Professional Documents
Culture Documents
Referensi Tesis Spesialis Grayson
Referensi Tesis Spesialis Grayson
Uploaded by
florensiaCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Referensi Tesis Spesialis Grayson
Referensi Tesis Spesialis Grayson
Uploaded by
florensiaCopyright:
Available Formats
Clinical Review & Education
JAMA Otolaryngology–Head & Neck Surgery | Review
Contemporary Classification of Chronic Rhinosinusitis
Beyond Polyps vs No Polyps
A Review
Jessica W. Grayson, MD; Claire Hopkins, MD; Eri Mori, MD; Brent Senior, MD; Richard J. Harvey, MD, PhD
Supplemental content
IMPORTANCE Chronic rhinosinusitis (CRS) is a broadly defined process that has previously
been used to describe many different sinonasal pathologic conditions from odontogenic
sinusitis and allergic fungal sinusitis to the more contemporary definition of broad
inflammatory airway conditions. Previous classification systems have dichotomized these Author Affiliations: Department of
Otolaryngology–Head and Neck
conditions into CRS with nasal polyps and CRS without nasal polyps. However, clinicians are Surgery, University of Alabama at
learning more about the inflammatory subtypes of CRS, which can lead to improved delivery Birmingham (Grayson); Department
and effectiveness of treatment. of Otolaryngology, Guy’s Hospital,
London, United Kingdom (Hopkins);
OBSERVATIONS In clinical practice, treatment decisions are often based on observable Department of Otorhinolaryngology–
findings, clinical history, presumed disease, and molecular pathophysiologic characteristics. Head and Neck Surgery, The Jikei
University School of Medicine, Tokyo,
A proposed classification system is simple and practical. It proposes that the functional Japan (Mori); Department of
anatomical compartments involved create the first level of separation into local and diffuse Otolaryngology–Head and Neck
CRS, which are usually unilateral or bilateral in distribution. Diffuse does not imply Surgery, University of North Carolina
“pansinusitis” but simply that the disease is not confined to a known functional anatomical at Chapel Hill (Senior); Rhinology
and Skull Base Research Group,
unit. This classification takes into account whether local anatomical factors are associated St Vincent’s Centre for Applied
with pathogenesis. Then the inflammatory endotype dominance is separated into a type 2 Medical Research, University of
skewed inflammation, as this has both causal and treatment implications. The non–type 2 New South Wales, Sydney,
New South Wales, Australia (Harvey);
CRS encompasses everything else that is not yet known about inflammation and may change
Department of Otolaryngology,
over time. The phenotypes or clinical examples are CRS entities that have been described and Faculty of Medicine and Health
how they align with this system. Sciences, Macquarie University,
Sydney, New South Wales, Australia
CONCLUSIONS AND RELEVANCE Although research continues to further define the subtypes of (Harvey).
CRS into phenotypes and endotypes, the proposed classification system of primary CRS by Corresponding Author: Jessica W.
anatomical distribution and endotype dominance allows for a pathway forward. Grayson, MD, Department of
Otolaryngology–Head and Neck
Surgery, University of Alabama at
JAMA Otolaryngol Head Neck Surg. doi:10.1001/jamaoto.2020.1453 Birmingham, 1720 Second Ave S,
Published online July 9, 2020. FOT 1155, Birmingham, AL 35294
(jessica.w.grayson@gmail.com).
C
hronic rhinosinusitis (CRS) is a broadly defined process that tory sinonasal disease have largely been associated with research
has previously been used to describe many different sino- in the lower respiratory tract and applied to the upper airway given
nasal pathologic conditions from odontogenic sinusitis and the unified airway theory.6,7 Determination of endotypes origi-
allergic fungal sinusitis to the more contemporary definition of broad nated with asthma and has been further extrapolated to the man-
inflammatory airway conditions. Previous classification systems have agement of CRS.6,8-11 An endotype in CRS is defined as a distinct
dichotomized these conditions into CRS with nasal polyps and CRS pathophysiologic mechanism that may be identified by specific
without nasal polyps.1,2 Although this classification system is simple, biomarkers.4,5,12 The delineation of these endotypes has the ability
it relies heavily on clinical findings for diagnosis without account- to streamline and improve treatment in patients with CRS as well
ing for the pathophysiologic differences between CRS subtypes and as allow for better analysis of treatment results.
