Professional Documents
Culture Documents
Cellular Response To MTA PDF
Cellular Response To MTA PDF
Uploaded by
Hazim rizkOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Cellular Response To MTA PDF
Cellular Response To MTA PDF
Uploaded by
Hazim rizkCopyright:
Available Formats
Clinical Practice
Cellular Response to Mineral Trioxide Aggregate
Root-End Filling Materials
Contact Author
Hiran Perinpanayagam, DDS, PhD Dr. Perinpanayagam
Email: hiran.
perinpanayagam@
schulich.uwo.ca
ABSTRACT
Endodontic surgical procedures involve the use of a root-end filling material to pro-
vide an apical seal and to facilitate the repair and regeneration of periradicular tissues.
Whereas earlier studies typically evaluated the cytotoxicity of these materials, contem-
porary research has focused on their capacity to support a favourable cellular response.
In vitro tissue culture techniques have demonstrated interactions at the cell surface that
may be conducive to periapical healing. Recent research with osteoblasts has confirmed
their affinity for novel endodontic mineral trioxide aggregate (MTA) materials, with evi-
dence of cell attachment and synthesis of bone matrix. This article reviews the current
state of knowledge about MTA as a root-end filling material, with particular emphasis
on cellular response to MTA materials.
For citation purposes, the electronic version is the definitive version of this article: www.cda-adc.ca/jcda/vol-75/issue-5/369.html
T
raditional efforts to repair tooth roots and biocompatible with the periradicular tis-
used materials that were originally de- sues. The ability to promote periapical healing
signed for the restoration of tooth could be particularly beneficial.
crowns, but these materials were barely toler- Periapical healing involves the repair and
ated by the surrounding ligament and bone. regeneration of alveolar bone and the peri-
Recent studies have indicated that mineral odontal ligament.6 Bone regeneration depends
trioxide aggregates (MTAs) are highly effective on differentiation of osteoblasts and synthesis
for filling root ends, repairing root perfora- and mineralization of extracellular bone
tions1 and restoring damaged root structure. matrix. However, little is known about the
Under physiological conditions, MTAs set effects of root-end filling materials on these
gradually (over a period of several hours) processes in alveolar bone. Therefore, con-
through hydration and subsequent inter- temporary studies have begun to examine the
locking of crystals within the tissue fluid. The influence of the root-end filling materials on
hardened material has little cytotoxicity and osteoblasts and other cells in the periradicular
good biocompatibility and appears to induce a tissues.
favourable tissue response.2-4
The purpose of these retrograde fillings is Limitations of Research and Study
to create an apical seal or protective barrier Design to Date
between the contents of the root canal system The ideal study to examine the effects of
and the periradicular tissues in the alveolar endodontic root-end filling materials on the
bone (Fig. 1).5 Therefore, the materials must periradicular tissues would be a clinical trial
possess physical properties conducive to cre- involving humans. However, clinical trials on
ating an apical seal, and they must be nontoxic patients requiring endodontic treatment are
JCDA • www.cda-adc.ca/jcda • June 2009, Vol. 75, No. 5 • 369
––– Perinpanayagam–––
They found that the cells were in con-
tact with the MTA after 1 and 3 days,
but remained small and rounded on
IRM (i.e., not in contact with the sub-
strate). Zhu and colleagues13 also used
scanning electron microscopy to look
at the effects of amalgam, IRM, resin
and MTA on Saos-2 osteosarcoma
cells. They found that the cells attached
to and spread very well over the resin
material, forming a monolayer within
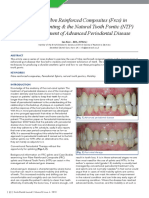
Figure 1: Surgical endodontics and root-end fillings. This patient had traumatic
crown fractures of the maxillary incisors, which were treated with root canal therapy, 24 hours. Similarly, they had good at-
prefabricated posts, core build-ups and crowns. Unsatisfied with the esthetic results, tachment and spreading on MTA.
