Professional Documents
Culture Documents
PIN 07 2013 Onsite Fuel and Lubricant Quality Control by Portable FTIR Analysers
PIN 07 2013 Onsite Fuel and Lubricant Quality Control by Portable FTIR Analysers
Uploaded by
chiralicOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
PIN 07 2013 Onsite Fuel and Lubricant Quality Control by Portable FTIR Analysers
PIN 07 2013 Onsite Fuel and Lubricant Quality Control by Portable FTIR Analysers
Uploaded by
chiralicCopyright:
Available Formats
34 Analytical Instrumentation
On-site Fuel and
Lubricant Quality Control
by Portable FTIR Analysers
Dr. Christoph Wagner, Eralytics GmbH
Lohnergasse 3, A - 1210 Vienna, Austria
Tel: +43 1 890 50 33-0 • Email: wagner@eralytics.com • Web: www.eralytics.com
In today’s quickly moving business there is an increased demand for rapid quality control and condition
monitoring analysis of fuels and lubricants. is has sparked the development of portable analysers allowing on-
site analysis with performances comparable to laboratory instruments. eir dedicated design also enables
complex analysis to be carried out by non-experts, and the time from sampling to decision can be shortened
significantly compared to testing at an off-site laboratory.
Quality control of gasoline, Diesel fuel and jet fuel includes fuel composition analysis as well as FTIR instruments use an interferometer for the measurement of the sample spectrum. It splits the
the determination of fuel properties, such as Octane numbers (RON, MON) or Cetane index. For light emitted from a source into two equal parts by a beam splitter typically made of ZnSe. This
condition monitoring of lubricant oils, the degradation of the base oil due to oxidation, allows the measurement of spectra in the range of 1.5-18 µm (6660-550 cm-1) covering all
concentrations of additives and contaminants like water and ethylene glycol as well as properties relevant signals for fuel and lubricant analysis. The two light beams are directed onto a mirror
such as Total Acid Number (TAN) and Total Base Number (TBN) are key parameters. reflecting the IR light back towards the beam splitter where they recombine and are directed
For such applications infrared (IR) spectroscopy is the method of choice due to several reasons. towards the detector. If both mirrors are positioned at the same distance from the beam splitter
The IR spectrum contains information about the sample on a molecular level. Because each no phase shift between the two parts is introduced. During a measurement one of the two
molecular species has a unique signature, or fingerprint, in the IR part of the spectrum, it is mirrors moves away from the beam splitter at a constant speed. When the displacement of the
possible to determine the concentration of multiple species from a single spectrum. The mirror equals one quarter of the wavelength of the light, the combined beams are out of phase
evaluation and interpretation of IR spectra can be fully automated, enabling non-experts to resulting in destructive interference. In this case no light hits the detector. As the mirror is
rapidly carry out complex chemical analysis. Additionally, no sample preparation steps are needed scanned, the light intensity measured by the detector will show a modulation. The intensity
resulting in easy-to-operate analysers that only require a power connection for operation. The measured by the detector as a function of the mirror displacement is called an interferogram. For
measurement device itself, called Fourier transform infrared (FTIR) spectrometer, can be made very a monochromatic source, the interferogram will be a sine wave. For polychromatic light, which is
compact, stable and robust so that it can be deployed in the field. emitted by typical IR light sources exhibiting a broad wavelength distribution, the interferogram
will be the sum of sine waves of all frequencies present in the source. A Fourier transform of the
interferogram gives the spectrum. By referencing the spectrum of the sample to that of the
Principle of FTIR spectroscopy source, the (sample) absorption spectrum is obtained. The absorption spectrum will show a set of
Infrared radiation is an electromagnetic radiation with a wavelength between 0.7 µm and bands corresponding to frequencies where the sample absorbed light.
