Professional Documents
Culture Documents
Nanoparticle-Mediated Drug Delivery To Tumor Vasculature Suppresses Metastasis
Uploaded by
ahmet kaçakOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Nanoparticle-Mediated Drug Delivery To Tumor Vasculature Suppresses Metastasis
Uploaded by
ahmet kaçakCopyright:
Available Formats
Nanoparticle-mediated drug delivery to tumor
vasculature suppresses metastasis
Eric A. Murphy*, Bharat K. Majeti*, Leo A. Barnes, Milan Makale, Sara M. Weis, Kimberly Lutu-Fuga,
Wolfgang Wrasidlo, and David A. Cheresh†
Department of Pathology, Moores Cancer Center, University of California at San Diego, 3855 Health Sciences Drive, La Jolla, CA 92093
Communicated by Dennis A. Carson, University of California at San Diego School of Medicine, La Jolla, CA, April 17, 2008 (received for review
March 7, 2008)
Integrin ␣3 is found on a subset of tumor blood vessels where terol, dioleoylphosphatidylethanolamine (DOPE), dis-
it is associated with angiogenesis and malignant tumor growth. tearoylphosphatidylethanolamine (DSPE)-mPEG2000, and
We designed an ␣3-targeted nanoparticle (NP) encapsulating DSPE-cyclic RGDfK. After dehydration/rehydration and soni-
the cytotoxic drug doxorubicin (Dox) for targeted drug delivery to cation, the NPs were stepwise extruded down through a mini-
the ␣3-expressing tumor vasculature. We observed real-time mum pore size of 100 nm. Dynamic light scattering demonstrated
targeting of this NP to tumor vessels and noted selective apoptosis that the NPs had a mean hydrodynamic diameter of 105 nm and
in regions of the ␣3-expressing tumor vasculature. In clinically a zeta potential close to neutrality [supporting information (SI)
relevant pancreatic and renal cell orthotopic models of spontane- Table S1]. A 6-(((4,4-difluoro-5-(2-thienyl)-4-bora-3a,4a-diaza-
ous metastasis, targeted delivery of Dox produced an antimeta- s-indacene-3-yl)styryloxy)acetyl) aminohexanoic acid, succin-
static effect. In fact, ␣3-mediated delivery of this drug to the imidyl ester (BODIPY) 630/650 fluorophore was conjugated to
tumor vasculature resulted in a 15-fold increase in antimetastatic DOPE, and this fluorescent lipid was incorporated into the
activity without producing drug-associated weight loss as ob- particle at low concentration for imaging both in vitro and in vivo
served with systemic administration of the free drug. These find- targeting. To address the specificity of the NPs for integrin ␣v3,
ings reveal that NP-based delivery of cytotoxic drugs to the we performed binding studies on human umbilical vein endo-
␣3-positive tumor vasculature represents an approach for treat- thelial cells (HUVECs) that express high levels of this integrin
ing metastatic disease. (Fig. 1B). HUVECs were pretreated with a 20-fold molar excess
of either cRGDfK (targeting peptide) or cRADfK (control
antiangiogenic 兩 intravital microscopy 兩 pancreatic cancer 兩 peptide), and the RGD-NPs were subsequently incubated with
renal cell carcinoma 兩 liposome the cells. RGD-NPs bound and internalized within 20 min in the
presence of the cRADfK peptide as expected, whereas the
soluble cRGDfK peptide completely inhibited the binding of
A ngiogenesis contributes to tumor malignancy and is linked
to a wide variety of inflammatory and ischemic diseases.
Integrin ␣v3, an internalization receptor for a number of
the RGD-NPs (Fig. 1B).
viruses (1, 2), was shown to be preferentially expressed on the In Vivo Targeting of the RGD-NPs. After establishing targeting in
angiogenic endothelium in malignant or diseased tissues (3, 4). vitro, the RGD-NPs were tested for targeting to the tumor
These characteristics of integrin ␣v3 make it an attractive vasculature. M21L-GFP mouse melanoma cells (integrin ␣v3
targeting molecule for molecular imaging and delivery of ther- negative) were implanted in dorsal skin-fold window chambers
apeutics for cancer. Previous studies have shown that ␣v3- and allowed to grow and become vascularized for 7 days. We then
targeted nanoparticles (NPs) coupled to contrast agents can injected RGD-NPs (targeted) or RAD-NPs (nontargeted) and
readily image the tumor vasculature revealing ‘‘hot spots’’ of imaged both the tumor (GFP) and NPs (BODIPY) by confocal
angiogenesis within the tumor (5, 6). Therapeutic studies using microscopy. RGD-NPs targeted the newly forming tips of the
the ␣v integrin-targeting peptide, RGD-4C, demonstrated that tumor neovasculature associated with the tumor margin within 2 h
this peptide effectively targeted doxorubicin (Dox) to the tumor and reached maximum binding ⬇5 h after i.v. injection, whereas the
neovasculature and enhanced efficacy in human breast cancer corresponding RAD-NP did not accumulate in the tumor neovas-
xenografts in mice (7). In another study, an ␣v3-targeted NP culature at all time points examined (Fig. 1C).
delivering a suicide gene to angiogenic blood vessels was capable of
producing an anticancer response (8). Although integrin ␣v3 is a RGD-Dox-NPs Are Antiangiogenic. To study the antiangiogenic
marker of angiogenic endothelium, histological analysis of breast effect of the targeted NP, mice containing s.c. Matrigel plugs
cancer biopsy tissue revealed that ␣v3 was a primary marker of loaded with basic fibroblast growth factor (bFGF) were i.v.
blood vessels within the most malignant tumors (4). In fact, a strong injected with NPs containing Dox. Angiogenesis was measured
correlation was established between the percent of ␣v3-positive after 7 days by labeling the vasculature with fluorescein-labeled
Griffonia Simplicifolia lectin (10). It is important to note that this
MEDICAL SCIENCES
vessels within the tumor and disease progression (9).
Here, we report the design and characterization of an ␣v3- is a model of normal angiogenesis induced by a proangiogenic
targeted NP capable of delivering various pharmacological agents factor and not a model of tumor angiogenesis. Animals treated
to the ␣v3-expressing tumor vasculature. Evidence is provided with RGD-Dox-NPs (1 mg/kg total Dox) demonstrated vascular
that an ␣v3-targeted NP carrying the cytotoxic drug Dox is
capable of controlling the metastatic behavior of pancreatic and Author contributions: E.A.M., B.K.M., S.M.W., W.W., and D.A.C. designed research; E.A.M.,
renal cell cancer in mice. Importantly, targeted delivery of Dox to B.K.M., L.A.B., M.M., and K.L.-F. performed research; E.A.M., B.K.M., S.M.W., and D.A.C.
the tumor vasculature provided a 15-fold increase in the efficacy of analyzed data; and E.A.M., B.K.M., and D.A.C. wrote the paper.
the drug while producing few, if any, side effects. The authors declare no conflict of interest.