therefore may not be able to assess response to different treat- Although all subtypes and causes of CRS have previously been
ment regimens. Furthermore, inflammatory nasal polyps are seen grouped together on the basis of the presence or absence of pol-
as an end point of inflammation and fibrin deposition rather than yps, we propose instead that CRS may be indicative of primary or
specific for any underlying pathophysiologic cause.3,4 Current thera- secondary sinus pathologic characteristics. These subtypes (pri-
pies are now chosen on the basis of presumed underlying causal mary vs secondary) can be further delineated by their anatomical
pathogenesis or endotypic subtype of CRS. Many different pheno- locations and endotype or inflammatory predominance (or causal
types of CRS, as well as the inflammatory disorders underlying them, mechanism). These further classifications ultimately result in the con-
have been proposed.5 stellation of a phenotype with clinical and radiographic findings. The
In clinical practice, treatment decisions are often based on ob- classification is a proposed system, based on our rhinologic prac-
servable findings, clinical history, presumed disease, and molecu- tices, that provides a framework in which to move forward and pro-
lar pathophysiology. These treatment algorithms used in inflamma- vide further data to support such a new classification system and
jamaotolaryngology.com (Reprinted) JAMA Otolaryngology–Head & Neck Surgery Published online July 9, 2020 E1
© 2020 American Medical Association. All rights reserved.
Downloaded From: https://jamanetwork.com/ by a Glasgow University Library User on 07/10/2020
Clinical Review & Education Review Contemporary Classification of Chronic Rhinosinusitis
Figure 1. Classification of Primary Chronic Rhinosinusitis (CRS)
Anatomical Endotype Clinical examples and
distribution dominance phenotypes
Type 2 AFRS
Localized
(unilateral)
OMC
Non-type 2 Isolated frontal
Isolated sphenoid
Primary CRS
CCAD
Type 2 eCRS
AFRS
Diffuse
(bilateral)
Non-eCRS
Poor corticosteroid
Non-type 2
response
Older or smoker
The functional anatomical compartments involved create the first level of separation into localized and diffuse. These compartments are usually unilateral or
bilateral in distribution. Diffuse does not imply pansinusitis but simply that the disease is not confined to a known functional anatomical unit, taking into account
whether local anatomical factors are associated with pathogenesis. Then the inflammatory endotype dominance separates into a type 2 skewed inflammation
because this delineation has both causal and treatment implications. The non–type 2 classification encompasses everything else not yet known about inflammation
and may change over time. The phenotypes or clinical examples are CRS entities that have been described and how they align with this system. AFRS indicates
allergic fungal rhinosinusitis; CCAD, central compartment atopic disease; eCRS, eosinophilic CRS; and OMC, ostiomeatal complex.
to evolve beyond the presence or absence of polyps. Although this conditions, although there may be circumstances in which a local-
system has not been fully tested, it provides a working classifica- ized or discrete process may occur synchronously on both sides, al-
tion on which we can continue to build. though it would be very uncommon. Surgery is likely to be a part of
We propose a CRS classification that allows for integrating con- the first-line management of localized CRS.
temporary scientific knowledge on the known causes of CRS, which
is based on anatomical distribution and the endotype dominance or Diffuse
mechanisms that produce previously described phenotypes of CRS. Diffuse airway inflammation describes a broader inflammatory dis-
The proposed classification system is integral in the European order that may affect both the upper and lower airways. Involve-
Position Paper on Rhinosinusitis 2020.13 ment of the sinuses may be patchy or diffuse on results of radio-
logic assessment and does not have to have complete opacification
of all sinuses but will not be limited by functional sinonasal units or
spaces. There will be variability in the degree to which the upper and
Primary CRS
lower airway are involved in a single patient, as one part of the air-
Primary CRS is contemporarily defined as a primary inflammatory dis- way may dominate the clinical presentation. It is common that some
order of “the airway” or respiratory system (Figure 1). Patients are de- degree of disease often exists across the entire airway. It is impor-
finedashavingprimaryCRSiftheyhaveadisorderthatislimitedtotheir tant to delineate whether or not the patient has lower airway symp-
airway or respiratory system only. Thus, patients who have CRS in the toms, as these symptoms may differentiate between a local sinus
setting of immunodeficiencies (eg, selective immunoglobulin defi- process or broader airway disease and thus the underlying mecha-
ciency), autoimmune conditions (eg, granulomatosis with polyangiitis nism associated with the disease process.
