Professional Documents
Culture Documents
Vaccine Development Infographic
Uploaded by
JuvvilyynGraceMahomatCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Vaccine Development Infographic
Uploaded by
JuvvilyynGraceMahomatCopyright:
Available Formats
vaccine
development
What is a vaccine?
Vaccines are biological products that generate vaccine history
acquired immunity to specific infectious diseases. The history of vaccine research is rich
with scientific breakthroughs.
1700s 1798 Edward Jenner coins the terms “vaccine” and
vaccines are saving lives “vaccination” as part of his work on a smallpox
vaccine.
The principal of vaccination is to train the immune 1800s 1800 Smallpox vaccination becomes
system to recognize and react to an infectious commonplace.
agent, and to induce an effective immune response. 1870s-80s Louis Pasteur develops first live bacterial
This ensures that the individual is protected against vaccine for chicken cholera and the first
the pathogen in the future. live viral vaccine for rabies.
1900s 1918 Spanish flu pandemic kills 25-50 million
Vaccines are critical in the prevention and worldwide.
control of infectious disease outbreaks. 1926-28 Developmet of tuberculosis, diphtheria,
tetanus and whooping cough vaccines.
The World Health Organization (WHO) states
that vaccines now prevent more than 20 1945 An inactivated influenza vaccine is licensed
in the US.
life-threatening diseases.
1950-60 First combination vaccines developed (DTP
Immunization prevents 2-3 million deaths – diphtheria, tetanus and whooping cough).
per year.
1952 Nearly 60,000 cases of polio are documented
in the US.
1955 First polio vaccine pioneered by Jonas Salk
is licensed in the US.
1961 An orally administered polio vaccine
The potential cost of developed by Albert Sabin is licensed in
the US.
illness avoided due to 1963 Measles vaccine is licensed in the US.
$63B
vaccinations from 2011-2020: 1974 Meningococcal polysaccharide vaccine is
licensed in the US.
1980 Smallpox is the first infectious disease
eradicated by vaccination.
1986 First genetically engineered vaccine
(hepatitis B) is developed.
2000s 2000s Measles and rubella are no longer endemic in
the US.
2006 Human papillomavirus vaccine (HPV) is
licensed in the US.
2009 Vaccines against 1009 H1N1 pandemic
strain and high-dose influenza vaccine are
licensed in the US. More than 12,000
Americans died during the H1N1
pandemic.
Haemophilus Pneumococcal
infections Rotavirus
influenzae type b
$27 B $24 B $12 B
Understanding the different types of vaccines
There are various different types of vaccines that are utilized to
fight against different infectious pathogens. Examples include:
Inactivated Toxoid Live-attenuated
Uses a dead form of A toxin produced A weakened form
a pathogen that has by the pathogen of the antigen that
been inactivated that causes a causes a disease is
using chemicals, disease is used. used to generate
heat or radiation. an immune
response.
Subunit, polysaccharide, conjugate mRNA
and recombinant Messenger ribonucleid acid (mRNA)
A certain piece of a pathogen, such as encoding a particular viral protein
a sugar, protein or capsid, is used to induces endogenous production of
generate an immune response. the protein in the recipient's cells.
vaccine development
There are several key stages involved in the development and approval of a new vaccine.
The specifics of each stage in this pipeline will differ according to the type of vaccine that is being developed.
Po on rc
st ito h
m sea
Ba
Pr
Cl
Re ta
-a rin
is
e-
in
re
si
gu ine
ob
co
pp g
ic
c
cl
la d
ve
al
re
ro &
in
to
ic
st
r
s
va
y
ea
ry
al
ud
l
rc
ap
st
ie
h
ud
pr
s/
ov
ie
tr
s
ia
al
ls
Phase: 1 2 3 4
Safety Efficacy Safety & Safety monitoring for
efficacy serious adverse events
Varies 2-4 years 6-8 years Approx 3 years Continuous
Traditional vs "new-era" vaccines
Under typical circumstances, the development of a new vaccine can take 10-15 years to
complete. In response to the COVID-19 global pandemic, this process has been accelerated
by carrying out specific stages in parallel with one another.
One outcome of this acceleration has been the rapid manufacturing, testing and emergency
use authorization (EUA) of mRNA-based vaccines. RNA-based vaccines are often referred to
as "new-era" or "next-generation" vaccines.