*E.A.M. and B.K.M. contributed equally to this work.
Results †To whom correspondence should be addressed. E-mail: dcheresh@ucsd.edu.
Design of NPs Targeted to Angiogenic Endothelium. A schematic This article contains supporting information online at www.pnas.org/cgi/content/full/
Downloaded by guest on June 23, 2021
representation of the targeted NP (RGD-NP) (Fig. 1A), which 0803728105/DCSupplemental.
is composed of distearoylphosphatidylcholine (DSPC), choles- © 2008 by The National Academy of Sciences of the USA
www.pnas.org兾cgi兾doi兾10.1073兾pnas.0803728105 PNAS 兩 July 8, 2008 兩 vol. 105 兩 no. 27 兩 9343–9348
A A
C y c lic
R G D fK
PBS
PBS RGD-Dox-NP
RGD-Dox-NP
PEG
2000
B RGD-NP
c(RGDfK)
20 fold molar
excess of peptide
B 16000 C1000
b F G F - in d u c e d A n g io g e n e s is (R a w O D )
900
b F G F- in d u c e d A n g io g e n e s is (R a w O D )
14000
c(RADfK) 800
12000
700
10000 600
8000 500
C 6000 400
*=0.004
* =0.04 300
4000
200
2000 100
RAD-NP 0 0
PBS RGD-Dox RGD c(RGDfK) PBS RAD-Dox RGD-Dox
-NP -NP -NP -NP
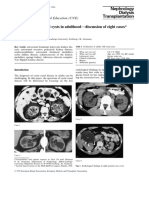
Fig. 2. Vascular disruption in the mouse Matrigel model with ␣v3-targeted
RGD-Dox-NP. Mice were injected s.c. on the flank with Matrigel containing
400 ng of human recombinant bFGF. NPs containing 1 mg/kg of Dox were
i.v.-injected on days 1, 3, and 5. (A) After the treatment, mice were i.v.-injected
with fluorescein-labeled G. simplicifolia lectin and the plugs were removed
and imaged by scanning confocal microscopy. (B and C) To quantify angio-
RGD-NP genesis, the matrigel plugs were removed and the fluorescent-lectin content
was quantified with a fluorimeter. *, P ⬍ 0.05 for RGD-Dox-NP vs. PBS. (Scale
bar: 100 m.)
pancreatic cancer cells derived from a spontaneous murine
Fig. 1. In vitro and in vivo ␣v3 targeting of RGD-NP. (A) Schematic pancreatic tumor (13) were injected into the tail of the
representation and transmission EM of Dox-loaded RGD-NP. (B) Competition pancreas. After 11 days of tumor growth, we injected f luores-
assay for in vitro ␣v3 targeting of RGD-NP in endothelial cells. HUVECs were cent RGD-NPs i.v. and observed accumulation of the RGD-
pretreated for 5 min with a 20-fold molar excess of either cRGDfK or cRADfK NPs in the pancreatic tumor vasculature but not in the
to test for inhibition of NP binding. Subsequently, the cells were incubated
vasculature of the adjacent normal pancreatic tissue (Fig. 3A).
with the RGD-NPs for 20 min, and binding was studied by scanning confocal
microscopy for the BODIPY 630/650 dye. (C) In vivo integrin ␣v3 targeting of
Additionally, tissues that manifest a filtration function exhibit
RGD-NPs within the tumor neovasculature was studied by intravital micros- detectable concentrations of NPs, albeit at significantly lower
copy with the dorsal skin-fold window chamber. M21L melanomas (␣v3 f luorescence intensities than the tumor tissue. Specifically, we
negative) were allowed to vascularize for 7 days, and mice were i.v.-injected observed minimal accumulation of the RGD-NPs in the
with 200 nmol of either RGD-NP or RAD-NP containing BODIPY 630/650. NPs kidney, liver, and lung but found no accumulation in the brain,
were imaged by confocal scanning microscopy at 5-h postinjection. GFP- heart, or spleen (Fig. 3 A and B).
labeled M21L melanomas are shown in green, and NPs are in blue. (Scale bars: For efficacy studies, mice implanted with orthotopic pancre-
A, 100 nm; B and C, 100 m.)
atic tumors were treated on days 5, 7, and 9, and the effects of
the RGD-Dox-NPs were observed on day 11. The primary
pancreatic tumors were sectioned and analyzed by immunohis-
pruning when compared with the normal vascular structure and tochemistry. The serial sections of primary tumors treated with
branching of animals treated with PBS (Fig. 2A) and inhibited RGD-Dox-NPs were stained with hematoxylin and eosin and
angiogenesis by ⬇70% (Fig. 2B). Although the cyclic RGDfK analyzed for total blood vessels, 3-positive blood vessels, and
peptide has been previously shown to be an integrin ␣v3 apoptosis (TUNEL stain) (Fig. 3C). For tumors treated with
antagonist (11, 12), minimal response was observed when testing RGD-Dox-NPs at 1 mg/kg (as in Fig. 3D), we observed apoptosis
RGD-NP without Dox or cyclic peptide alone (Fig. 2B). Also, in angiogenic hot spots that corresponded to the 3 positive
RAD-Dox-NPs demonstrated only a marginal reduction (24%) blood vessels associated with the tumor margin (Fig. 3C). No
in neovascularization relative to control animals (Fig. 2C). These apoptosis was observed in areas corresponding to 3-negative
findings reveal that integrin ␣v3 targeting of Dox containing vessels. Additionally, no apoptosis was observed in tumors
NPs produces a strong antiangiogenic effect. treated with the RAD-Dox-NPs, even when 3-expressing blood
vessels were present (data not shown).
Comparison of RGD-Dox-NP Efficacy on Primary vs. Metastatic Sites in Orthotopically implanted R40P cells typically form metastatic
Pancreatic Carcinoma. We next evaluated the targeting and lesions in the hepatic hilar lymph node, which is important
Downloaded by guest on June 23, 2021
efficacy of the RGD-Dox-NPs in a syngeneic murine ortho- because regional lymph node metastasis is an important predic-
topic tumor model of pancreatic carcinoma. R40P murine tor for survival of pancreatic adenocarcinoma patients (14, 15).
9344 兩 www.pnas.org兾cgi兾doi兾10.1073兾pnas.0803728105 Murphy et al.