or sarcoidosis), genetic abnormalities (eg, cystic fibrosis), odontogenic
sinusitis, or local neoplasm do not have primary CRS, as their sinona- Endotype Dominance
sal mucosal disease is secondary to another process. At the sinus mucosal level, the adaptive immune response is typically
divided into a T-cell response and a B-cell response; these will be dis-
Anatomical Distribution cussed as they relate to CRS. Several patterns of inflammatory immune
Localized response have been described, but the most recognized are the helper
Local airway inflammation occurs at the level of the sinonasal mu- Tcell,type1(TH1),TH2,andTH17immuneresponses.14 Thesearesome-
cosa without direct involvement of the lower airway or even the con- times referred to as type 1, type 2, and type 3. In the pathogenesis of
tralateral paranasal sinuses. The key feature is that the sinus cavi- CRS, these immune responses may go awry.
ties involved are anatomically discrete and almost always follow the
known ostial or functional drainage pathways. Examples are iso- Type 2
lated sphenoid, isolated frontal, or the ostiomeatal unit group of si- The type 2 inflammatory response seen in CRS is likely associated with
nuses: maxillary, anterior ethmoid, and frontal. These are ex- TH2 cells, cytotoxic T cells, and innate lymphoid cells.4 Type 2 immune
amples of a localized disease process and are usually unilateral responses are associated with upregulated production of interleukin
E2 JAMA Otolaryngology–Head & Neck Surgery Published online July 9, 2020 (Reprinted) jamaotolaryngology.com
© 2020 American Medical Association. All rights reserved.
Downloaded From: https://jamanetwork.com/ by a Glasgow University Library User on 07/10/2020
Contemporary Classification of Chronic Rhinosinusitis Review Clinical Review & Education
4 (IL-4), IL-5, IL-13, local immunoglobulin E (IgE), and profound eosino- sitis, to have broader airway disease and bronchpulmonary
philia. The threshold of eosinophilia (or other markers) that might de- aspergillosis.26 Although classically unilateral, allergic fungal rhino-
fine type 2 responses is likely to change over time and has been inten- sinusitis can be diffuse (eFigure 1 in the Supplement). The mainstay
tionally avoided.15,16 Type 2 responses are also associated with high of treatment for these diseases includes early surgical intervention
asthma comorbidity. Interleukin 5 is a key activator and factor for sur- with creation of a “neo-sinus” cavity in addition to adequate post-
vival of the eosinophil. Interleukin 4 and IL-13 are responsible for mu- operative medical management (ie, corticosteroid irrigations).
cusproductioninepithelialcells.Apresumedmechanismofthisinflam- Localized, non–type 2 pathologic conditions include the follow-
mation is through stimulation of nasal epithelial cells to secrete thymic ing: isolated frontal disease (Figure 2A), isolated sphenoid disease
stromallymphopoietin,IL-25,andIL-33.Interleukin25isintheIL-17fam- (Figure 2B), and ostiomeatal complex involvement (Figure 2C and
ily; however, it is responsible for inducing a type 2 immune response. D). These clinical entities are not associated with eosinophils or IgE.
Thymic stromal lymphopoietin, IL-25, and IL-33 induce the release of The remodeling events that occur with inflammation (fibrosis, base-
IL-4, IL-5, and IL-13 from the epithelial and mucosal mast cells. These ment membrane thickening, fibrin deposition, and metaplasia) may
interleukins then go on to stimulate immunoglobulin class, switching not be fully reversible with medical therapy.27 When these remod-
to IgE and IgG4 and mast cell degranulation into eosinophils.17 Mast cell eling events occur with functional drainage pathways, they create
andeosinophildegranulationtriggerinflammationandsubsequenttis- anatomically discrete or localized sinus disease. These conditions
suedamage,whichmayhavelong-termnegativeconsequences.Treat- usually require surgical intervention and have a good prognosis from
ment interventions are being developed that are based on inhibition surgery as a single-modality intervention.