100 µm. This range is further divided in the Near-IR (NIR) range (wavelengths 0.7 µm to roughly 4 The spectral resolution, a parameter influencing the separation of two nearby bands in the final
µm), mid-IR (wavelengths 4 µm to roughly 20 µm), and far-IR (wavelengths 20 µm to 100 µm). spectrum, depends on how far the mirror is moved. For a spectral resolution of 4 wavenumbers
For analytical applications, mostly NIR and mid-IR spectroscopy are used. (cm-1) the mirror needs to be moved 0.25 cm whereas it has to be moved 0.5 cm for the higher
In IR spectroscopy, IR radiation is directed through the sample to be analysed and the intensity of spectral resolution of 2 cm-1.
the transmitted radiation is measured as a function of wavelength. When the wavelength of the
light matches the energy difference between the ground and excited vibrational state of a
molecule in the sample the light will be absorbed. The absorption of light is given by the Beer-
Considerations for building the ideal FTIR system
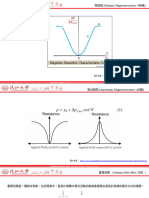
Lambert law, stating that the absorbance A is given by The goal for building an FTIR analyser is always to measure the desired samples with the best
signal to noise ratio (SNR) for the most accurate quantitative analysis. Therefore the careful
A=c*l*α(v), selection and attuning of the spectrometer’s components, such as light source, detector,
where c is the concentration, l is the sample path length and α(v) is a molecule specific constant. interferometer and electronics, are crucial for building high quality instruments.
From this equation we can conclude that A increases linearly with both sample concentration and Because absorption means attenuation of light, very high absorption means that little light
absorption of IR light. This implies that α(v) depends on wavelength. This is how concentrations
the sample path length. Generally, a molecule has multiple vibrational modes resulting in reaches the detector. This implies that very high absorption cannot be measured accurately. On
the other hand, if the absorption is too low, meaning that little light is absorbed by the sample,
of many different substances can be determined simultaneously by IR spectroscopy. the signal might be smaller than the noise of the instrument. In both cases the SNR will limit the
In mid-IR spectroscopy the fundamental molecular vibrations, which are typically narrow and accuracy of the qualitative analysis. The best SNR ratio is obtained when approximately 70% of
molecules with highly different α(v) or concentrations, the value of the path length (l) of the
substance specific signals, are excited. Moreover, a substance exhibits different vibrational modes the light is absorbed by the sample. It is therefore obvious from the formula A = c*l*α(v) that for
at different frequencies helping to identify individual compounds even if partial overlap of bands
occurs. Evaluating the whole spectrum enables the separation of chemically similar substances, system has to be adopted so that the absorption falls within a range where it can be measured
such as ethanol and methanol and quantitative analysis of both substances at the same time. with high accuracy.
Contrary to mid-IR spectroscopy, near-IR spectroscopy probes vibrational overtone and To be able to separate similar spectral features, it is necessary to measure the spectrum at a
combination transitions. These bands are typically much broader than in the mid-IR and only sufficient resolution. Figure 1 shows spectra from a methanol-ethanol mixture at different spectral
characteristic for some groups, such as CH or OH groups. The near-IR spectrum shows only a few resolutions (2, 4, 8 and 16 cm-1). If the spectral resolution is too low the two signals (one for
broad and highly overlapping bands. The lack of chemical selectivity in this spectral region makes methanol and one for ethanol) cannot be separated anymore. It becomes impossible to
it difficult to measure additives present at lower concentrations and the analysis of the spectra distinguish between them at a spectral resolution of around 16 cm-1. It can also be seen that the
relies heavily on chemometric methods requiring extensive calibration and validation. intensity of the signals decreases as the resolution is lowered. The spectral resolution not only
affects the signal strength but also the noise level. Figure 2 shows how the noise of the baseline
Due to the nature of near-IR signals all the information is present in the mid-IR spectrum as well.
increases going from a spectral resolution of 4 to 2 cm-1. For quantitative analysis the parameter
The mid-IR, however, has the advantage of more distinct features that exhibit higher signal
that needs to be optimised is the repeatability i.e. the variation between consecutive
intensities. Therefore mid-IR spectroscopy is better suited for quantitative analysis resulting in
measurements of the same sample. Figure 3 shows the repeatability as a function of resolution
lower limits of detection due to the higher signal intensities observed.