A B 25%
Nanoparticle Accumulation
20%
(%of total pixels)
15%
L iv e r H e a rt Pancreas 10%
5%
0%
s
r
rt
ng
in
ve
ne
ea
ea
cr or
s)
ra
Lu
Li
id
cr
an m
ea
B
H
K
an
(P Tu
P
K id n e y Lung Pancreas (Tumor)
H ila r L N T u m o r (m g )
C
T umor Weig ht (mg )
500 40
35
400 30
300 25
20
200 15
10 *=0.02
Tumor 100
5
0 0
P ox
P ox
P ox
S
P ox
S
ox
ox
B
-N -D
-N -D
B
-N -D
-N -D
P
D
P
D
AD
D
AD
G
G
R
R
H&E B lood vessels
E Primary Tumor Metastasis
25
H ila r L N T u m o r (m g )
A poptosis 700
T umor Weig ht (mg )
600 20
500
400 15
300 10
200 ** =0.014
b3 100 5
0 0
PBS 1 7.5 15 PBS 1 7.5 15
T reatmen t (D o x mg /k g ) T reatment (D ox mg /kg )
Fig. 3. Suppression of metastasis in an orthotopic model of pancreatic cancer. (A) R40P pancreatic carcinoma cells were injected into the tail of the pancreas
in Tie-2 GFP mice and allowed to grow for 11 days. RGD-NPs labeled with BODIPY 630/650 were i.v.-injected, and the primary tumor, pancreas, liver, lung, kidney,
and heart were imaged by confocal microscopy. Red represents rhodamine-labeled G. simplicifolia lectin for staining the endothelium, and green represents NP
binding. (B) Quantitation of the fluorescent NPs in various tissues expressed as the percent of pixels from the confocal images. (C) Immunohistochemistry of serial
sections from the tumors treated with the RGD-Dox-NPs demonstrates areas of apoptosis (TUNEL staining) that colocalize only with blood vessels that express
the target (3). (D) Integrin ␣v3-targeted RGD-Dox-NPs prevent metastasis to the hepatic hilar lymph node. After surgical implantation of the cells, mice were treated
on days 5, 7, and 9 with RGD-Dox-NP, RAD-Dox-NP, free Dox, or PBS (each with 1 mg/kg total Dox per dose). On day 11, the primary tumor and the hepatic hilar lymph
node were resected and weighed. (E) Effect of free Dox at various concentrations (1, 7.5, and 15 mg/kg dosed on days 5, 7, and 9) on primary tumor growth and metastasis
to the hepatic hilar lymph node. *, P ⬍ 0.05 for RGD-Dox-NP vs. PBS. **, P ⬍ 0.05 for 15 mg/kg free Dox vs. PBS. (Scale bars: A and C, 100 m.)
To assess the effects of the RGD-Dox-NP on primary tumor containing 1 mg/kg Dox (Fig. 3 D and E). However, dosing
growth and metastasis, tumor-bearing mice were injected with animals with 15 mg/kg of free Dox caused severe weight loss
NPs on days 5, 7, and 9 after orthotopic tumor implantation. We (18% reduction in body weight), whereas the RGD-Dox-NP
then measured the size of the primary tumors and metastatic treatment at 1 mg/kg showed only a 0.8% decline in body weight
lesions on day 11. While the RGD-Dox-NPs containing 1 mg/kg (Fig. S2). Additionally, like the RGD-Dox-NP-treated animals,
of Dox produced a modest reduction of the primary tumor control animals did not gain or lose any weight during the
MEDICAL SCIENCES
growth (23%), it significantly reduced metastasis to the hepatic experiment.
hilar lymph node (82%) as compared with control animals (Fig. As an alternate treatment regimen, we increased the fre-
3D). The untargeted particle, RAD-Dox-NPs containing 1 quency of doses by treating animals on days 7, 8, and 9 after
mg/kg Dox, had no significant effect on the primary tumor or the tumor implantation. In this case, we observed a more robust
metastatic lesions (Fig. 3D). Importantly, 1 mg/kg of free Dox impact on the primary tumor (43% reduction vs. control) while
had no effect on either primary tumor growth or metastasis (Fig. maintaining a substantial antimetastatic effect (90% reduction
3D). Similarly, no antitumor effect was seen after separate vs. control) (Fig. S3). These studies suggest that targeting Dox to
administration of the empty RGD-NP together with 1 mg/kg free the tumor vasculature greatly affects metastasis.
Dox (Fig. S1). To determine the concentration of free Dox that
was necessary to duplicate the anticancer effects observed with Comparison of RGD-Dox-NP Efficacy on Primary vs. Metastatic Sites in
the RGD-Dox-NPs we injected varying concentrations of free Renal Cell Carcinoma. We further tested the NPs in an orthotopic
Downloaded by guest on June 23, 2021
Dox and found that 15 mg/kg was required to achieve a similar model of renal cell carcinoma. In this model, human SN12C
reduction in metastasis as demonstrated with the RGD-NP renal carcinoma cells expressing red fluorescent protein (RFP)
Murphy et al. PNAS 兩 July 8, 2008 兩 vol. 105 兩 no. 27 兩 9345
Table 1. Incidence of metastasis in the orthotopic renal cell
carcinoma model
A
P rim a ry tu m o r w e ig h t (m g )
Primary Tumor Metastasis
Metastatic site RGD-DOX-NP RAD-DOX-NP 900
800
M e ta s ta tic b u rd e n
Pancreas 5/8 6/8 700 1600
Spleen 3/8 5/8 600
500 1200
Diaphragm 1/8 4/8 400
800
Liver 1/8 4/8 300
200 400
Lungs 1/8 2/8 100 * =0.014
Gut 4/8 6/8 0 0
RAD-Dox-NP RGD-Dox-NP RAD-Dox-NP RGD-Dox-NP
Primary 8/8 8/8
PANCREAS SPLEEN DIAPHRAGM LIVER
B
RGD-Dox RAD-Dox RGD-Dox RAD-Dox RGD-Dox RAD-Dox RGD-Dox RAD-Dox
were injected into the left kidney of athymic nude mice. After 8 -NP -NP -NP -NP -NP -NP -NP -NP
days, mice were dosed i.v. with either the RAD-Dox-NP or
RGD-Dox-NP at 2 mg/kg total Dox every other day until the
experiment was terminated on day 42. At that time point, control
animals typically demonstrated widespread metastasis to the
liver, spleen, diaphragm, and pancreas. However, treatment with
the RGD-Dox-NP significantly reduced both the incidence of
metastasis (Table 1) and the total metastatic burden (91%) (Fig.
4A). Images of the RFP-expressing metastatic lesions are shown
for each of the metastatic sites in all animals (Fig. 4B). Metas-
tasis was quantified by measuring the total pixels from each
organ in both treatment groups. Similar to the pancreatic
carcinoma results (Fig. 3D), there was only a modest reduction
of primary tumor burden (35%) in the renal cell carcinoma
model in animals treated with RGD-Dox-NPs compared with
the RAD-Dox-NPs (P ⫽ 0.27) (Fig. 4A). Nevertheless, during
the course of the NP treatment, the mice gained weight normally
and displayed no obvious side effects (data not shown).
