Professional Documents
Culture Documents
Certificate Haccp 2
Uploaded by
Malaika Shaikh0 ratings0% found this document useful (0 votes)
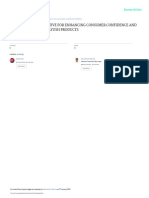
17 views1 pageThis certificate certifies that DrG WELLNESS PVT. LTD.'s Food Safety Management System has been independently assessed and found to be compliant with Hazard Analysis and Critical Control Points (HACCP) requirements for the import, export, manufacturing, and supply of ayurvedic medicines, nutraceuticals, health/dietary supplements, and proprietary food products from its registered office in Delhi and manufacturing unit in Bawana Industrial Complex, Delhi. The certificate is valid until September 24, 2021.
Original Description:
Original Title
certificate-haccp-2
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis certificate certifies that DrG WELLNESS PVT. LTD.'s Food Safety Management System has been independently assessed and found to be compliant with Hazard Analysis and Critical Control Points (HACCP) requirements for the import, export, manufacturing, and supply of ayurvedic medicines, nutraceuticals, health/dietary supplements, and proprietary food products from its registered office in Delhi and manufacturing unit in Bawana Industrial Complex, Delhi. The certificate is valid until September 24, 2021.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
17 views1 pageCertificate Haccp 2
Uploaded by
Malaika ShaikhThis certificate certifies that DrG WELLNESS PVT. LTD.'s Food Safety Management System has been independently assessed and found to be compliant with Hazard Analysis and Critical Control Points (HACCP) requirements for the import, export, manufacturing, and supply of ayurvedic medicines, nutraceuticals, health/dietary supplements, and proprietary food products from its registered office in Delhi and manufacturing unit in Bawana Industrial Complex, Delhi. The certificate is valid until September 24, 2021.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 1
This is to Certify that the
Food Safety Management System of
DrG WELLNESS PVT. LTD.
REGD. OFFICE: 100/28, SURAJMAL MARKET, RAZAPUR,
SECTOR - 09, ROHINI, DELHI-110085, INDIA
MFG. UNIT: C-44, SECTOR - 2, BAWANA INDUSTRIAL COMPLEX,
BAWANA, DELHI-110039, INDIA
has been independently assessed and is compliant with the requirements of:
HACCP
(Hazard Analysis & Critical Control Points)
This certificate is applicable to the following scope of operations:
IMPORT - EXPORT, MANUFACTURING & SUPPLY OF AYURVEDIC
MEDICINES, NUTRACEUTICALS, HEALTH / DIETARY SUPPLEMENTS &
PROPRIETARY FOOD PRODUCTS
Certificate No.: SPC18H2130 Date of Initial Registration: 24-09-2018
1st Surveillance audit on or before: 23-09-2019
2nd Surveillance audit on or before: 23-09-2020
Date of Re-Certification: 24-09-2021
You might also like
- Healing Cancer Vol. 1 IIDocument36 pagesHealing Cancer Vol. 1 IIMalaika ShaikhNo ratings yet
- Food Control System Assessment Tool: Dimension B – Control FunctionsFrom EverandFood Control System Assessment Tool: Dimension B – Control FunctionsNo ratings yet
- No. True/False: FSSC QuestionnaireDocument3 pagesNo. True/False: FSSC Questionnairechan5264100% (2)
- Certificate of Registration: Quality Management SystemDocument1 pageCertificate of Registration: Quality Management Systemrohan rohanNo ratings yet
- Pcod Homeo TherapeuticsDocument21 pagesPcod Homeo TherapeuticsMalaika ShaikhNo ratings yet
- BRC Issue 8 Whitepaper WebDocument12 pagesBRC Issue 8 Whitepaper WebGaganpreet KaurNo ratings yet
- Audit Report: Prime Xynergies Food CorporationDocument35 pagesAudit Report: Prime Xynergies Food CorporationPhuong Nguyen100% (1)
- BRC Cetificate 2017-KarjanDocument1 pageBRC Cetificate 2017-KarjanSachin KothariNo ratings yet