for MTBE (a gasoline additive). Higher spectral resolutions lead to an increased deviation of the
The aforementioned mid-IR signals can be measured in different ways. A cheap but limited way is measured absorbance deteriorating the repeatability of the measurement. These figures strikingly
the use of optical filters selecting only one of the various bands in the mid-IR range specific for illustrate that a compromise has to be made between repeatability and spectral separation of
one substance. Such analysers do not obtain a whole spectrum, which highly limits their use for overlapping features.
complex samples such as fuels. Nowadays the instrument of choice to measure complete spectra
Inspecting figure 4, showing a spectrum of gasoline, it becomes clear that no spectral
is the FTIR instrument which replaced the older technique of dispersive grating spectrometers.
information is lost at a spectral resolution of 4 cm-1. This is not surprising because the natural
JUNE / JULY 2013 • WWW.PETRO-ONLINE.COM
Analytical Instrumentation 35
linewidth of vibrations for molecules found in gasoline is larger than 10 cm-1. To resolve these
bands a resolution of 4 cm-1 as described in ASTM standards is sufficient. Increasing the spectral
resolution further only leads to a decrease of the signal to noise ratio as illustrated in figure 3.
Additionally spectra at a spectral resolution of 4 cm-1 are measured in half the time as at a
resolution of 2 cm-1 because the moving mirror has to move a shorter distance. This helps to
shorten analysis time and to increase sample throughput.
Fuel and lubricant analysis using FTIR spectroscopy
Fuels are complex mixtures of alkanes (paraffins), alkenes (olefins) and aromatics. Moreover, they
are blended with a wide range of additives to enhance combustion and exhaust emission
parameters. Therefore typically oxygenates are used such as MTBE, ethanol or methanol, which
can be readily monitored by IR spectroscopy according to ASTM D5845. Other components have
to be monitored because they are strictly regulated by authorities due to environmental and
health concerns. Benzene for example can be monitored according to ASTM D6277 and EN 238
using IR spectroscopy.
In recent years the increasing desire to use renewable fuel sources led to blending of bio-ethanol
to gasoline. FAME, an ester produced from vegetable oil, is often added as a CO2-neutral
component to Diesel fuel (ASTM D7806, EN 14078). All these components are present in modern
fuels in typically high concentrations. However, there are fuel additives such as MMT or 2-EHN
Figure 1: Spectra of a mixture of ethanol and methanol at different spectral resolutions which have a significant effect on fuel properties already at very low concentrations. 50 mg/l
MMT for example boosts the RON by typically 2.5 and 250 mg/l of 2-EHN the Cetane number by
1.7. It is therefore of high importance to measure these parameters as accurately as possible
alongside other additives that are present at much higher concentrations.
As described earlier, longer path lengths of the transmission cell lead to higher signals allowing
lower limits of detections. Components of high concentrations will lead to strong or even total
absorption of light if the path length is too long. To be able to measure additives present in
both high and low concentrations accurately, ERASPEC, an FTIR fuel analyser by eralytics, offers a
dual cell system. Typically, additives present in high concentrations like ethanol (up to 25 Vol%)
are measured in a 20 µm cell while additives present at low concentration like MMT (added in the
ppm range) are measured in a 100 µm cell.
Component concentrations, however, are not the only interesting values of fuels. There are
various parameters such as RON, MON, distillation properties, evaporation points and vapour
pressure, which are of high interest as well. Such fuel parameters cannot be pinned to specific
signals in a spectrum because they do not correlate with single fuel components. Using various
different signals and applying advanced statistical methods such parameters can be calculated
from a spectrum. For most accurate results it is not enough to measure the spectrum as
accurately as possible but the analyser has to use a spectral database for the correlation of the
parameters. Therefore the result also highly depends on the quality of the reference data, for
example the knock engine results for the octane numbers or the vapour pressure measured using
one of the available mini methods. To obtain reliable predictions using such a model the predicted
value also has to fall within the calibrated range of the model. If only samples with octane
numbers between 90 and 92 have been used for calibration, the analyser will not be capable to
Figure 2: Noise increase of the spectral baseline with higher spectral resolution calculate accurate results for gasoline with an octane number of 95. Additionally, due to the
complexity of fuels and their regional variations no overall database can be created. ERASPEC,
with several hundred installations worldwide, therefore offers comprehensive databases for
different regions in order to achieve most accurate results.