Note: Although the COVID-19 vaccines are the first examples authorized for human use,
scientists have been studying and manufacturing RNA-based vaccines for many years for
their potential use against infectious diseases and cancer.1
How does the development and
manufacturing of traditional vs mRNA-based
vaccines compare?
Let's take a look. This schematic summarizes the upstream and downstream production of
the cell-based inactivated influenza vaccine.2
WHO candidate virus
Seed virus
preparation
Virus infection
Preculture
High density
culture
Virus propagation
Filling Centrifugation DNA removal
The vaccine is filled into
a vial or syringe
All components of the
final vaccine are Whole virus Virus inactivation
combined uniformly
Split vaccine
Formulation Polishing
Virus disruption
Subunit vaccine
Manufacturing mRNA vaccines
The mRNA is then
Genome
sequencing
AATGCGCTA
The sequence corresponding
to the mRNA is inserted into a
3 purified through
methods such as
Target CGCTACGAT chromatography to
pathogen CGACTCGAT plasmid within a cell. The
CAGCTAGCA plasmid is placed in a remove enzymes,
TTCGATCGA
reactor where enzymatic remaining nucleotides
reactions trigger the and defective mRNA.
synthesis of the mRNA.
1 2
The mRNA encoding
a specific antigen of the
infectious pathogen is
created from a DNA
template. This DNA
sequence can be shared
globally computer 4
systems in an instant.
mRNA delivery: As mRNA is
unstable, scientists are working on
various methods to encapsulate it
for delivery into the body.
Examples of delivery methods being explored are:3
Lipid-based Polymer-based Peptide-based Virus-like Cationic Naked mRNAs Dendritic cell-based
delivery delivery delivery replicon particle nanoemulsion mRNA vaccines
Challenges in vaccine development and manufacturing
Due to the inherent variability, development and manufacturing of a vaccine is exceptionally challenging. Here is an
overview of the key challenges at the global scale:
Viral mutations: As
seen with the
SARS-CoV-2 virus,
Vaccine development pathogens can
is an expensive mutate and new
process – if a strains evolve,
candidate fails it can which can mean
have huge financial redevelopment of
repercussions. already distributed
vaccines.
Manufacturing
requires highly
Failure in supply chains of skilled and
essential resources such as trained
culture media, filtration personnel.
equipment or vials can stall
manufacturing.
Knowledge on infectious
exposures of the intended
Vaccines are vaccine recipients required.
harder to
standardize
compared to
other biologics.
Low success rate Require large clinical
of early-phase trials to evaluate
development. immunogenicity and
safety in humans.
Challenges in vaccine distribution
There are also challenges specific to the distribution of a vaccine. In the case of the COVID-19 global pandemic, the
key difficulties that have been encountered include:
Communication
Racial and ethnic and trust
disparities
Funding
Federal, state,
Coverage and local roles
Supply and and costs
logistics
Future perspectives
The COVID-19 global pandemic has demonstrated that it is possible to manufacture and distribute a vaccine at
unprecedented speeds. Lessons learnt from this experience include the flexibility of mRNA vaccine platforms and the
value of single-use technologies that enable fast repurposing of manufacturing facilities. Continuous investment in
Tools strategies expertise
that enable the expansion of manufacturing capabilities will be essential in the future of
vaccine development and manufacturing.
Sponsored by
References:
1. Pardi N, Hogan MJ, Porter FW, Weissman D. mRNA vaccines — a new era in vaccinology. Nat Rev Drug Discov. 2018;17(4):261-279. doi: 10.1038/nrd.2017.243.
2. Gomez PL, Robinson JM. Vaccine Manufacturing. In: Plotkin SA, Orenstein WA, Offit PA, Edwards KM, eds. Plotkin’s Vaccines. 7 ed. Elsevier; 2018:51-60.e1. doi: 10.1016/B978-0-323-35761-6.00005-5.
3. Verbeke R, Lentacker I, De Smedt SC, Dewitte H. Three decades of messenger RNA vaccine development. Nano Today. 2019;28:100766. doi: 10.1016/j.nantod.2019.100766.