he sought retreatment several years later. Specialists in endodontics, periodontics In contrast, most of the cells applied
and prosthodontics performed nonsurgical retreatment of the root canals, surgical on amalgam or IRM were small and
lengthening of the crowns, and placement of cast post, cores and crowns. Despite
the nonsurgical retreatment of the root canal of tooth 22, a periradicular radiolu- rounded, with little evidence of attach-
cency persisted (a). Endodontic surgery revealed a radicular cyst (b). Root-end resec- ment and spreading.
tion and filling were followed by periapical healing and near-complete resolution of The authors of these studies used
the lesion (c). carefully controlled in vitro condi-
tions to examine the microscopic
interactions between cells and root-
end filling materials. However, their
limited by obvious ethical constraints. For example, pa- choice of osteosarcoma cell lines is a cause for concern.
tients should not be exposed to potentially hazardous Although osteosarcoma cells resemble osteoblasts in
materials, should not receive suboptimal treatment and some respects, their underlying biological regula-
should not be left untreated as experimental controls. tory mechanisms may differ fundamentally from those
Furthermore, the information that could be obtained of primary osteoblasts. For example, when Perez and
from such a clinical trial would be limited unless the sur- colleagues14 compared the responses of MG-63 osteo-
gical site was periodically resampled. sarcoma cells and primary rat calvarial osteoblasts to
Given these constraints on clinical studies, several MTAs, there were noticeable differences. Whereas the
investigators have used animal models to study the effects osteosarcoma cells readily made contact with both
of endodontic materials on tissues.4 These animal studies ProRoot MTA (Dentsply Tusla Dental, Tulsa, Okla.) and
have provided valuable information, but extrapolation of white (tooth-coloured) MTA (Dentsply Tulsa Dental), the
their findings to the human condition should be under- primary rat osteoblasts could not be maintained on the
taken with caution. Accordingly, such studies have been white MTA for the study period of 13 days. Furthermore,
supplemented by in vitro tissue culture analyses. In vitro the primary rat cells formed mineral nodules and ex-
studies offer a more rigorous and carefully controlled pressed an osteogenic phenotype, whereas the osteosar-
environment in which to investigate the underlying cel- coma cell cultures failed to mineralize. Accordingly, these
lular and molecular mechanisms of tissue responses to investigators concluded that primary cell cultures may be
endodontic materials. a more sensitive and appropriate model for studying cel-
lular interactions with endodontic materials.
Tissue Culture Studies These 3 studies with osteosarcoma cells12–14 and the
Most tissue culture studies have focused on the cyto- single study that used primary cells from the calvaria of
toxicity of endodontic materials, particularly in the freshly rats14 were limited to microscopic examinations for cell
mixed state, before setting.7–10 When freshly mixed, these contact and a few other analyses of limited scope. In con-
materials release a host of chemical by-products that are trast, a more recent study used more clinically relevant
cytotoxic to the cells in culture. However, under clinical cells and undertook more complex and rigorous analyses
(in vivo) conditions, these by-products are diluted in the to determine the specific effects of root-end filling ma-
interstitial tissue fluids and are eliminated through the terials on cells in the periradicular tissues.11 Bonson and
vasculature. Therefore, recent studies have examined the colleagues11 studied the effects of amalgam (Tytin, Kerr
effect of preset, washed materials on cells in culture.11 Corporation, Orange, Calif.), MTA (ProRoot, Dentsply
In one of these studies, Koh and colleagues12 used Tusla Dental), zinc-oxide eugenol cement (SuperEBA,
scanning electron microscopy to examine the effects of Bosworth Corporation, Chicago, Ill.) and hybrid ionomer
2 root-end filling materials, MTA and intermediate re- composite resin (Geristore, DenMat, Santa Monica,
storative material (IRM), on MG-63 osteosarcoma cells. Calif.) on clinically derived human gingival fibroblasts
370 JCDA • www.cda-adc.ca/jcda • June 2009, Vol. 75, No. 5 •
––– Cellular Response to MTA –––
lying MTA surface and with adjacent cells (Fig. 2). These
cellular interactions with the surface of MTA appeared
to be at least as extensive as those seen when cells were
grown on serum-coated plastic surfaces. Similarly, other
researchers have recently found that human periodontal
ligament fibroblasts attached to MTAs.11,17 Bonson and
colleagues11 found that clinically derived human gingival
fibroblasts and periodontal ligament fibroblasts survived
and proliferated in the presence of MTA and made direct
contact with MTA particles. Balto17 found that human
periodontal ligament fibroblasts attached to MTA within
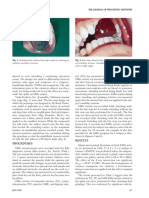
Figure 2: Human bone cell interactions with mineral trioxide
4 hours and then spread out over the surface during the
aggregates (MTAs). Human alveolar bone cells were grown
on tooth-coloured MTA (Dentsply Tulsa Dental) over 24 hours subsequent 20 hours. Likewise, earlier studies12,13,18 with
in vitro. Scanning electron microscopy showed that the cells osteosarcoma cells showed that they readily attached to
attached to and spread onto the material. Numerous MTA. One of the studies12 found that MG-63 osteosar-
cellular projections were intimately associated with the MTA
coma cells were in contact with MTA within 24 hours.
