Canoe And KayakNo ratings yet