Another field of application of IR spectroscopy is the condition monitoring of lubricants. In
contrast to fuel analysis the main interest is to monitor chemical changes of the oil during use
affecting its performance. Parameters such as oxidation (ASTM D7414), nitration (ASTM D7624),
sulphation (ASTM D7415) and phosphate antiwear additive depletion (ASTM D7412) as well as
antioxidant depletion (ASTM D2668) can be readily monitored in the mid-IR spectrum. In
addition, IR offers a quick screening for various contaminants like water, glycol, fuels or soot
according to ASTM E2412. ERASPEC OIL also calculates total acid and base numbers using a
database spanning all relevant values of the sample range.
IR spectroscopy is a versatile tool for various analytical questions concerning fuels and lubricants.
Thorough design regarding the spectrometer itself combined with intelligent spectral analysis
using extensive databases is the key element for reliable fuel or lubricant analysis. ERASPEC and
ERASPEC OIL by eralytics are portable and rugged analysers offering tailor-made solutions for fuel
and lubricant analysis. They deliver precise analysis results in the lab as well as in the field.
Figure 3: Influence of spectral resolution on the repeatability of the measurement
Figure 4: Gasoline spectra at different spectral resolutions Figure 5: Portable ERASPEC analyser by eralytics (www.eralytics.com)
JUNE / JULY 2013 • WWW.PETRO-ONLINE.COM
You might also like
- Infrared SpectrosDocument59 pagesInfrared SpectrosMohammed Muzaffar MoinuddinNo ratings yet
- Handbook of Instrumental Techniques For Analytical CHemistry - Fran A.Settle PDFDocument728 pagesHandbook of Instrumental Techniques For Analytical CHemistry - Fran A.Settle PDFGuto Steinhorst100% (5)
- The Physics and Technology of Diagnostic Ultrasound: Study Guide (Second Edition)From EverandThe Physics and Technology of Diagnostic Ultrasound: Study Guide (Second Edition)No ratings yet
- Gyro-Compass: TG-8000 / 8500 Operator's ManualDocument96 pagesGyro-Compass: TG-8000 / 8500 Operator's ManualGeorgios Mariolis100% (1)
- FTIRDocument6 pagesFTIRAnubhav ShuklaNo ratings yet
- ASTM D7653 gas-phase-ftir-spectroscopy-introduction-BR52338Document8 pagesASTM D7653 gas-phase-ftir-spectroscopy-introduction-BR52338Treerat VacharanukrauhNo ratings yet
- Ir SpectrometersDocument10 pagesIr SpectrometersVinícius FavrettoNo ratings yet
- TN50674 E 0215M FT IR AdvantagesDocument3 pagesTN50674 E 0215M FT IR AdvantagesAliyyus Sya'niNo ratings yet
- 08 Chapter3 PDFDocument34 pages08 Chapter3 PDFprathaps1987No ratings yet
- Diapositivas Ing. MaterialesDocument44 pagesDiapositivas Ing. MaterialesnataliaNo ratings yet
- Pro CH-1Document7 pagesPro CH-1Barkhad MohamedNo ratings yet
- FT-IR vs. Dispersive Infrared Theory of Infrared Spectroscopy InstrumentationDocument2 pagesFT-IR vs. Dispersive Infrared Theory of Infrared Spectroscopy InstrumentationtulitahkayNo ratings yet
- Fourier Transform Infrared (FT-IR) Spectroscopy: Theory and ApplicationsDocument35 pagesFourier Transform Infrared (FT-IR) Spectroscopy: Theory and ApplicationsLeeyie LimNo ratings yet
- Fourier Transform Infrared Spectroscopy: Introduction ToDocument8 pagesFourier Transform Infrared Spectroscopy: Introduction ToLai LaiNo ratings yet