surface.
Cells from another osteosarcoma cell line, Saos-2, spread
out to form a monolayer on MTA within 24 hours.13
Saos-2 cells attached within 12 hours and then prolif-
and periodontal ligament fibroblasts. As expected, the erated over MTA-coated cover slips during a 72-hour
freshly mixed materials were cytotoxic, but this effect was period.18 Additionally, Thomson and colleagues19 found
reduced by washing. Through molecular techniques in- that an immortalized mouse cementoblast cell line at-
volving reverse transcription polymerase chain reaction, tached to and spread out over the MTA surface in a
these authors were able to observe the effects of these fashion similar to that observed on glass cover slips.
In our research, cell attachment to and propagation on
materials on the cellular expression of genes. There were
ProRoot (grey) MTA were similar to what was observed
some differences in the responses of the gingival and
on tooth-coloured (white) MTA.16 Scanning electron mi-
periodontal ligament fibroblasts to each material, as well
croscopy showed that comparable numbers of alveolar
as differences in their responses to different materials.
bone cells attached to the grey and to the white MTA
For example, one of the materials appeared to promote
surfaces and that elaboration of cellular extensions and
an osteogenic phenotype, whereas another had the op-
processes on the 2 surfaces was similar. Furthermore, in
posite effect.
extended culture the cells remained attached to the white
In addition to the periodontal ligament fibroblasts,
MTA surface and formed an extensive matrix-like layer
cells from the surrounding alveolar bone15 probably play
that appeared similar to that observed on grey MTA. This
an important role in repair and regeneration at the endo-
cellular matrix remained intimately associated with the
dontic surgical site. Accordingly, we have examined the
white MTA surface for the full 2-week duration of this
effects of root-end filling materials on primary cell cul- experiment. In contrast, in an earlier study, primary rat
tures derived from human alveolar bone, as well as mouse osteoblasts did not remain attached to the white MTA
pre-osteoblasts that are capable of differentiating into in extended culture.14 In that study, fetal rat calvarial
mature cells that synthesize bone matrix. These studies osteoblasts and MG-63 osteosarcoma cells were grown
will help to ensure that the choice of root-end filling in the presence of grey and white MTA particles for 6,
materials is based on a thorough understanding of their 9 and 13 days. Both the primary rat osteoblasts and the
effects on the repair and regeneration of periradicular osteosarcoma cells proliferated and attached to the grey
tissues. This research will also aid in the development of and to the white MTA particles after 6 and 9 days of in-
novel endodontic materials that can actively promote the cubation. After 13 days, the osteosarcoma cells remained
healing process. attached to the grey and to the white MTA particles, but
the primary rat cells were seen only on the grey MTA.
