Professional Documents
Culture Documents
Binary Phase Diagrams: Binary Phase Diagram For Completely Soluble System (Isomorphous Systems)
Binary Phase Diagrams: Binary Phase Diagram For Completely Soluble System (Isomorphous Systems)
Uploaded by
Thaya GanapathyOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Binary Phase Diagrams: Binary Phase Diagram For Completely Soluble System (Isomorphous Systems)
Binary Phase Diagrams: Binary Phase Diagram For Completely Soluble System (Isomorphous Systems)
Uploaded by
Thaya GanapathyCopyright:
Available Formats
BINARY PHASE DIAGRAMS
BINARY PHASE DIAGRAM FOR COMPLETELY SOLUBLE SYSTEM
(ISOMORPHOUS SYSTEMS)
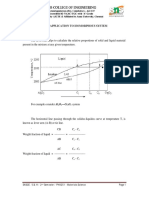
Binary phase diagrams involve temperature, pressure and composition as variables.
Then, a 3-dimensional representation is required for explaining the system, which is
complicated. In order to simplify, the binary phase diagrams are drawn at atmospheric
pressure. Also, pressure changes in vapor phase does not affect much the system, where,
high melting point components are present, hence, pressure variable can be ignored.
Then, the modified form of the phase rule equation for binary system is
F=C–P+1
Al2O3—Cr2O3, exhibits the simplest binary system where, they are completely
soluble with both liquid solubility and solid solubility.
The two components, namely alumina and chromia dissolve each other in all
proportions, both in liquid and solid states. There are only two phases in the above phase
diagram, liquid phase and solid phase.
The single phase regions are separated by a two-phase region (L+S). in this region,
both solid and liquid exist together. In all binary phase diagrams, the two phase region
SNSCE /S & H / 2nd Semester / PH8251 - Materials Science Page 1
SNS COLLEGE OF ENGINEERING, Coimbatore
separates the single phase regions. This is given by “1—2—1” rule, i.e., from single phase
(solid) to combined phase (L+S) to liquid phase (single phase).
The phase boundary between liquid phase and the two phase region is call liquidus.
The phase boundary between solid and two phase region is called solidus.
When only one phase is present, composition at particular temperature is given by
intersecting with composition axis. When two phases are present (L+S region), a horizontal
line called tie-line gives both liquid and solid composition.
For example, at particular temperature, T, Cl gives liquid composition, Cs gives solid
composition and Co gives overall composition.
SNSCE /S & H / 2nd Semester / PH8251 - Materials Science Page 2
You might also like
- Electricity and MagnetismDocument275 pagesElectricity and MagnetismThaya Ganapathy100% (3)
- Phase Rule and EquilibriaDocument23 pagesPhase Rule and EquilibriaRuchika RajaniNo ratings yet
- High-Pressure Fluid Phase Equilibria: Phenomenology and ComputationFrom EverandHigh-Pressure Fluid Phase Equilibria: Phenomenology and ComputationNo ratings yet
- PhysicalDocument79 pagesPhysicallividiveNo ratings yet
- Phase RuleDocument45 pagesPhase RuleSrinivasanNo ratings yet
- Makalah Termo Kleompok 1Document28 pagesMakalah Termo Kleompok 1Fadlan RasyidNo ratings yet
- UNIT-5 Phase EquilibriaDocument13 pagesUNIT-5 Phase EquilibriaALOK KUMARNo ratings yet
- Phase Diagrams NotesDocument8 pagesPhase Diagrams NotesRamesh PotnuriNo ratings yet
- Phase RuleDocument3 pagesPhase Rulekalyanivishal777No ratings yet