Discussion
Nontargeted long circulating liposomes, e.g., Doxil, have been Fig. 4. RGD-Dox-NP reduces overall metastasis in an orthotopic renal cell
extensively used for delivering chemotherapeutic drugs to tu- carcinoma model. (A) Human SN12C-RFP cells were injected into the left
mors via the enhanced permeability and retention mechanism kidney and primary tumors were established for 8 days. At that time, NPs
(16). Although liposomal delivery of cytotoxic drugs can im- containing 2 mg/kg Dox were dosed every other day from day 8 until day 42.
At the endpoint, both kidneys were removed and the difference in weight was
prove antitumor activity, targeted delivery of these particles
attributed to primary tumor burden. Additionally, the diaphragm, liver, pan-
represents a potential approach to further enhance efficacy and creas, and spleen were resected and imaged for RFP-expressing metastatic
minimize toxicity. Recent studies have described the design of lesions by using an OV-100 whole animal imaging system. The total pixels in
NPs that target the tumor endothelium to improve diagnosis via each image were quantified by using ImageJ software and the average pixels
imaging (5, 6, 17) or deliver therapeutics to solid tumors (8, 18, of the metastatic lesions/animal are represented as the metastatic burden.
19). The majority of the therapeutic research has focused on *, P ⬍ 0.05 for RGD-Dox-NPs vs. RAD-Dox-NPs. (B) Images of the RFP fluores-
using various forms of RGD peptides for targeting integrin ␣v3, cence from the metastatic lesions present in the diaphragm, liver, pancreas,
which is present on the tumor neovasculature (20). and spleen used for the quantification of the metastatic burden in A are
presented for each animal. (Scale bars: 5 mm.)
Integrin ␣v3 represents an ideal vascular targeting receptor
because it is highly expressed on the angiogenic endothelium and
expression of this receptor on tumor vessels correlates with express integrin ␣v3. The modest effect on the primary tumor
disease progression (9). Additionally, this receptor is used by is perhaps not surprising, because treatment was initiated after
viruses for internalization into cells, making it an optimal the primary tumors had established a blood supply. Tumors with
targeting receptor for NP-mediated drug delivery (21). By
preestablished vasculature typically express integrin ␣v3 on a
displaying targeting ligands such as cyclic RGD peptides in a
subset of vessels associated with the tumor margin as we (Fig.
multivalent array on the surface of NPs, avidity for the target is
3C) and others have observed (4, 9). Because apoptosis is
greatly increased as the binding to integrins causes both lateral
diffusion and clustering of multimeric complexes (22, 23). This induced only in areas of the tumor colocalizing with 3, only a
increase in avidity leads to active targeting even in the presence fraction of the primary tumor (i.e., that near the margin) is
of shear stress generated by the flowing blood at the surface of expected to be impacted by the 3-targeted NPs. Importantly,
the endothelium. The combination of these properties make even though the hydrodynamic diameter of the untargeted
integrin ␣v3 a particularly useful target for delivering chemo- RAD-Dox-NPs was 100–120 nm, no effect was observed on
therapeutic molecules to the tumor endothelium. inhibition of primary tumor growth because of nonspecific
In this study, we evaluated the impact of integrin ␣v3- accumulation in the tumor via the enhanced permeability and
targeted NP drug delivery on primary tumor growth and spon- retention mechanism. Additionally, the untargeted RAD-Dox-
taneous metastasis of two orthotopic cancer models. Although NPs did not induce apoptosis in any area of the tumor (data not
we observed a reproducible, but modest, effect on primary tumor shown), confirming that targeting integrin ␣v3 was necessary
growth, we noted a substantial impact on metastatic disease. for localized tumor cell apoptosis.
Downloaded by guest on June 23, 2021
This effect was not based on integrin ␣v3 expression on the Several hypotheses might help to explain the preferential
tumor cells because the tumor cells examined in this study do not effect of the RGD-Dox-NPs on the metastatic lesions in these
9346 兩 www.pnas.org兾cgi兾doi兾10.1073兾pnas.0803728105 Murphy et al.
orthotopic tumor models. First, establishment of the blood in a bath sonicator (ULTRAsonik 28X) for 2–3 min at room temperature to
supply might be much more critical at the newly forming produce multilamellar vesicles (MLVs). MLVs were then sonicated with a
metastatic sites relative to the established primary tumor, cre- Ti-probe (Branson 450 sonifier) for 1–2 min to produce small unilamellar
vesicles (SUVs) as indicated by the formation of a translucent solution. To
ating a differential sensitivity to the targeted treatment. Second,
reduce the size of the SUVs, stepwise extrusion was performed with the
we demonstrate that RGD-Dox-NPs induce apoptosis in the final step being extrusion through a polycarbonate filter with 100-nm pore
3-positive tumor vasculature (Fig. 3C), and this effect on the size (Whatman). The buffer was exchanged with 20 mM Hepes, 150 mM
primary tumor could influence the invasive behavior of the tu- NaCl, pH 7.4 by using gel-filtration chromatography (PD-10; GE Health-
mor and its ability to metastasize. Third, in the tumor models care). Dox (Fluka) encapsulation was performed as described (27) with one
examined here, to some degree primary tumor growth correlated slight variation in loading temperature. After addition of Dox to the NPs,
with metastatic potential. For example, treatment of the pan- the solution was heated at 55°C for 1h and then left overnight at room
creatic carcinomas with 7.5 or 15 mg/kg of free Dox reduced both temperature before purification by PD-10 column, eluted with water. The
amount of encapsulated Dox was quantified by adding 1.5% Triton X-100
the size of the primary tumor and the metastatic burden in the
to disrupt the NPs and comparing the absorbance at 476 nm on a spectro-
hepatic hilar lymph node (Fig. 3E). Further studies are necessary photometer to a standard curve of free Dox.
to elucidate the mechanism of the antimetastatic effect that
might become important for other targeting strategies. In Vitro Competition Assay. HUVECs were seeded and grown overnight in
Using NP-mediated Dox delivery to the tumor vasculature, 48-well plates coated with 10 g/ml collagen type I and blocked with 5% BSA.
we were able to observe a 15-fold improvement in drug efficacy For competition, a 20-fold molar excess of either cRGDfK or cRADfK peptide
relative to animals treated with free drug (Fig. 3). Dox is was preincubated with the cells for 5 min in serum-free medium. NPs contain-
dose-limited by cardiotoxicity (24), and lower doses greatly ing 1% of a BODIPY 630/650 (Invitrogen)-linked DOPE for fluorescence mi-
reduce the side effects of this chemotherapeutic agent. By croscopy were added to the HUVECs for 20 min at 37°C. Cells were fixed and
NP binding was visualized by confocal microscopy (Nikon C1si).