of these type 2 inflammatory mediators, including duplimab (IL-4 and Diffuse type 2 pathologic conditions include central compart-
IL-13), mepoluzimab (IL-5), benraluzimab (IL-5 receptor), and omali- ment atopic disease, eosinophilic CRS, and allergic fungal rhinosinu-
zumab (IgE).18 sitis. A newly described phenotype is central compartment atopic
disease, which is an IgE-associated disease process that is associ-
Non–Type 2 ated with inhalant allergy.28,29 Patients with central compartment
Non–type 2 inflammation in the setting of CRS is a mix of type 1 and atopic disease will have specific radiographic findings, including cen-
type 3 inflammation. Interleukin 6, IL-8, and tumor necrosis factor tral thickening of the turbinates and septum with superior and lat-
have been shown to stimulate the production of interferon gamma eral sparing of the sinus cavities.30 This centralized inflammation with
(IFN-γ) and IL-8, which further recruit immune responses. Interleu- otherwise normal surrounding mucosa has been described as a halo
kin 8 recruits neutrophils to the area, which then release further cy- sign31 (eFigure 2 in the Supplement). The sinus dysfunction is mainly
tokines, including IL-1β, IL-6, IL-8, and myeloperoxidase. The re- postobstructive with mucus trapping. Barotrauma and pressure
lease of IFN-γ leads to the differentiation of CD4-positive T cells into symptoms are common for individuals with central compartment
the TH1 inflammatory response through further production of IL-2 atopic disease. In contrast, eosinophilic CRS is associated primarily
and IFN-γ.17 The immune-modulating effects of macrolides are an with tissue and serum eosinophila, and patients will have or go on
example of the anti–IL-8 effect that have been shown to benefit pa- to develop concomitant lower airway disease (adult-onset eosino-
tients with this skew in inflammation.19,20 philic asthma). These patients typically have nasal polyps and se-
The proposed mechanism of type 1 inflammation involves an epi- vere olfactory dysfunction, and their symptoms are sensitive to
thelial response to environmental stimuli that leads to osteopontin corticosteroids.32 Their loss of smell is often profound and very sen-
stimulation of dendritic cells, which induce TH1-cell differentiation sitive to corticosteroids. They will have thick, glue-like eosinophilic
and TH17-cell differentiation. This mechanism further orchestrates mucin (Figure 3). These patients’ symptoms are not associated with
noneosinophilic inflammation through production of IFN-γ and tu- inhalant allergy in contrast to central compartment atopic disease,
mor necrosis factor and lymphotoxin α. This production leads to mac- and serum IgE is not directly associated with eosinophilic response.33
rophage activation with enhancement of phagocytic properties, class Diffuse allergic fungal rhinosinusitis is similar to the previously de-
switching to production of IgG subclasses with complement-fixing scribed localized allergic fungal rhinosinusitis except that there is
abilities, antigen presentation to macrophages, and local tissue in- broad paranasal sinus involvement (eFigure 3 in the Supplement).
flammation and neutrophil activation.21 Diffuse non–type 2 pathologic conditions include non–
The proposed mechanism of type 3 inflammation involves TH17 eosinophilic CRS. Patients with non–eosinophilic CRS are often older
responses that are preferentially invoked with the production of large (age, 50-60 years)5 and do not have the same benefit from the use
amounts of IL-17A, IL-17F, and IL-22, with several protective effects of corticosteroids.19 These patients may have polyps or polypoid
that include neutrophil recruitment, activation, and proliferation and edema, but they will lack the eosinophilic mucin seen in patients with
innate antimicrobial production by airway epithelial cells.22 eosinophilic CRS.16,34 These patients will less often have smell loss
as a presenting symptom and may have an uncontrolled lower air-
Clinical Phenotype way when using corticosteroid inhalers.19,20,32 They will often have
Localized, type 2 pathologic conditions associated with inflamma- postobstructive infective phenomena with purulence noted on en-
tion can include examples such as allergic fungal rhinosinusitis, a lo- doscopic evaluation35 (Figure 4).