Cellular Response to MTA Materials The failure of the primary rat cells to remain attached
In our research, we found that human alveolar bone to the white MTA for 13 days was a cause for concern.
cells15 attached and spread out on the surface of ProRoot Therefore, demonstration of sustained attachment and
MTA (Dentsply Tulsa Dental) (Fig. 2).16 In particular, propagation of human cells on white MTA in our study
when the cells were seeded onto MTA disks, they at- was a valuable finding. It reassures us that both the grey
tached and spread over the surface within 24 hours. and the white formulations of MTA are capable of sup-
Scanning electron microscopy revealed cellular exten- porting the cell attachment and proliferation that cul-
sions and processes in intimate contact with the under- minate in the synthesis of a matrix-like layer.
JCDA • www.cda-adc.ca/jcda • June 2009, Vol. 75, No. 5 • 371
––– Perinpanayagam–––
Molecular Analysis of the Cellular Response Correspondence to: Dr. Hiran Perinpanayagam, Schulich School of
Medicine & Dentistry, The University of Western Ontario, Dental Sciences
In addition to this research on interactions at the cell Building 0079, London, ON N6A 5C1
surface, several studies have examined the molecular re-
The author has no declared financial interests in any company manufac-
sponses of osteoblasts to MTA materials in vitro.11,20,21 In turing the types of products mentioned in this article.
their comprehensive 2004 study, Bonson and colleagues11
This article has been peer reviewed.
used human gingival fibroblasts and periodontal ligament
fibroblasts, the latter of which possess some osteogenic
potential. The periodontal ligament fibroblasts that were References
1. Roberts HW, Toth JM, Berzins DW, Charlton DG. Mineral trioxide aggre-
exposed to MTA expressed osteoblast-associated pro- gate material use in endodontic treatment: a review of the literature. Dent
teins such as alkaline phosphatase, bone sialoprotein and Mater 2008; 24(2):149–64.
periostin. Similarly, Nakayama and colleagues20 found 2. Torabinejad M, Hong CU, Lee SJ, Monsef M, Pitt Ford TR. Investigation
of mineral trioxide aggregate for root-end filling in dogs. J Endod 1995;
expression of alkaline phosphatase and osteopontin by 21(12):603–8.
rat femoral bone marrow cells exposed to MTA. However, 3. Torabinejad M, Hong CU, Pitt Ford TR, Kettering JD. Cytotoxicity of four
root end filling materials. J Endod 1995; 21(10):489–92.
their impression was that MTA had an inhibitory ef-
4. Torabinejad M, Pitt Ford TR. Root end filling materials: a review. Endod
fect on osteoblast differentiation. More recently, Tani- Dent Traumatol 1996; 12(4):161–78.
Ishii and colleagues21 detected the expression of bone 5. Gartner AH, Dorn SO. Advances in endodontic surgery. Dent Clin North
sialoprotein and osteocalcin from mouse MC3T3-E1 pre- Am 1992; 36(2):357–78.
6. Fouad AF, Walton RE, Rittman BR. Healing of induced periapical lesions in
osteoblasts exposed to MTA. ferret canines. J Endod 1993; 19(3):123–9.
Likewise, we found that human alveolar bone cells 7. Zhu Q, Safavi KE, Spangberg LS. Cytotoxic evaluation of root-end filling
grown on MTA disks in vitro expressed type I collagen materials in cultures of human osteoblast-like cells and periodontal ligament
cells. J Endod 1999; 25(6):410–2.
and synthesized an extensive collagenous matrix during
8. Osorio RM, Hefti A, Vertucci FJ, Shawley AL. Cytotoxicity of endodontic
2 weeks of growth. 22 Concomitantly, the osteoblast- materials. J Endod 1998; 24(2):91–6.
specific transcription factor Runx2 was expressed within 9. Chong BS, Owadally ID, Pitt Ford TR, Wilson RF. Cytotoxicity of potential
a week of culture on MTA. Furthermore, this pattern retrograde root-filling materials. Endod Dent Traumatol 1994; 10(3):129–33.
10. Bruce GR, McDonald NJ, Sydiskis RJ. Cytotoxicity of retrofill materials.
of gene expression was largely unaltered by interactions J Endod 1993; 19(6):288–92.
with alternative formulations of MTA. 11. Bonson S, Jeansonne BG, Lallier TE. Root-end filling materials alter fibro-
blast differentiation. J Dent Res 2004; 83(5):408–13.