targeting NPs to the tumor neovasculature, we observed an
antitumor effect at 1 mg/kg of NP-encapsulated Dox with no
Dorsal Skin-Fold Window Chamber. Intravital microscopy of NP binding to the
appreciable weight loss, whereas 15 mg/kg of free Dox was tumor neovasculature was monitored in the dorsal skin-fold window chamber
required for similar efficacy, leading to an 18% decrease in as described (21, 22). Two days after surgical implantation of the window
body weight (Fig. S2). It will be interesting to use this approach chamber, a concentrated (4 l) suspension of 800,000 M21L-GFP melanoma
with a number of the newer pharmacological agents designed cells was injected into the retractor muscle within the chamber. After 7 days
to suppress tumor or vascular cell signal transduction (25). and the appearance of tumor-associated angiogenesis, fluorescent RGD-NPs
Although some of these agents are beginning to show promise or RAD-NPs were injected i.v., and the animals were imaged 5 h postinjection
in the clinic, targeted NP delivery would likely reduce the by confocal microscopy (Nikon C1si).
concentration needed for efficacy and minimize problems
associated with pharmacokinetics and/or side effects. In Vivo Angiogenesis. The Matrigel assay was performed to assess in vivo
angiogenesis (10). Briefly, mice were injected s.c. on the flank with 400 l of
Methods growth factor-reduced Matrigel (BD Biosciences) containing either sterile
saline or 400 ng of human recombinant bFGF (Millipore). After 7 days, mice
Animal Studies. All animal procedures were conducted in accordance with all were injected i.v. with 20 g fluorescein-labeled G. simplicifolia lectin that
appropriate regulatory standards under protocol SO5018 and approved by binds selectively to mouse endothelial cells (GSL I–BSL I; Vector Labs). The
the University of California San Diego Institutional Animal Care and Use Matrigel plugs were removed, photographed, viewed whole-mount by con-
Committee. focal microscopy (Nikon C1si), and then fixed and stained for fluorescence
microscopy or homogenized, and the fluorescence content was quantified
Cell Culture. M21L-GFP melanomas were maintained under standard culture with a fluorimeter by using standard fluorescein filters (Tecan).
conditions in DMEM supplemented with 10% FBS. R40P cells were isolated and
cultured from a spontaneous pancreatic tumor in Pdx1-Cre,LSL- Pancreatic Carcinoma Model. The orthotopic pancreatic carcinoma model has
KRasG12D,Ink4a/Arflox/lox mice after 7 weeks. Both the mice and tumor cell been described (28, 29). Briefly, 6- to 10-week-old Tie2-GFP mice (30) were
isolation procedure have been described (13). The R40P cells were maintained injected with 1 million syngeneic murine R40P cells in the tail of the pancreas.
under standard culture conditions in RPMI medium 1640 supplemented with NPs containing Dox were injected i.v. on days 5, 7, and 9 postsurgical implan-
10% FBS. SN12C-RFP cells were a gift from Robert Hoffman at AntiCancer, Inc. tation of the cells. On day 11, the primary tumor and the hepatic hilar lymph
(San Diego). The SN12C-RFP renal carcinomas were grown in RPMI medium node were resected and weighed. To quantify metastasis, the combined
1640 supplemented with 10% FBS and maintained under standard culture weight of both the lymph node and metastatic lesions is reported. Standard
conditions. HUVECs were purchased from Lonza and maintained in EBM-2 immunohistochemistry on frozen sections was performed. Images were ac-
with EGM-2 singlequots. HUVECs were propagated on plates coated with 10 quired by using either a standard light microscope equipped with a CCD
g/ml of collagen type I (Millipore), and all experiments were conducted at camera or confocal microscopy. The 3 integrin antibody was from BD Bio-
passage ⬍6. sciences (550541). Blood vessels were labeled for ‘‘EC markers’’ with a mix of
rat anti-mouse antibodies recognizing Flk-1 (BD Biosciences; 550549), CD31
Synthesis of Peptide-Lipid Conjugates. The cyclic peptides, cRGDfK and (BD Biosciences; 550274), VE-cadherin (BD Biosciences; 555289), and CD105
cRADfK (f denotes D-phenylalanine), were synthesized by using standard (Millipore; CBL1358). The Apoptag kit from Millipore (S7165) was used as
Fmoc solid-phase chemistry as described (26). Peptides were purified by directed to assess apoptosis on frozen tissue sections. For targeting studies and
reverse-phase HPLC, and exact mass was confirmed by mass spectroscopy. biodistribution, fluorescent NPs (as described above) were injected on day 11,
MEDICAL SCIENCES
For conjugation to lipid, the peptides were conjugated to a short linker, and the organs were resected at 5-h postinjection. The organs were imaged via
succinimidyl ester-(PEO)4-maleimide (Pierce). DSPE was reacted with imi- whole mount on glass slides by confocal microscopy (Nikon C1si). Fluorescence
nothiolane (Sigma–Aldrich) to produce a free thiol. The DSPE containing was observed from both the NP and the blood vessels of the Tie2-GFP mice in
the free thiol group was reacted with the cRGDfK-(PEO)4-maleimide or which GFP was expressed in the endothelium.
cRADfK-(PEO)4-maleimide to produce the peptide-lipid conjugates, and
those conjugates were recrystallized in methanol/diethyl ether 1:9 at 4°C Renal Cell Carcinoma Model. The renal cell carcinoma model was used as
overnight. The exact mass of the peptide-lipid conjugates was verified by described (31). One million tumor cells in 20 l of a 1:1 PBS/Matrigel mixture
mass spectroscopy. were injected into the lower pole of the kidney just below the renal subcap-
sule in male nu/nu mice. The needle was removed after a visible blister formed
NP Preparation. The particle formulation of cholesterol/DOPE/DSPC/DSPE- and leakage of the tumor cell suspension was minimal. Animals that formed
(PEO)4-cRGDfK/DSPE-mPEG2000 (6:6:6:1:1 molar ratio) in chloroform was visible blisters upon injection in the subcapsule with minimal leakage were
evaporated under argon gas, and the dried lipid film was hydrated in sterile used for the study. Mice with orthotopic injections of the SN12C-RFP cells were
300 mM ammonium phosphate buffer (pH 7.4) in a total volume of 5 ml at imaged by using the Olympus OV100 Small Animal Imaging System and
Downloaded by guest on June 23, 2021
a total lipid concentration of 3.32 mM for a minimum of 1 h. Liposomes grouped on day 7 based on weight and imaging results. NPs were injected i.v.
were vortexed for 2–3 min to remove any adhering lipid film and sonicated every other day beginning on day 8 and continuing until the end of the
Murphy et al. PNAS 兩 July 8, 2008 兩 vol. 105 兩 no. 27 兩 9347
experiment when the kidneys were resected and weighed. The difference in Statistical Analysis. Error bars represent mean values ⫾ SEM. The statistical
weight between the tumor-bearing kidney and the normal kidney was re- significance of the experiments was determined by two-tailed Student’s t test.
ported. To observe metastasis, various organs, including the spleen, pancreas, P ⬍ 0.05 were considered significant.
diaphragm, lungs, liver, and intestines, were removed, and the RFP expression
was imaged by using the OV100 system. Metastatic disease was quantified by ACKNOWLEDGMENTS. This work was supported by National Cancer Institute
measuring the total pixels in each organ with ImageJ software and quantify- Grant U54-CA119335 (National Cancer Institute Alliance for Nanotechnology
ing the number of pixels in each image above a threshold of fluorescence in Cancer) (to D.A.C.) and National Institutes of Health Grants P01-CA078045
intensity. and P01-CA104898 (to D.A.C.).