- Introduction To Fourier Transform Infrared SpectrometryDocument8 pagesIntroduction To Fourier Transform Infrared SpectrometryKwanchaiBuaksuntearNo ratings yet
- 2014 Ftir LabDocument10 pages2014 Ftir LabAnurak OnnnoomNo ratings yet
- IR QualitativeDocument4 pagesIR QualitativeredaelwanNo ratings yet
- Analytical ChemistryDocument7 pagesAnalytical ChemistrySamra ButtNo ratings yet
- Ft-Ir Spectroscopy Principle, Technique and MathematicsDocument7 pagesFt-Ir Spectroscopy Principle, Technique and MathematicsYisi LrNo ratings yet
- Introduction To FTIR SpectrosDocument12 pagesIntroduction To FTIR SpectrosĐào Văn HoạtNo ratings yet
- Spandana (19CH60R49) - FTIR LABDocument4 pagesSpandana (19CH60R49) - FTIR LABdiary dearNo ratings yet
- Handbook of Instrumental Techniques For Analytical Chemistry (F. Settle)Document230 pagesHandbook of Instrumental Techniques For Analytical Chemistry (F. Settle)Leandregg100% (1)
- IR InstrumentationDocument8 pagesIR InstrumentationVedika SeepersadNo ratings yet
- Gas Phase Ftir Spectroscopy Introduction BR52338Document9 pagesGas Phase Ftir Spectroscopy Introduction BR52338MelixaxNo ratings yet
- Chap2 IR LectureNote DRK 110929Document28 pagesChap2 IR LectureNote DRK 110929Aby JatNo ratings yet
- Chapter 2 Infrared Spectroscopy IRDocument28 pagesChapter 2 Infrared Spectroscopy IRAmeerRashidNo ratings yet
- Services Copy: Idaho State Police Forensic Laboratory Training Manual Infared SpectrophotometryDocument9 pagesServices Copy: Idaho State Police Forensic Laboratory Training Manual Infared SpectrophotometryMark ReinhardtNo ratings yet
- FTIRDocument25 pagesFTIRAnubhav ShuklaNo ratings yet
- Introduction Fourier Transform Infrared 1Document8 pagesIntroduction Fourier Transform Infrared 1Karim Khan DaddaNo ratings yet
- Fourier Transform Infra Red (FTIR) : Group 3 Dwi Yuliantiningsih Wifqul Laili Riski Ayu Candra Rina RukaenahDocument19 pagesFourier Transform Infra Red (FTIR) : Group 3 Dwi Yuliantiningsih Wifqul Laili Riski Ayu Candra Rina RukaenahWifqul LailyNo ratings yet
- Introduction Fourier Transform Infrared Spectroscopy br50555Document8 pagesIntroduction Fourier Transform Infrared Spectroscopy br50555ASSASNo ratings yet
- FT SpectrosDocument5 pagesFT SpectrosSaptarshi SenguptaNo ratings yet
- Infra Red Absorption SpectrosDocument18 pagesInfra Red Absorption SpectrosSukumar PaniNo ratings yet
- Hospital Management AppDocument12 pagesHospital Management AppAnu VinodNo ratings yet
- FTIR (Things We Need To Know)Document3 pagesFTIR (Things We Need To Know)Keith Jaszer AgarradoNo ratings yet
- Explainer - The Difference Between IR, FTIR, and ATR-FTIRDocument5 pagesExplainer - The Difference Between IR, FTIR, and ATR-FTIRArt AuthenticationNo ratings yet
- Color Wavelength NM Violet Blue Cyan Green Yellow Orange RedDocument4 pagesColor Wavelength NM Violet Blue Cyan Green Yellow Orange RedJhenard John Lansangan BeltranNo ratings yet
- FTIR: Introdution and Application To ForensicsDocument23 pagesFTIR: Introdution and Application To ForensicsLJ GicoleNo ratings yet
- 3.2 Optical Absorption and Emission SpectrosDocument22 pages3.2 Optical Absorption and Emission SpectrosAnnNo ratings yet
- EE471 F2016 Lec8Document21 pagesEE471 F2016 Lec8Ahmed RekikNo ratings yet
- Infrared SpectrosDocument17 pagesInfrared SpectrosLisbeth Roos RoosNo ratings yet
- Kuliah Infra MerahDocument110 pagesKuliah Infra Merahmuhlisun azimNo ratings yet