- 2012 HACCP Accreditation Program RequirementsDocument32 pages2012 HACCP Accreditation Program Requirementssadbad6No ratings yet
- China - Food and Agricultural Import Regulations and Standards Report - Export Certificate - 2!26!2019Document15 pagesChina - Food and Agricultural Import Regulations and Standards Report - Export Certificate - 2!26!2019Annisa Rahma FatmalaNo ratings yet
- FAM814 Module 14 Meeting Costco Requirements v3 24.10.2022Document9 pagesFAM814 Module 14 Meeting Costco Requirements v3 24.10.2022Lutviana Ema100% (1)
- GMP in Pharmaceutical Industry: Global cGMP & Regulatory ExpectationsFrom EverandGMP in Pharmaceutical Industry: Global cGMP & Regulatory ExpectationsRating: 5 out of 5 stars5/5 (2)
- BRC Food Safety Quality Management System Training GuideDocument61 pagesBRC Food Safety Quality Management System Training GuideFederico Leon100% (1)
- FSSC 22000 V5Document1 pageFSSC 22000 V5SupriyaNo ratings yet
- M.Sc. Foods and Nutrition: Semester I Max. MarksDocument21 pagesM.Sc. Foods and Nutrition: Semester I Max. MarksMalaika ShaikhNo ratings yet
- Malaysian Certification Scheme For HACCPDocument25 pagesMalaysian Certification Scheme For HACCPneli00100% (2)
- BRC - 2019-Shuaiba Industrial Company (Jafza)Document1 pageBRC - 2019-Shuaiba Industrial Company (Jafza)VincentNo ratings yet
- TSN 282862 enDocument1 pageTSN 282862 enarabelle.valeskaNo ratings yet
- Certificate of Registration: DR HealthtechDocument2 pagesCertificate of Registration: DR HealthtechDR HealthtechNo ratings yet
- Talc PowderDocument1 pageTalc Powdereurop pharmNo ratings yet
- Certificate of Analysis, Quality and Conformity: Certified: ISO 9001:2008, ISO 13485-2003 and WHO GMPDocument2 pagesCertificate of Analysis, Quality and Conformity: Certified: ISO 9001:2008, ISO 13485-2003 and WHO GMPMitha AriantiNo ratings yet
- 54.procedure For Workers Health ControlDocument3 pages54.procedure For Workers Health Controldileepkumar.gNo ratings yet
- HACCP Guidelines From APEDADocument20 pagesHACCP Guidelines From APEDASuresh KumarNo ratings yet
- Certificate of Compliance: Who-GmpDocument1 pageCertificate of Compliance: Who-GmpKSBNo ratings yet
- CXG 060eDocument4 pagesCXG 060eMonching AdecerNo ratings yet
- Lafiya Biotech Who-GmpDocument1 pageLafiya Biotech Who-GmpRakesh RockzzNo ratings yet
- Certificate of Registration: AB AZUCARERA IBERIA, S.L. Sociedad UnipersonalDocument1 pageCertificate of Registration: AB AZUCARERA IBERIA, S.L. Sociedad UnipersonalDomingoNo ratings yet
- IndiaHACCP Certification Criteria Oct 2017Document13 pagesIndiaHACCP Certification Criteria Oct 2017ANKUSHNo ratings yet
- Principles For Traceability Product Tracing As A Tool Within A Food Inspection and Certification SystemDocument2 pagesPrinciples For Traceability Product Tracing As A Tool Within A Food Inspection and Certification Systemcurious_leneNo ratings yet
- Haccp Form RenewDocument1 pageHaccp Form RenewHarits As SiddiqNo ratings yet
- Certificate of RegistrationDocument1 pageCertificate of RegistrationJavita CertificationsNo ratings yet
- Bgba HimediaDocument2 pagesBgba HimediaadiazcalidadNo ratings yet
- Hangzhou Focus Corporation: Certificate CN23/00000800Document1 pageHangzhou Focus Corporation: Certificate CN23/00000800shaoxin.bdNo ratings yet
- ISO22000Document1 pageISO22000arabelle.valeskaNo ratings yet
- Heineken Vietnam Brewery-Vung Tau Limited Company: Food Safety System Certification 22000 (Version 5 - May 2019)Document1 pageHeineken Vietnam Brewery-Vung Tau Limited Company: Food Safety System Certification 22000 (Version 5 - May 2019)kieutien782No ratings yet
- Es-Form-Annex522-Ago2022 (QP Declaration CHX)Document4 pagesEs-Form-Annex522-Ago2022 (QP Declaration CHX)marinaNo ratings yet
- I491 Khodiyar Agro IndustriesDocument1 pageI491 Khodiyar Agro Industriesansarmulla711No ratings yet