1. Wang X, Huang DY, Huong SM, Huang ES (2005) Integrin ␣v3 is a coreceptor for 18. Pastorino F, et al. (2003) Vascular damage and antiangiogenic effects of tumor
human cytomegalovirus. Nat Med 11:515–521. vessel-targeted liposomal chemotherapy. Cancer Res 63:7400 –7409.
2. Wickham TJ, Mathias P, Cheresh DA, Nemerow GR (1993) Integrins ␣v3 and ␣v5 19. Schiffelers RM, et al. (2003) Antitumor efficacy of tumor vasculature-targeted liposo-
promote adenovirus internalization but not virus attachment. Cell 73:309 –319. mal doxorubicin. J Controlled Release 91:115–122.
3. Brooks PC, et al. (1994) Integrin ␣v3 antagonists promote tumor regression by 20. Temming K, Schiffelers RM, Molema G, Kok RJ (2005) RGD-based strategies for selective
inducing apoptosis of angiogenic blood vessels. Cell 79:1157–1164. delivery of therapeutics and imaging agents to the tumor vasculature. Drug Resist
4. Brooks PC, et al. (1995) Antiintegrin ␣v3 blocks human breast cancer growth and Updat 8:381– 402.
angiogenesis in human skin. J Clin Invest 96:1815–1822. 21. Stewart PL, Nemerow GR (2007) Cell integrins: Commonly used receptors for diverse
5. Sipkins DA, et al. (1998) Detection of tumor angiogenesis in vivo by ␣v3-targeted viral pathogens. Trends Microbiol 15:500 –507.
magnetic resonance imaging. Nat Med 4:623– 626. 22. Montet X, Funovics M, Montet-Abou K, Weissleder R, Josephson L (2006) Multiva-
6. Winter PM, et al. (2003) Molecular imaging of angiogenesis in nascent Vx-2 rabbit lent effects of RGD peptides obtained by nanoparticle display. J Med Chem 49:6087–
tumors using a novel ␣v3-targeted nanoparticle and 1.5 tesla magnetic resonance 6093.
imaging. Cancer Res 63:5838 –5843. 23. Garanger E, Boturyn D, Coll JL, Favrot MC, Dumy P (2006) Multivalent RGD synthetic
7. Arap W, Pasqualini R, Ruoslahti E (1998) Cancer treatment by targeted drug delivery to peptides as potent ␣v3 integrin ligands. Org Biomol Chem 4:1958 –1965.
tumor vasculature in a mouse model. Science 279:377–380.
24. Takemura G, Fujiwara H (2007) Doxorubicin-induced cardiomyopathy from the car-
8. Hood JD, et al. (2002) Tumor regression by targeted gene delivery to the neovascula-
diotoxic mechanisms to management. Progr Cardiovas Dis 49:330 –352.
ture. Science 296:2404 –2407.
25. Steeghs N, Nortier JW, Gelderblom H (2007) Small molecule tyrosine kinase inhibitors
9. Gasparini G, et al. (1998) Vascular integrin ␣v3: A new prognostic indicator in breast
in the treatment of solid tumors: An update of recent developments. Ann Surg Oncol
cancer. Clin Cancer Res 4:2625–2634.
14:942–953.
10. Weis SM (2007) Evaluating integrin function in models of angiogenesis and vascular
26. Dai X, Su Z, Liu JO (2000) An improved synthesis of a selective ␣v3-integrin antagonist
permeability. Methods Enzymol 426:505–528.
cyclo(-RGDfK-). Tetrahedron Lett 41:6295– 6298.
11. Haubner R, et al. (1996) Structural and functional aspects of RGD-containing cyclic
27. Fritze A, Hens F, Kimpfler A, Schubert R, Peschka-Suss R (2006) Remote loading of
pentapeptides as highly potent and selective integrin ␣v3 antagonists. J Am Chem Soc
118:7461–7472. doxorubicin into liposomes driven by a transmembrane phosphate gradient. Biochim
12. Pfaff M, et al. (1994) Selective recognition of cyclic RGD peptides of NMR defined Biophys Acta 1758:1633–1640.
conformation by ␣IIb3, ␣v3, and ␣51 integrins. J Biol Chem 269:20233–20238. 28. Grimm J, Potthast A, Wunder A, Moore A (2003) Magnetic resonance imaging of the
13. Aguirre AJ, et al. (2003) Activated Kras and Ink4a/Arf deficiency cooperate to produce pancreas and pancreatic tumors in a mouse orthotopic model of human cancer. Int J
metastatic pancreatic ductal adenocarcinoma. Genes Dev 17:3112–3126. Cancer 106:806 – 811.
14. House MG, et al. (2007) Prognostic significance of pathologic nodal status in patients 29. Tsutsumi S, Yanagawa T, Shimura T, Kuwano H, Raz A (2004) Autocrine motility factor
with resected pancreatic cancer. J Gastrointest Surg 11:1549 –1555. signaling enhances pancreatic cancer metastasis. Clin Cancer Res 10:7775–7784.
15. Schwarz RE, Smith DD (2006) Extent of lymph node retrieval and pancreatic cancer survival: 30. Motoike T, et al. (2000) Universal GFP reporter for the study of vascular development.
Information from a large U.S. population database. Ann Surg Oncol 13:1189 –1200. Genesis 28:75– 81.
16. Allen TM, Cullis PR (2004) Drug delivery systems: Entering the mainstream. Science 31. An Z, Jiang P, Wang X, Moossa AR, Hoffman RM (1999) Development of a high
303:1818 –1822. metastatic orthotopic model of human renal cell carcinoma in nude mice: Benefits of
17. Hu G, et al. (2007) Imaging of Vx-2 rabbit tumors with ␣v-integrin-targeted 111In fragment implantation compared to cell-suspension injection. Clin Exp Metastasis
nanoparticles. Int J Cancer 120:1951–1957. 17:265–270.
Downloaded by guest on June 23, 2021
9348 兩 www.pnas.org兾cgi兾doi兾10.1073兾pnas.0803728105 Murphy et al.