Summary of Research and Future Directions 12. Koh ET, McDonald F, Pitt Ford TR, Torabinejad M. Cellular response to
mineral trioxide aggregate. J Endod 1998; 24(8):543–7.
Contemporary researchers have shifted the direction
13. Zhu Q, Haglund R, Safavi KE, Spangberg LS. Adhesion of human osteo-
of investigations on endodontic materials, from basic blasts on root-end filling materials. J Endod 2000; 26(7):404–6.
measurements of cytotoxicity to more complex analyses 14. Perez AL, Spears R, Gutmann JL, Opperman LA. Osteoblasts and MG-63
of tissue response. These studies have demonstrated fa- osteosarcoma cells behave differently when in contact with ProRoot MTA
and White MTA. Int Endod J 2003; 36(8):564–70.
vourable and extensive cell-surface interactions with 15. Perinpanayagam H, Martin T, Mithal V, Dahman M, Marzec N, Lampasso J,
MTA materials, including cell attachment, cell prolifera- and other. Alveolar bone osteoblast differentiation and Runx2/Cbfa1 expres-
sion. Arch Oral Biol 2006; 51(5):406–15.
tion and gene expression. The recent utilization of sur-
16. Al-Rabeah E, Perinpanayagam H, MacFarland D. Human alveolar
gically derived human cells has extended these findings bone cells interact with ProRoot and tooth-colored MTA. J Endod 2006;
with clinically relevant material. Ongoing research into 32(9):872–5.
the molecular events involved in cellular responses to 17. Balto HA. Attachment and morphological behavior of human periodontal
ligament fibroblasts to mineral trioxide aggregate: a scanning electron
these materials are expected to unravel the underlying microscope study. J Endod 2004; 30(1):25–9.
mechanisms and will facilitate the design and synthesis 18. Abdullah D, Ford TR, Papaioannou S, Nicholson J, McDonald F. An evalu-
of novel biomaterials that fully exploit these exemplary ation of accelerated Portland cement as a restorative material. Biomaterials
2002; 23(19):4001–10.
properties. a 19. Thomson TS, Berry JE, Somerman MJ, Kirkwood KL. Cementoblasts
maintain expression of osteocalcin in the presence of mineral trioxide ag-
gregate. J Endod 2003; 29(6):407–12.
20. Nakayama A, Ogiso B, Tanabe N, Takeichi O, Matsuzaka K, Inoue T.
THE AUTHOR Behaviour of bone marrow osteoblast-like cells on mineral trioxide ag-
gregate: morphology and expression of type I collagen and bone-related
protein mRNAs. Int Endod J 2005; 38(4):203–10.
21. Tani-Ishii N, Hamada N, Watanabe K, Tujimoto Y, Teranaka T, Umemoto T.
Dr. Perinpanayagam is associate professor in dentistry, Expression of bone extracellular matrix proteins on osteoblast cells in the
Schulich School of Medicine & Dentistry, The University of presence of mineral trioxide. J Endod 2007; 33(7):836–9.
Western Ontario, London, Ontario. 22. Perinpanayagam H, Al-Rabeah E. Osteoblasts interact with MTA surfaces
and express Runx2. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2009;
107(4):590–6.
Acknowledgements: Dr. Perinpanayagam’s research has been supported
by an Internal Research Grant from Schulich Dentistry, a New Research
and Scholarly Initiatives Award from the University of Western Ontario,
and 2 research grants from the American Association of Endodontists
Foundation.