You might also like
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- Christ Be Our LightDocument2 pagesChrist Be Our LightJuvvilyynGraceMahomatNo ratings yet
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- Role and Function of Nucleotides in Infant Nutrition: Julio J. Boza and Olga Martinez-AugustinDocument20 pagesRole and Function of Nucleotides in Infant Nutrition: Julio J. Boza and Olga Martinez-AugustinJuvvilyynGraceMahomatNo ratings yet
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Sars-Cov 2Document12 pagesSars-Cov 2JuvvilyynGraceMahomatNo ratings yet
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- 1888 Fathers of The Catholic ChurchDocument392 pages1888 Fathers of The Catholic ChurchArmando CottimNo ratings yet
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- Chemical Biology 03 Blood: Biomolecular Structure Lecture 3: 9/13/10Document23 pagesChemical Biology 03 Blood: Biomolecular Structure Lecture 3: 9/13/10JuvvilyynGraceMahomatNo ratings yet
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (399)
- WoodsDocument6 pagesWoodsJuvvilyynGraceMahomatNo ratings yet
- Chapter 18: Carbohydrates: Instructional ObjectivesDocument25 pagesChapter 18: Carbohydrates: Instructional ObjectivesSalma NurrabbaniNo ratings yet
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (73)
- Confocal Scanning MicrosDocument3 pagesConfocal Scanning MicrosJuvvilyynGraceMahomatNo ratings yet
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Biochemistry of Carbohydrates: Part IDocument90 pagesBiochemistry of Carbohydrates: Part IJuvvilyynGraceMahomatNo ratings yet
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- FLU Pandemic: November March May July September November January March May JanuaryDocument1 pageFLU Pandemic: November March May July September November January March May JanuaryJuvvilyynGraceMahomatNo ratings yet
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Experiment 8 Lab Manual PDFDocument2 pagesExperiment 8 Lab Manual PDFJuvvilyynGraceMahomat100% (2)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- Lessons From The Call of JeremiahDocument2 pagesLessons From The Call of JeremiahJuvvilyynGraceMahomat100% (1)
- Nuhra Region4 Agenda PDFDocument32 pagesNuhra Region4 Agenda PDFJing CruzNo ratings yet
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (344)
- 24 APJABSS Feb BRR755 Social-Sciences-250-260 PDFDocument11 pages24 APJABSS Feb BRR755 Social-Sciences-250-260 PDFJuvvilyynGraceMahomatNo ratings yet
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- SummaryaDocument8 pagesSummaryaIna AguilarNo ratings yet
- Copperplate Practice Sheet W Letters PDFDocument1 pageCopperplate Practice Sheet W Letters PDFJuvvilyynGraceMahomatNo ratings yet
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- Sample Food Business Plan Oklahoma StateDocument61 pagesSample Food Business Plan Oklahoma StateJuvvilyynGraceMahomatNo ratings yet
- Final Simp Gothic PDFDocument8 pagesFinal Simp Gothic PDFJuvvilyynGraceMahomatNo ratings yet
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- Wellness and Lifestyles Questionnaire PDFDocument3 pagesWellness and Lifestyles Questionnaire PDFJuvvilyynGraceMahomatNo ratings yet
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- DR701SD14 PDFDocument7 pagesDR701SD14 PDFJuvvilyynGraceMahomatNo ratings yet
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- Sample Food Business PlanDocument17 pagesSample Food Business PlanTapan Jain100% (1)
- Copperplate Practice Sheet PDFDocument1 pageCopperplate Practice Sheet PDFJuvvilyynGraceMahomatNo ratings yet
- Idealism PlatoDocument10 pagesIdealism PlatoJuvvilyynGraceMahomat100% (1)
- Modern Calligraphy Practice Sheet Lowercase PDFDocument2 pagesModern Calligraphy Practice Sheet Lowercase PDFJuvvilyynGraceMahomat100% (1)