You might also like
- Caregiving Elderly Care TRDocument170 pagesCaregiving Elderly Care TRRodolfo Corpuz67% (3)
- Edmund Bergler - Divorce Won't HelpDocument256 pagesEdmund Bergler - Divorce Won't Helpbmx0100% (2)
- RA 6969 OverviewDocument76 pagesRA 6969 OverviewKaren Feyt Mallari100% (1)
- 00017Document5 pages00017arypwNo ratings yet
- The Laryngoscope - 2009 - Guang Wu - The Relationship Between Microvessel Density The Expression of Vascular EndothelialDocument4 pagesThe Laryngoscope - 2009 - Guang Wu - The Relationship Between Microvessel Density The Expression of Vascular EndothelialWening Dewi HapsariNo ratings yet
- GPR5CD Atamaniuk Et Al-2012-European Journal of Clinical InvestigationDocument8 pagesGPR5CD Atamaniuk Et Al-2012-European Journal of Clinical Investigationben rosenthalNo ratings yet
- 42b3 PDFDocument7 pages42b3 PDFExaminenNo ratings yet
- Antiangiogenic and Antitumoral Effects Mediated by ADocument10 pagesAntiangiogenic and Antitumoral Effects Mediated by AMiss AmyNo ratings yet
- 1513 FullDocument19 pages1513 Fullgerielle mayoNo ratings yet
- Carvalho 2022Document16 pagesCarvalho 2022docadax848No ratings yet
- 10 1016@j Urolonc 2019 03 007Document10 pages10 1016@j Urolonc 2019 03 007Marijana KnezovicNo ratings yet
- Development and Experimental Verification of A.24Document18 pagesDevelopment and Experimental Verification of A.24Michael UNo ratings yet
- Conferences and Reviews Brain Tumors: JdiazepineDocument12 pagesConferences and Reviews Brain Tumors: JdiazepineFlorentina AdascaliteiNo ratings yet
- Fonc 11 709525Document16 pagesFonc 11 709525Bikash Ranjan SamalNo ratings yet
- VDR Gene Single Nucleotide Polymorphisms and Their: Association With Risk of Oral Cavity CarcinomaDocument4 pagesVDR Gene Single Nucleotide Polymorphisms and Their: Association With Risk of Oral Cavity CarcinomaFrancine Rossetti DelboniNo ratings yet
- 10 1126@scitranslmed Aah6510Document14 pages10 1126@scitranslmed Aah6510翁子貽No ratings yet
- INTRO Rev Neuroseñalización en Tumor de PróstataDocument12 pagesINTRO Rev Neuroseñalización en Tumor de Próstatasarahi venturaNo ratings yet
- Fonc 11 664848Document10 pagesFonc 11 664848m8f5mwpzwyNo ratings yet
- Utilizing Targeted Gene Therapy With Nano Particles Binding Alpha V Beta 3 For Imaging and Treating Choroidal NeovascularizationDocument9 pagesUtilizing Targeted Gene Therapy With Nano Particles Binding Alpha V Beta 3 For Imaging and Treating Choroidal Neovascularizationtennisboy92No ratings yet
- Primary Nephrotic Syndrome NewDocument15 pagesPrimary Nephrotic Syndrome Newpj rakNo ratings yet
- Single-Cell Heterogeneity in Ductal Carcinoma in Situ of BreastDocument12 pagesSingle-Cell Heterogeneity in Ductal Carcinoma in Situ of BreastrohitNo ratings yet
- Polycystic Kidney - Eight CasesDocument11 pagesPolycystic Kidney - Eight CasesKAINAT ANJUMNo ratings yet
- Xiao2018 PDFDocument27 pagesXiao2018 PDFBüşra TuranNo ratings yet
- Pedf Pancreatic CancerDocument8 pagesPedf Pancreatic CancerMuhammad Jahanzaib AnwarNo ratings yet
- Srep 45623Document14 pagesSrep 45623javi222222No ratings yet
- Association Between A CTGF Gene Polymorphism andDocument8 pagesAssociation Between A CTGF Gene Polymorphism andAhmad ShamyNo ratings yet
- Art:10.1007/s00401 013 1088 7Document11 pagesArt:10.1007/s00401 013 1088 7TT WeiNo ratings yet
- Tenascin-C Promotes Tumor Cell Migration and Metastasis Through Integrin A9b1-Mediated YAP InhibitionDocument12 pagesTenascin-C Promotes Tumor Cell Migration and Metastasis Through Integrin A9b1-Mediated YAP InhibitionSayda DhaouadiNo ratings yet
- Leptin 1Document9 pagesLeptin 1vestibuleNo ratings yet
- Oral Oncology: SciencedirectDocument8 pagesOral Oncology: SciencedirectDenis OverlöperNo ratings yet
- 10 1038@nature13906Document14 pages10 1038@nature13906Diego Benitez RiquelmeNo ratings yet
- Influence of Morinda Citrifolia (Noni) On Expression of DNA Repair Genes in Cervical Cancer CellsDocument5 pagesInfluence of Morinda Citrifolia (Noni) On Expression of DNA Repair Genes in Cervical Cancer CellsAtika Nur NabilaNo ratings yet
- Li2021 - Article - PETCTImagingOf89Zr N Sucdf PemDocument10 pagesLi2021 - Article - PETCTImagingOf89Zr N Sucdf PemWSNo ratings yet
- Expression of A Breast-Cancer-Associated Protein (Ps2) in Human Neuro-Endocrine TumoursDocument5 pagesExpression of A Breast-Cancer-Associated Protein (Ps2) in Human Neuro-Endocrine TumoursSarly FebrianaNo ratings yet
- AtomicBomb Scid MiceDocument7 pagesAtomicBomb Scid MiceDavid MartínezNo ratings yet
- 1997 FullDocument15 pages1997 FullVignesh RavichandranNo ratings yet
- GBM Terapi PoliovirusDocument20 pagesGBM Terapi PoliovirusSafitri MuhlisaNo ratings yet
- Sciadv Abh2445Document14 pagesSciadv Abh2445Valen EstevezNo ratings yet
- DCE and DW-MRI Monitoring of Vascular Disruption Following VEGF-Trap Treatment of A Rat Glioma ModelDocument22 pagesDCE and DW-MRI Monitoring of Vascular Disruption Following VEGF-Trap Treatment of A Rat Glioma ModelDong DongNo ratings yet
- Ali Et Al-2016-Cancer ScienceDocument8 pagesAli Et Al-2016-Cancer ScienceTatiana Sanchez AlvarezNo ratings yet
- Retrovirus Producer Cells Encoding Antisense Vegf Prolong Survival of Rats With Intracranial GS9L GliomasDocument14 pagesRetrovirus Producer Cells Encoding Antisense Vegf Prolong Survival of Rats With Intracranial GS9L GliomasDong DongNo ratings yet