- Summarized Notes-Phase DiagramsDocument22 pagesSummarized Notes-Phase DiagramsLogan DabumaNo ratings yet
- Phase Rule Water & CO2systemsDocument9 pagesPhase Rule Water & CO2systemsAtul GautamNo ratings yet
- New Civil Module5 PDF NotesDocument18 pagesNew Civil Module5 PDF NotesDrMohan KumarNo ratings yet
- Unit 3-Cy19241Document28 pagesUnit 3-Cy19241Suresh Kumar A PNo ratings yet
- Unit-2 AnsDocument25 pagesUnit-2 AnsV.Nagaraju 19O17-M-O5ONo ratings yet
- A Phase Diagram in Physical ChemistryDocument12 pagesA Phase Diagram in Physical Chemistryreeta1981No ratings yet
- Phase DiagramsDocument80 pagesPhase DiagramsWilliams AkandiNo ratings yet
- Unit 1 MsDocument126 pagesUnit 1 MsHarishNo ratings yet
- Phase Diagram: From Wikipedia, The Free EncyclopediaDocument7 pagesPhase Diagram: From Wikipedia, The Free EncyclopediaPawan PatelNo ratings yet
- EMM NoteCLASS NOTESsDocument61 pagesEMM NoteCLASS NOTESsNallappan Rajj ANo ratings yet
- Lecture No. 5 Phase DiagramDocument32 pagesLecture No. 5 Phase DiagramarslanNo ratings yet
- Unit 7 PhaseruleDocument11 pagesUnit 7 Phaseruleengineeringchemistry100% (2)
- PDF Utils PrintDocument15 pagesPDF Utils PrintAvinash UpadhyayNo ratings yet
- Week 9 10 - 1 - IPE 2203-LecturesDocument86 pagesWeek 9 10 - 1 - IPE 2203-LecturesMD Al-AminNo ratings yet
- Critical So TemperatureDocument49 pagesCritical So TemperatureThakur Aditya PratapNo ratings yet
- Phase RuleDocument35 pagesPhase RuleABHINAVNo ratings yet
- Phase RuleDocument21 pagesPhase RuleelxnkitsNo ratings yet
- Phase Rule 1Document23 pagesPhase Rule 1SarojNo ratings yet
- 2-Phase RuleDocument18 pages2-Phase RuleSomesh Ashok BagalNo ratings yet
- Phase DiagramsDocument21 pagesPhase DiagramsJnrNo ratings yet
- Phase RuleDocument27 pagesPhase RulejaiminNo ratings yet
- Two ComponentsDocument19 pagesTwo ComponentsMumtaz AhmadNo ratings yet
- 5) Phase RuleDocument17 pages5) Phase RuleSHANJIDA ALI RIA100% (1)
- Capili Jefferson 9Document13 pagesCapili Jefferson 9Christian Al EncarnacionNo ratings yet
- Module 2 - 11-02-2020 Phase DiagramDocument57 pagesModule 2 - 11-02-2020 Phase DiagramsharadNo ratings yet
- Phase EquilibriumDocument32 pagesPhase EquilibriumSaif Khan100% (1)
- Binary Phase DiagramsDocument20 pagesBinary Phase DiagramsRaja AKNo ratings yet
- Phase RuleDocument29 pagesPhase RuleSruthi ShineyNo ratings yet
- Phase' Behavior in Binary and Ternary Water-Salt Systems at High Temperatures and PressuresDocument10 pagesPhase' Behavior in Binary and Ternary Water-Salt Systems at High Temperatures and PressuresPutra PratamaNo ratings yet
- Emm HandoutsDocument58 pagesEmm HandoutsDr.A.Maniram KumarNo ratings yet
- MS NotesDocument107 pagesMS Notessrinithims78No ratings yet
- Phase Diagrams: Example, Draw MacrostructureDocument54 pagesPhase Diagrams: Example, Draw MacrostructureMilan NaikNo ratings yet
- Propiedades Del ArgonDocument11 pagesPropiedades Del ArgonJuanVelaverdeNolazcoSalasNo ratings yet
- Bahir Dar Universtity Department of Material Science and EngineeringDocument5 pagesBahir Dar Universtity Department of Material Science and EngineeringKetemaw ZemeneNo ratings yet
- Phase Diagram 1Document23 pagesPhase Diagram 1Tony StarkNo ratings yet
- Phase EquilibriaDocument29 pagesPhase EquilibriaM.LOKESSH 18BME10029No ratings yet
- Phase Diagrams For Metallic SystemsDocument5 pagesPhase Diagrams For Metallic SystemsZesi Villamor Delos SantosNo ratings yet