- FTIR SpectrosDocument7 pagesFTIR SpectrosI. Murali KrishnaNo ratings yet
- 2 PDFDocument7 pages2 PDFAnnisa KarimahNo ratings yet
- Fourier Transform InfraRed Spectroscopy 1Document22 pagesFourier Transform InfraRed Spectroscopy 1Uma kNo ratings yet
- Infra Red 1Document18 pagesInfra Red 1Sukumar PaniNo ratings yet
- XRF and FTIR PresentationDocument21 pagesXRF and FTIR PresentationusamaNo ratings yet
- Inductively Coupled PlasmaDocument14 pagesInductively Coupled PlasmaAnibal RiosNo ratings yet
- 1525950185module-2 Unit5 COM-IDocument8 pages1525950185module-2 Unit5 COM-ISimranNo ratings yet
- Class: Integrated Physics: 6 .: 18532030019: Incharge: Mrs. Anshul Uppal Topic: FTIR and Vibrational Analysis of LabdaneDocument23 pagesClass: Integrated Physics: 6 .: 18532030019: Incharge: Mrs. Anshul Uppal Topic: FTIR and Vibrational Analysis of LabdaneArthansh KumarNo ratings yet
- 2-UV-Vis Molecular SpectrometryDocument27 pages2-UV-Vis Molecular SpectrometryWahyuni EkaNo ratings yet
- Lab MannualDocument24 pagesLab MannualAnila Aftab100% (1)
- InfaredDocument6 pagesInfareddulararashmika00No ratings yet
- Shining a Light on Cool Standards: Infrared Spectroscopy Theory Explored.From EverandShining a Light on Cool Standards: Infrared Spectroscopy Theory Explored.No ratings yet
- Infrared and Raman Spectroscopy: Principles and Spectral InterpretationFrom EverandInfrared and Raman Spectroscopy: Principles and Spectral InterpretationRating: 3 out of 5 stars3/5 (1)
- Application of Spectral Studies in Pharmaceutical Product development: (Basic Approach with Illustrated Examples) First Revised EditionFrom EverandApplication of Spectral Studies in Pharmaceutical Product development: (Basic Approach with Illustrated Examples) First Revised EditionNo ratings yet
- Laser Metrology in Fluid Mechanics: Granulometry, Temperature and Concentration MeasurementsFrom EverandLaser Metrology in Fluid Mechanics: Granulometry, Temperature and Concentration MeasurementsNo ratings yet
- Multispectral Imaging: Unlocking the Spectrum: Advancements in Computer VisionFrom EverandMultispectral Imaging: Unlocking the Spectrum: Advancements in Computer VisionNo ratings yet
- Optical Sources, Detectors, and Systems: Fundamentals and ApplicationsFrom EverandOptical Sources, Detectors, and Systems: Fundamentals and ApplicationsNo ratings yet
- Plastics Identification Flow ChartDocument1 pagePlastics Identification Flow ChartchiralicNo ratings yet
- ISO Standards and Bias Pierce DrorDocument10 pagesISO Standards and Bias Pierce DrorchiralicNo ratings yet
- The World Clock - WorldwideDocument1 pageThe World Clock - WorldwidechiralicNo ratings yet
- Microsart Manifolds: The Filtration Stand That Adapts To Your NeedDocument8 pagesMicrosart Manifolds: The Filtration Stand That Adapts To Your NeedchiralicNo ratings yet
- Fungi: Candida Auris: A Review of Recommendations For Detection and Control in Healthcare SettingsDocument11 pagesFungi: Candida Auris: A Review of Recommendations For Detection and Control in Healthcare SettingschiralicNo ratings yet
- Simultaneous Estimation of Residual Solvents (Ethanol, Acetone, Dichloromethane and Ethyl Acetate) in Dosage Form by GC-HS-FIDDocument7 pagesSimultaneous Estimation of Residual Solvents (Ethanol, Acetone, Dichloromethane and Ethyl Acetate) in Dosage Form by GC-HS-FIDchiralicNo ratings yet