- Effect of Platelet-Derived Growth Factor-B On Renal Cell Carcinoma Growth and ProgressionDocument11 pagesEffect of Platelet-Derived Growth Factor-B On Renal Cell Carcinoma Growth and ProgressionOZGE DENIZ YESILNo ratings yet
- Lee 2012Document11 pagesLee 2012JA VelascoNo ratings yet
- STEM CELLS - 2009 - Jen - Suppression of Gene Expression by Targeted Disruption of Messenger RNA Available Options andDocument13 pagesSTEM CELLS - 2009 - Jen - Suppression of Gene Expression by Targeted Disruption of Messenger RNA Available Options andHAMNA ZAINABNo ratings yet
- Octreosc Vs FDGDocument7 pagesOctreosc Vs FDGLaura PredescuNo ratings yet
- Neuropilins Intumor Biology: Molecular PathwaysDocument5 pagesNeuropilins Intumor Biology: Molecular PathwaysdupuytrenNo ratings yet
- 1 1Document11 pages1 1Nejc KovačNo ratings yet
- Newton 2019Document9 pagesNewton 2019vmdcabanillaNo ratings yet
- 10 1016@j Jconrel 2019 12 052Document13 pages10 1016@j Jconrel 2019 12 052Oscar OsorioNo ratings yet
- Richardson 2012Document8 pagesRichardson 2012Abhilash VemulaNo ratings yet
- Diffusion-Weighted Imaging in The Follow-Up of Treated High-Grade Gliomas: Tumor Recurrence Versus Radiation InjuryDocument9 pagesDiffusion-Weighted Imaging in The Follow-Up of Treated High-Grade Gliomas: Tumor Recurrence Versus Radiation InjuryJheny LopezNo ratings yet
- Abstract BookDocument90 pagesAbstract BookPopescu RoxanaNo ratings yet
- 1 s2.0 S0360301619310193 MainDocument2 pages1 s2.0 S0360301619310193 MainStem EducationNo ratings yet
- 2021 Coating An Adenovirus With Functionalized AuNPDocument12 pages2021 Coating An Adenovirus With Functionalized AuNPDenisse IuliaNo ratings yet
- Next Generation Sequencing ArticleDocument3 pagesNext Generation Sequencing ArticleHareeshNo ratings yet
- L19 DNAtech Rev Genetics 2013 FDocument27 pagesL19 DNAtech Rev Genetics 2013 FjdazuelosNo ratings yet
- 2663 Full PDFDocument6 pages2663 Full PDFleilalargate_3780929No ratings yet
- Ceju 69 00726Document9 pagesCeju 69 00726IkaNo ratings yet
- Blastic Plasmacytoid Dendritic Cell NeoplasmeCurrent InsightsDocument10 pagesBlastic Plasmacytoid Dendritic Cell NeoplasmeCurrent InsightsPaúl Otañez MolinaNo ratings yet
- Reviews: DNA Methylation Profiling in The Clinic: Applications and ChallengesDocument14 pagesReviews: DNA Methylation Profiling in The Clinic: Applications and ChallengesVivi ZhouNo ratings yet
- Benitez-Ribas 2018Document9 pagesBenitez-Ribas 2018docadax848No ratings yet
- Flucke 2013Document8 pagesFlucke 2013FANIA PARRANo ratings yet
- Targeted Therapies for Lung CancerFrom EverandTargeted Therapies for Lung CancerRavi SalgiaNo ratings yet
- Operating Room Personnel OrganizationDocument4 pagesOperating Room Personnel OrganizationantihistamiiineNo ratings yet
- Hannahs CVDocument2 pagesHannahs CVabbief1No ratings yet
- Yasmeen Sitxfa002Document35 pagesYasmeen Sitxfa002ranu jain100% (1)
- Essay For TBIDocument7 pagesEssay For TBINatalie DrubachNo ratings yet
- Home Care Catalogue UK Edition 4 Web PDFDocument60 pagesHome Care Catalogue UK Edition 4 Web PDFmanuales tecnicosNo ratings yet
- Auricular Causative Diagnosis-Finding The Roots of Diseases: A Study of Clinical CasesDocument11 pagesAuricular Causative Diagnosis-Finding The Roots of Diseases: A Study of Clinical Casesyan92120No ratings yet
- Gouty GCPDocument48 pagesGouty GCPKylie GolindangNo ratings yet
- Formulation and Evaluation of A Syrup Based On BalDocument10 pagesFormulation and Evaluation of A Syrup Based On BalemilNo ratings yet
- HIV and YogaDocument3 pagesHIV and YogaAbhaya Raj UpadhayaNo ratings yet
- Tennessee Hazard Mitigation Plan 2018 FINALDocument388 pagesTennessee Hazard Mitigation Plan 2018 FINALAnonymous GF8PPILW5100% (1)
- Case Study APFDocument64 pagesCase Study APFJayselle ArvieNo ratings yet
- Pharmacology 3 IMPDocument14 pagesPharmacology 3 IMPYash TadseNo ratings yet
- Department of Pharmaceutical ChemistryDocument13 pagesDepartment of Pharmaceutical ChemistrySuvarna KiniNo ratings yet
- BIOBASE SCSJ-V Series User ManualDocument25 pagesBIOBASE SCSJ-V Series User Manualsoporte03No ratings yet
- Prep ColonosDocument13 pagesPrep ColonosKarlNo ratings yet
- ANGIOSARCOMA pdf3 PDFDocument9 pagesANGIOSARCOMA pdf3 PDFعبدالعزيز عماد محمودNo ratings yet
- Nestle AnalysisDocument20 pagesNestle AnalysisDiv Kabra100% (1)
- 675 MSDSDocument4 pages675 MSDSAhmed Emad AhmedNo ratings yet
- Clinical Surgical 1Document2 pagesClinical Surgical 1Ohana NanaNo ratings yet
- Head and NeckDocument21 pagesHead and NeckUday PrabhuNo ratings yet
- MRM Minutes 2014Document7 pagesMRM Minutes 2014maheshNo ratings yet
- What Has Psychotherapy Inherited From Carl RogersDocument4 pagesWhat Has Psychotherapy Inherited From Carl Rogerscris kalitoNo ratings yet
- Below Knee AmputationDocument6 pagesBelow Knee AmputationAmacayoNo ratings yet
- Say No To PlasticDocument28 pagesSay No To Plasticpavithra nirmalaNo ratings yet
- Staphylococcus Aureus: Antiseptic Resistance Gene Distribution Antimicrobial Agent of Susceptibilities andDocument8 pagesStaphylococcus Aureus: Antiseptic Resistance Gene Distribution Antimicrobial Agent of Susceptibilities andRisna IrvianiNo ratings yet
- P RM 01 Incident Reporting PolicyDocument44 pagesP RM 01 Incident Reporting PolicypatientsafetyNo ratings yet
- Analytical Exposition ExerciseDocument7 pagesAnalytical Exposition ExerciseECC SMAN 1 BaleendahNo ratings yet