- Crayola Lowercase Brush Practice Sheet PDFDocument4 pagesCrayola Lowercase Brush Practice Sheet PDFJuvvilyynGraceMahomatNo ratings yet
- SOPHIA The RobotDocument9 pagesSOPHIA The RobotJuvvilyynGraceMahomatNo ratings yet
- SISSY PH - Frequently Asked Questions (FAQ) : PDFDocument6 pagesSISSY PH - Frequently Asked Questions (FAQ) : PDFJuvvilyynGraceMahomatNo ratings yet
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (120)
- Diagrams of The Renewable Energy ResourcesDocument11 pagesDiagrams of The Renewable Energy ResourcesJuvvilyynGraceMahomatNo ratings yet
- 9arts GR 10 LM - Qtr4 (8 Apr 2015)Document18 pages9arts GR 10 LM - Qtr4 (8 Apr 2015)John Carlo Benitez100% (2)
- Approach To Jaundice (1) .PPT ALAWAJIDocument28 pagesApproach To Jaundice (1) .PPT ALAWAJIayuNo ratings yet
- A Cross-Sectional Study of The Prevalence ofDocument9 pagesA Cross-Sectional Study of The Prevalence ofKurnia pralisaNo ratings yet
- Pathology Polycystic Kidney DiseaseDocument4 pagesPathology Polycystic Kidney DiseaseOnyedika EgbujoNo ratings yet
- Chain of InfectionDocument2 pagesChain of Infectionapi-251976647No ratings yet
- Role of Biotechnology in Improving Human HealthDocument7 pagesRole of Biotechnology in Improving Human HealthdeepaksagarNo ratings yet
- Coronavirus (Covid-19)Document2 pagesCoronavirus (Covid-19)Shompa ShikderNo ratings yet
- Kaya Kalp House Keeping StandardsDocument131 pagesKaya Kalp House Keeping StandardsAnuragMishraNo ratings yet
- AMOEBIASISDocument36 pagesAMOEBIASISKhei Laqui SNNo ratings yet
- Outbreak Student Worksheet AnswersDocument9 pagesOutbreak Student Worksheet AnswersArlie Espeña DucducanNo ratings yet
- Chiron & NAT Introduction KL 29APR08Document35 pagesChiron & NAT Introduction KL 29APR08api-3750327100% (1)
- Interpret HbA2 With Caution Per DR Barbara BainDocument50 pagesInterpret HbA2 With Caution Per DR Barbara Baindamadol100% (1)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- Konsep Dasar Hais Dan Program PpiDocument38 pagesKonsep Dasar Hais Dan Program PpibotaNo ratings yet
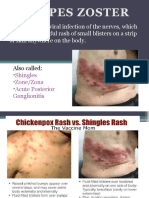
- Herpes ZosterDocument12 pagesHerpes ZosterCharlz ZipaganNo ratings yet
- The Family Enterobactericeae PDFDocument62 pagesThe Family Enterobactericeae PDFAngelica Alejandra De La Torre AnayaNo ratings yet
- Colouratlasofpoultrydiseases PDFDocument126 pagesColouratlasofpoultrydiseases PDFthanh ba matNo ratings yet
- Intratect 50 G L SPC 05 02 2016 - UkDocument12 pagesIntratect 50 G L SPC 05 02 2016 - UkCarla CaldararuNo ratings yet
- SZL2111 PDFDocument153 pagesSZL2111 PDFoscarNo ratings yet
- LimfadenopatiDocument8 pagesLimfadenopatiZulkarnain PrakosoNo ratings yet
- Diversity of Microorganisms 1 - AcellularDocument39 pagesDiversity of Microorganisms 1 - AcellularCarl Elexer Cuyugan Ano100% (1)
- Wood's Lamp - IjdvlDocument5 pagesWood's Lamp - IjdvlKunal PaulNo ratings yet
- Module SC f5 p2 CHAPTER 1Document10 pagesModule SC f5 p2 CHAPTER 1norasiah6500No ratings yet
- Biotics: Biotics Refers To BacteriaDocument14 pagesBiotics: Biotics Refers To BacteriaDinaNo ratings yet
- CestodesDocument86 pagesCestodesPidchayathanakorn Paemika0% (1)
- Aranjarea BibliografieiDocument12 pagesAranjarea BibliografieialinaNo ratings yet
- Design Poster - Group 11 - A3Document1 pageDesign Poster - Group 11 - A3HahahahaNo ratings yet
- Mission Urinalysis Strips InsertDocument1 pageMission Urinalysis Strips Insertquirmche70No ratings yet
- Biotechnology and Gene Tech Questions F215Document74 pagesBiotechnology and Gene Tech Questions F215tigerdentistNo ratings yet
- Restating Pharses and Sentences by Sigit Riyadi (Autosaved)Document26 pagesRestating Pharses and Sentences by Sigit Riyadi (Autosaved)srNo ratings yet
- Bovine Respiratory DiseaseDocument4 pagesBovine Respiratory DiseaseDamir BrankovicNo ratings yet
- Prometric High-Yield NOTES PDFDocument135 pagesPrometric High-Yield NOTES PDFDr-Jahanzaib Gondal100% (3)