cal response to common fungal species including Alternaria, Bipo-
laris, and Curvularia. Diagnosis requires eosinophilic mucin, which
has a typical appearance of peanut butter, fungal hyphae with Char-
Secondary CRS
cot-Leyden crystals on histologic examination, and serum-specific
IgE to fungal pathogens.23-25 It is uncommon for patients with lo- Less commonly, CRS is secondary to an established systemic dis-
calized type 2–dominant disease, such as allergic fungal rhinosinu- ease or local pathologic condition (odontogenic or neoplasm)
jamaotolaryngology.com (Reprinted) JAMA Otolaryngology–Head & Neck Surgery Published online July 9, 2020 E3
© 2020 American Medical Association. All rights reserved.
Downloaded From: https://jamanetwork.com/ by a Glasgow University Library User on 07/10/2020
Clinical Review & Education Review Contemporary Classification of Chronic Rhinosinusitis
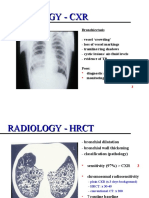
Figure 2. Primary Localized Non–Type 2 Chronic Rhinosinusitis
A Left frontal sinus disease B Left sphenoid disease
R L
R
C Purulence in middle meatus D Left OMC disease
A, Coronal computed tomography
(CT) scan showing isolated left frontal
sinus disease. B, Axial CT scan
showing isolated left sphenoid
disease. C, Nasal endoscopy with
purulence within the middle meatus
for this non–type 2 disease process.
D, Coronal CT scan showing isolated
left ostiomeatal complex (OMC)
disease. These images are examples
of traditional ostial occlusion chronic
B rhinosinusitis disease or isolated
sinusitis.
(Figure 5). Secondary CRS represents a group of clinical entities Mechanism
in which the sinus disease observed is part of another disease Although the list and categorization here is not designed to be com-
process. Secondary CRS is simply an expression of another condi- prehensive, most systemic conditions that are associated with the
tion, and correction of that mechanism will result in resolution of development of mucosal inflammation predominantly fall into con-
the CRS. ditions that impair the mechanical function of the sinuses to clear
mucus (mucociliary), the development of an abnormal immune re-
Anatomical Distribution sponse (autoimmune), and the lack of an appropriate immune re-
Localized sponse (immunodeficiency). It is important to highlight that these
Local sinus pathologic conditions, such as periapical dental ab- are processes that occur outside the respiratory system as well.
scess, neoplasm, facial trauma, and foreign bodies, are all ex-
amples of CRS that may occur locally in anatomically discrete sinus Mucociliary
cavities as a result of these pathologic conditions.36 These condi- Conditions that fit under this classification will affect the clearance of
tions are usually unilateral, but uncommon situations may arise, such mucusfromtheparanasalsinusesandwillleadtomucostasis.Mechani-
as advanced neoplasms,37 in which both sides are involved, but the cal clearance of mucus is an important part of the innate immune de-
CRS and pathologic condition remain anatomically related. Sur- fense, and mucostasis can serve as a source for secondary infection.
gery will likely play a role in first-line therapy in the management of Pseudomonas and Staphylococcus aureus are common bacteria that
these conditions. colonize those with mucostasis. The mechanical clearance of mucus
is an important part of the innate immune defense. Cystic fibrosis is a
Diffuse disorder of impaired mucociliary clearance. Primary ciliary dyskinesia
Secondary CRS that is diffuse is almost always the result of a is an example of loss of mechanical action of the cilia.38
broader systemic disorder. Examples include cystic fibrosis, selec-
tive immunodeficiency, and granulomatosis with polyangiitis; Autoimmune
these are diseases in which inflammation in the sinus cavity is Autoimmunepathologicconditionsarisefromautoantibodiesthatlead
an expression of the underlying disease and are thus considered to vasculitis or systemic deposition of inflammatory cells. These patho-
secondary CRS. logic conditions occur systemically and can have effects in the upper
E4 JAMA Otolaryngology–Head & Neck Surgery Published online July 9, 2020 (Reprinted) jamaotolaryngology.com
© 2020 American Medical Association. All rights reserved.