- A Review On Recent Developments in The Determination of Residual Solvents by Gas ChromatographyDocument9 pagesA Review On Recent Developments in The Determination of Residual Solvents by Gas ChromatographychiralicNo ratings yet
- And The Correct Answers Are:: You Got All Good Values: !!! You Are AnDocument3 pagesAnd The Correct Answers Are:: You Got All Good Values: !!! You Are AnchiralicNo ratings yet
- Grain and Processed Grain Solutions: The Complete Product RangeDocument8 pagesGrain and Processed Grain Solutions: The Complete Product RangechiralicNo ratings yet
- Candida Auris: A Brief ReviewDocument7 pagesCandida Auris: A Brief ReviewchiralicNo ratings yet
- Attune NXT Flow Cytometer: Efficient. Flexible. TransformativeDocument36 pagesAttune NXT Flow Cytometer: Efficient. Flexible. TransformativechiralicNo ratings yet
- Application Compare Sipper Cuvettes Cary-3500 Uv-Vis 5994-1951en Us AgilentDocument6 pagesApplication Compare Sipper Cuvettes Cary-3500 Uv-Vis 5994-1951en Us AgilentchiralicNo ratings yet
- 425-06 - Duo Sas 360 - UkDocument1 page425-06 - Duo Sas 360 - UkchiralicNo ratings yet
- Quality Control Solutions For The Feed IndustryDocument4 pagesQuality Control Solutions For The Feed IndustrychiralicNo ratings yet
- KAM LKF and PKF: Karl Fischer Moisture AnalyzerDocument22 pagesKAM LKF and PKF: Karl Fischer Moisture AnalyzerchiralicNo ratings yet
- Baths - Chillers - Resource GuideDocument8 pagesBaths - Chillers - Resource GuidechiralicNo ratings yet
- AN362Document2 pagesAN362chiralicNo ratings yet
- Bring The Benefits of Ultra Inert Liners To Your Whole LabDocument12 pagesBring The Benefits of Ultra Inert Liners To Your Whole LabchiralicNo ratings yet
- Quantifying The Impact of Operator Error in PipettingDocument1 pageQuantifying The Impact of Operator Error in PipettingchiralicNo ratings yet
- HPLC003 Adamas 10-2018-LRDocument20 pagesHPLC003 Adamas 10-2018-LRchiralicNo ratings yet
- SP10 - Automated Smoke Point: Smoke Point of Kerosene and Aviation Turbine Fuel ASTM D1322Document2 pagesSP10 - Automated Smoke Point: Smoke Point of Kerosene and Aviation Turbine Fuel ASTM D1322chiralicNo ratings yet
- CO Incubator: Resource GuideDocument8 pagesCO Incubator: Resource GuidechiralicNo ratings yet
- TRB 624Document2 pagesTRB 624chiralicNo ratings yet
- PDF Cecil BrochureDocument20 pagesPDF Cecil Brochurechiralic100% (1)
- From Inspiration To InnovationDocument4 pagesFrom Inspiration To InnovationchiralicNo ratings yet
- Natgas C Analyzer: With Additional He/H ChannelDocument15 pagesNatgas C Analyzer: With Additional He/H ChannelchiralicNo ratings yet
- Foods 09 01264Document15 pagesFoods 09 01264chiralicNo ratings yet
- Supplies For Former Varian GC Systems QRGDocument16 pagesSupplies For Former Varian GC Systems QRGchiralicNo ratings yet
- Teknokroma Capillary Columns: Trb-WaxDocument1 pageTeknokroma Capillary Columns: Trb-WaxchiralicNo ratings yet
- Task 1Document3 pagesTask 1Dhiviyansh Punamiya OT3 - 433No ratings yet
- Atomic Structure-DTS-1 Main (Archive) SolDocument2 pagesAtomic Structure-DTS-1 Main (Archive) SolHalfborn GundersonNo ratings yet
- Surface Roughness Analysis in Milling Machining Using Design of ExperimentDocument9 pagesSurface Roughness Analysis in Milling Machining Using Design of Experiment21P410 - VARUN MNo ratings yet