- Phase Diagrams and SolidificationDocument13 pagesPhase Diagrams and Solidificationvenkatesh198910No ratings yet
- Cooling & Heating CurrveDocument2 pagesCooling & Heating CurrveManju KumariNo ratings yet
- Phase EquilibriaDocument11 pagesPhase EquilibriaNandhanNo ratings yet
- Phase Rule 1Document62 pagesPhase Rule 1arpitpandey494No ratings yet
- Written Report Chapter 9Document8 pagesWritten Report Chapter 9Engineer ZephyrusNo ratings yet
- Extending The Diagrams To Include The Solid PhaseDocument4 pagesExtending The Diagrams To Include The Solid PhaseawaisahmedkhanNo ratings yet
- Module V Phase & Chem EqbDocument26 pagesModule V Phase & Chem Eqbarhanbhandawat66No ratings yet
- Unit - III: Me432P: Material EnggDocument37 pagesUnit - III: Me432P: Material Engghans groupNo ratings yet
- Phase RuleDocument12 pagesPhase RuleFaria Sultana MimiNo ratings yet
- I. Title of Experiment: Equilibrium Phase Two Component II. Date of Experiment: Friday, 19 III. Purpose of ExperimentDocument7 pagesI. Title of Experiment: Equilibrium Phase Two Component II. Date of Experiment: Friday, 19 III. Purpose of ExperimentUtari Ika CahyaniNo ratings yet
- TwocomponentsystemDocument28 pagesTwocomponentsystemarun231187No ratings yet
- New Microsoft Office Word DocumentDocument17 pagesNew Microsoft Office Word DocumentmanojNo ratings yet
- Phase Equilibrium and CorrosionDocument5 pagesPhase Equilibrium and CorrosionPrabhanshu PareNo ratings yet
- Volumetric Properties of Pure Fluids: ThermodynamicDocument9 pagesVolumetric Properties of Pure Fluids: ThermodynamicLulav BarwaryNo ratings yet
- Session 5 Topic 5 Isomorphous Systems, The Tie-Line RuleDocument2 pagesSession 5 Topic 5 Isomorphous Systems, The Tie-Line RuleThaya GanapathyNo ratings yet
- Microstructure of A Lead-Tin AlloyDocument55 pagesMicrostructure of A Lead-Tin AlloyThaya GanapathyNo ratings yet
- PH8251-Material Science QN Bank ValliammaiDocument10 pagesPH8251-Material Science QN Bank ValliammaiThaya GanapathyNo ratings yet
- Modern Engineering Materials: Unit - VDocument11 pagesModern Engineering Materials: Unit - VThaya GanapathyNo ratings yet
- Physics For Bioscience: Rajalakshmi Engineering College (Autonomous) Thandalam, Chennai - 602105Document193 pagesPhysics For Bioscience: Rajalakshmi Engineering College (Autonomous) Thandalam, Chennai - 602105Thaya GanapathyNo ratings yet
- Unit I MCQDocument18 pagesUnit I MCQThaya GanapathyNo ratings yet
- Unit Iii Thermal Physics: Department of Science and Humanities MCQ For Regulations 2017Document19 pagesUnit Iii Thermal Physics: Department of Science and Humanities MCQ For Regulations 2017Thaya GanapathyNo ratings yet
- Topic 2.1 Oscillatory Motion: Department of Science and Humanities MCQ For Regulations 2017Document13 pagesTopic 2.1 Oscillatory Motion: Department of Science and Humanities MCQ For Regulations 2017Thaya GanapathyNo ratings yet
- Question Bank US03CPHY01 Unit1 To 4 Optics PMPDocument13 pagesQuestion Bank US03CPHY01 Unit1 To 4 Optics PMPThaya GanapathyNo ratings yet
- Solid State Physics 1 PGDocument4 pagesSolid State Physics 1 PGThaya GanapathyNo ratings yet
- 08 - Chapter 1Document19 pages08 - Chapter 1Thaya GanapathyNo ratings yet
- 10 - Chapter 1Document24 pages10 - Chapter 1Thaya GanapathyNo ratings yet
- Exam1 SolutionsDocument4 pagesExam1 SolutionsThaya GanapathyNo ratings yet
- 11 Fluids 1Document13 pages11 Fluids 1Thaya GanapathyNo ratings yet