372 JCDA • www.cda-adc.ca/jcda • June 2009, Vol. 75, No. 5 •
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5819)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1092)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (845)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (590)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (897)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (540)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (348)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (822)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (122)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (401)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- GLOC Health Claim Form Fillable Form NewDocument2 pagesGLOC Health Claim Form Fillable Form NewRyanRamroopNo ratings yet
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- Smile Dent J 2013 8 4 32 36Document5 pagesSmile Dent J 2013 8 4 32 36nakhitasyafiraNo ratings yet
- ITI Decision Trees and ChecklistsDocument6 pagesITI Decision Trees and ChecklistsNay AungNo ratings yet
- User Guide - ACMDocument9 pagesUser Guide - ACMsaleh ahmedNo ratings yet
- FELINE-Dental Radio Graphic Technique in CatsDocument7 pagesFELINE-Dental Radio Graphic Technique in Catstaner_soysurenNo ratings yet
- Ron Greenspan DDS - Braces 101Document10 pagesRon Greenspan DDS - Braces 101Ron Greenspan DDSNo ratings yet
- Research Essay Final DraftDocument9 pagesResearch Essay Final Draftapi-670223407No ratings yet
- MM - Ejgd 360 BlindDocument32 pagesMM - Ejgd 360 BlindMuhammad IhsanNo ratings yet
- Intro To Pulp Space Morphology 2022Document24 pagesIntro To Pulp Space Morphology 2022محمد ربيعيNo ratings yet
- PN 1Document1 pagePN 1Florin Eugen ConstantinescuNo ratings yet
- Lynal: Tissue Conditioner & Temporary RelinerDocument2 pagesLynal: Tissue Conditioner & Temporary RelinerGonzalo Urrutia ElsoNo ratings yet
- Autorotation of MandibleDocument7 pagesAutorotation of Mandibleخالد الجدياويNo ratings yet
- Mapeh Grade 6 Active Body: Quarter 1 Week 4 Module 1Document39 pagesMapeh Grade 6 Active Body: Quarter 1 Week 4 Module 1reena joyceNo ratings yet
- Ceramic Dental Biomaterials and CAD/CAM Technology: State of The ArtDocument10 pagesCeramic Dental Biomaterials and CAD/CAM Technology: State of The ArtMaria Fernanda Romero RinconNo ratings yet
- Impression Techniques: For Implant ProsthesesDocument38 pagesImpression Techniques: For Implant Prosthesesbaqer hussain0% (1)
- DentalUM Spring/Summer 2008Document87 pagesDentalUM Spring/Summer 2008umdentNo ratings yet
- Immediate Vs Delayed Implant PlacementDocument60 pagesImmediate Vs Delayed Implant PlacementYashica KalyaniNo ratings yet
- Martha Alemayehus Resume LeadersipDocument3 pagesMartha Alemayehus Resume Leadersipapi-316750582No ratings yet
- 10 .Principles of Tooth PreparationDocument72 pages10 .Principles of Tooth PreparationDr Tejashree CNo ratings yet
- CPE Candidate Handbook PDFDocument43 pagesCPE Candidate Handbook PDFVirendra Singh Rathore RathoreNo ratings yet
- Personal Identification l1Document6 pagesPersonal Identification l1Apple AsneNo ratings yet
- Dentistry: A Case of Drug - Induced Xerostomia and A Literature Review of The Management OptionsDocument4 pagesDentistry: A Case of Drug - Induced Xerostomia and A Literature Review of The Management OptionsSasa AprilaNo ratings yet
- Dental Assistants in OrthodonticsDocument2 pagesDental Assistants in Orthodonticsmaryam fatimaNo ratings yet
- Biokompatibilitas PhilipDocument23 pagesBiokompatibilitas PhilipKrisna MertaNo ratings yet
- An Esthetic Removable Inclined Plane - Jco - Pearls - Volume IV Number 5 May 2020 Page 275Document2 pagesAn Esthetic Removable Inclined Plane - Jco - Pearls - Volume IV Number 5 May 2020 Page 275APinhaoFerreiraNo ratings yet
- Becker1999 2Document1 pageBecker1999 2Zhiao LiuNo ratings yet
- Rubber DamDocument78 pagesRubber DamDevanshi Sharma100% (1)
- Stunting Dan CariiesDocument5 pagesStunting Dan CariiessitoresmiNo ratings yet
- Repair Versus Replacement of RestorationDocument77 pagesRepair Versus Replacement of Restorationmrkhalid.sobhy67100% (3)
- 313 Classification of Periodontal and Peri-Implant Diseases and Conditions 2020 - Part IIDocument52 pages313 Classification of Periodontal and Peri-Implant Diseases and Conditions 2020 - Part IIAbdulrahman100% (1)