- BS-soil Classifications SlidesDocument4 pagesBS-soil Classifications SlidesEngr Ishfaque TunioNo ratings yet
- Generating FunctionsDocument13 pagesGenerating Functionscompiler&automataNo ratings yet
- 實驗報告1Document6 pages實驗報告1SuzanneNo ratings yet
- MFTMW Chapter 3 PROBLEM SOLVING AND REASONINGDocument17 pagesMFTMW Chapter 3 PROBLEM SOLVING AND REASONINGMarilou RepatoNo ratings yet
- Module Fizik Form 4 2013 Printing PotraitDocument404 pagesModule Fizik Form 4 2013 Printing PotraitMuis MuliaNo ratings yet
- Electromotive Force (E.m.f.) of The Battery. Like Potential DifferenceDocument9 pagesElectromotive Force (E.m.f.) of The Battery. Like Potential DifferenceAbdoulie JoofNo ratings yet
- GEO3701 Unit 3Document94 pagesGEO3701 Unit 3Asithandile Punqunqu MpayipheliNo ratings yet
- And Q Fixed Along The y Axis, Exert A NetDocument7 pagesAnd Q Fixed Along The y Axis, Exert A NetUjwal C UjwalNo ratings yet
- SMDDocument8 pagesSMDGeorge UrdeaNo ratings yet
- Supported Cone Roof (CS) Add.4Document21 pagesSupported Cone Roof (CS) Add.4skhurramq100% (2)
- Flexural Strength of Ferrocement PannelDocument44 pagesFlexural Strength of Ferrocement PannelAishwarya ShedgeNo ratings yet
- Homework 2Document1 pageHomework 2Nguyễn Hoàng Khánh LinhNo ratings yet
- Temperature Sensor NTC M12 Datasheet 51 en 2782569739pdfDocument3 pagesTemperature Sensor NTC M12 Datasheet 51 en 2782569739pdfJimena DriNo ratings yet
- Refinement of Timoshenko Beam Theory For Composite and Sandwich Beams Using Zigzag KinematicsDocument45 pagesRefinement of Timoshenko Beam Theory For Composite and Sandwich Beams Using Zigzag KinematicsPablo Silva VásquezNo ratings yet
- Review of Materials For Photothermal Energy ConversionDocument29 pagesReview of Materials For Photothermal Energy ConversionW BogaertsNo ratings yet
- Hdpe Pe80 Din 8074 / Iso 4427Document79 pagesHdpe Pe80 Din 8074 / Iso 4427Steve WanNo ratings yet
- Experimental Analysis of Fibre Reinforced Concrete Using AnsysDocument30 pagesExperimental Analysis of Fibre Reinforced Concrete Using AnsysShivaNo ratings yet
- Sheet 2 Properties of Pure SubstancesDocument2 pagesSheet 2 Properties of Pure SubstancesZainhumNo ratings yet
- Effective Nuclear ChargeDocument3 pagesEffective Nuclear Chargekalloli100% (1)
- A Barrel Straightness Measurement System For Medium CaliberDocument18 pagesA Barrel Straightness Measurement System For Medium CaliberMian Muhammad JazibNo ratings yet
- Vtu No. ECE/Exam Cell/002Document2 pagesVtu No. ECE/Exam Cell/002SrivalliNo ratings yet
- Four-Tier Diagnostic Test With Certainty of Response Index On The Concepts of FluidDocument10 pagesFour-Tier Diagnostic Test With Certainty of Response Index On The Concepts of FluidAlimatu FatmawatiNo ratings yet
- Mathematics Junior Secondary School (Jss 3) First TermDocument8 pagesMathematics Junior Secondary School (Jss 3) First TermAdepoju PrimeNo ratings yet
- Wa0046Document185 pagesWa0046Christian KelechiNo ratings yet
- Dat E: 23/04/2015 Annual 2015 Examinat Ion Result SDocument1 pageDat E: 23/04/2015 Annual 2015 Examinat Ion Result SRsp SrinivasNo ratings yet
- Two-Layer Obstacle Collision Avoidance With Machine Learning For More Energy-Efficient Unmanned Aircraft TrajectoriesDocument16 pagesTwo-Layer Obstacle Collision Avoidance With Machine Learning For More Energy-Efficient Unmanned Aircraft TrajectoriesMarcelo RodriguesNo ratings yet