- HACCP AuditDocument1 pageHACCP Audithariprem26No ratings yet
- Bluesun-Mark Organic Standard: Packaging and Storing of Fruit and VegetablesDocument10 pagesBluesun-Mark Organic Standard: Packaging and Storing of Fruit and VegetablesSerghei DeliuNo ratings yet
- Guideline For HACCP AuditorDocument28 pagesGuideline For HACCP AuditorLee Wi SyuenNo ratings yet
- Decree No. 155:2018:ND-CPDocument43 pagesDecree No. 155:2018:ND-CPHieu NgNo ratings yet
- An Introduction To Haccp Food SafetyDocument24 pagesAn Introduction To Haccp Food SafetypeterlimttkNo ratings yet
- 2019 - Certificate - Linea Pet Peru - BRC Packaging & Packaging Material CertificateDocument1 page2019 - Certificate - Linea Pet Peru - BRC Packaging & Packaging Material CertificatenatyloliNo ratings yet
- Haccp FormDocument4 pagesHaccp FormMohd Khairul Md DinNo ratings yet
- Who GMPDocument44 pagesWho GMPYoussef KaidNo ratings yet
- Cases HD 2011 Including Answers PDFDocument11 pagesCases HD 2011 Including Answers PDFAhmedElSayedNo ratings yet
- fssc22000 Features v3.2 2015 PDFDocument16 pagesfssc22000 Features v3.2 2015 PDFApple Sirinart ThaNo ratings yet
- Reporte Auditoria BRC GAMCORP 2018Document53 pagesReporte Auditoria BRC GAMCORP 2018aprador2No ratings yet
- CDF20403 CH 3Document21 pagesCDF20403 CH 3fdhln sakinahNo ratings yet
- 2020LR91a - Compact Dry LM Enumeration - Certificate 2022-2026Document1 page2020LR91a - Compact Dry LM Enumeration - Certificate 2022-2026jupablex.ton2114No ratings yet
- EUL 0653-143-00 OnSiteCOVID-19 Ag RapidTest v1.0Document18 pagesEUL 0653-143-00 OnSiteCOVID-19 Ag RapidTest v1.0Maciej OONo ratings yet
- Ilovepdf Merged-9Document6 pagesIlovepdf Merged-9Wandysson SantosNo ratings yet
- Dupont Nutrition Newark Delaware USA SGSDocument1 pageDupont Nutrition Newark Delaware USA SGSAbhishek VermaNo ratings yet
- AyushmarksDocument7 pagesAyushmarkscosmpiroNo ratings yet
- BV CER Profile 2016Document3 pagesBV CER Profile 2016Harthwell CapistranoNo ratings yet
- FSSC 22000 La Bañeza - 06.04.2020Document1 pageFSSC 22000 La Bañeza - 06.04.2020DomingoNo ratings yet
- BRC Certificate 2018Document1 pageBRC Certificate 2018Khalil ValhallaNo ratings yet
- Importing Food Products Into IndiaDocument4 pagesImporting Food Products Into IndiaanupamkhannaNo ratings yet
- Final Amrit Herbals Organic QvaDocument1 pageFinal Amrit Herbals Organic QvaKartik RajputNo ratings yet
- Quality AssuranceDocument30 pagesQuality Assurancedavid oj100% (1)
- Gluten and Celiac Disease: Gluten-Free Certification Guide & Terms of CertificationsDocument9 pagesGluten and Celiac Disease: Gluten-Free Certification Guide & Terms of CertificationsKSXNo ratings yet
- Desain Sentra Pangan Sesuai Penerapan Cara Produksi Pangan Olahan Yang Baik (Cppob) / Good Dak Kementerian Perindustrian Ta.2023Document74 pagesDesain Sentra Pangan Sesuai Penerapan Cara Produksi Pangan Olahan Yang Baik (Cppob) / Good Dak Kementerian Perindustrian Ta.2023Zaiful HamzahNo ratings yet
- Invoice: Product Quantity PriceDocument1 pageInvoice: Product Quantity PriceMalaika ShaikhNo ratings yet
- Health and Nutrition History Client Information: Today's DateDocument6 pagesHealth and Nutrition History Client Information: Today's DateMalaika ShaikhNo ratings yet
- Book Reviews: Williams' Basic Nutrition and Diet Therapy. 13th EditionDocument1 pageBook Reviews: Williams' Basic Nutrition and Diet Therapy. 13th EditionMalaika ShaikhNo ratings yet
- Essential Duas PDFDocument84 pagesEssential Duas PDFMalaika ShaikhNo ratings yet
- CURE AND RECOVERY - DR - AMRUTHA MANOHARANDocument27 pagesCURE AND RECOVERY - DR - AMRUTHA MANOHARANMalaika ShaikhNo ratings yet
- Death - Think AboutDocument25 pagesDeath - Think AboutMalaika ShaikhNo ratings yet
- WHA43 - TD-5 - Eng VeryDocument50 pagesWHA43 - TD-5 - Eng VeryMalaika ShaikhNo ratings yet