Downloaded From: https://jamanetwork.com/ by a Glasgow University Library User on 07/10/2020
Contemporary Classification of Chronic Rhinosinusitis Review Clinical Review & Education
Figure 3. Primary Diffuse Type 2 Dominant Chronic Rhinosinusitis (CRS): Eosinophilic CRS (eCRS)
or Adult-Onset Eosinophilic Nasal Polyposis
A Pansinus involvement of eCRS B Middle meatal mucosal edema and eosinophilic secretions
A, Coronal computed tomography
scan showing the typical pansinus
involvement of eCRS. B, Nasal
endoscopy showing extensive middle
meatal mucosal edema and
eosinophilic secretions. The size of
the polyps does not always reflect
the severity of the inflammatory
process. This type of CRS is often
associated with adult-onset asthma.
These patients obtain significant
relief from corticosteroid use.
and lower airways. Examples include granulomatosis with polyangiitis autosomal recessive genetic mutations. Primary ciliary dyskin-
(formerly known as Wegener granulomatosis) and eosinophilic granu- esia is highlighted by defective respiratory cilia; however, other
lomatosis with polyangiitis (formerly known as Churg-Strauss cilia are defective as well. These patients have poor mucociliary
syndrome).39,40 clearance and experience upper and lower airway disease.46,47
They often have polypoid edema or polyps on results of
Immunodeficiency endoscopy and have diffuse sinus involvement48,49 (eFigure 5 in
Immunodeficiencies lead to the inability to clear pathogens and per- the Supplement).
sistence or recurrence of infections. The upper airway serves as a Patients with cystic fibrosis have impaired chloride transport
filter for some of these pathogens and can be inoculated easily given leading to decreased air-surface liquid interface, which leads to mu-
the route of transmission. Classic immunodeficiencies identified with cociliary clearance defects owing to defective ciliary beat fre-
sinus involvement include selective immunoglobulin deficiencies, quency. These patients often have polypoid edema or true middle
common variable immune deficiency (CVID), and type 1 and 2 meatal polyps on results of endoscopy with universal pansinus in-
diabetes.41 volvement on results of imaging. In many cases, depending on the
specific genetic mutation, there is hypoplasia of the maxillary and
Clinical Phenotype frontal sinuses on results of radiography.50
Localized involvement of the paranasal sinuses may occur due to Diffuse disease due to autoimmune disease includes eosino-
certain local pathologic conditions (eg, odontogenic sources, fun- philic granulomatosis with polyangiitis. Patients with eosinophilic an-
gal ball, or neoplasia). Fungal ball affects the maxillary sinus most giitis have eosinophilia, asthma, and evidence of vasculitis. The up-
often; however, it can be seen in other isolated sinuses.42-44 Given per airway of these patients will look similar to the upper airway of
the predilection of fungal ball for the maxillary sinus, patients those with eosinophilic CRS; however, patients with eosinophilic an-
undergoing endodontic treatment are at higher risk. 45 These giitis have other organ involvement such as skin, cardiac, gastroin-
patients may also have purulence draining from the sinus owing to testinal, and musculoskeletal.51
postobstructive infective phenomena. Odontogenic sinusitis is Patients with granulomatosis with polyangiitis will experience
typically seen in patients older than 40 years with a history of pre- nasal ulcers and crusting, saddle-nose deformity, and/or other sys-
vious dental work or need for dental work. These patients may temic symptoms (fatigue, weight loss, and night sweats). Septal ul-
also report cachosmia. When the maxillary sinus is the primary cerations and perforations may be present in patients with active
sinus affected, the anterior ethmoid and frontal sinus can be disease and those with disease in remission. Nasal crusting is com-
involved in a postobstructive manner; however, the disease is ana- mon in patients with active disease.52 Patients with granulomato-
tomically discrete or localized to a functionally related group of sis with polyangiitis will have other organs involved such as skin, kid-
sinuses (eFigure 4 in the Supplement). Benign and malignant neo- ney, musculoskeletal, ophthalmologic, central nervous system,
plasia can affect the sinus from which it arises by causing local and/or cardiac.53-55
obstruction for normal mucociliary flow but also increased inflam- Diffuse disease associated with immunodeficiencies includes
mation in the area. The sinuses surrounding the neoplasia may selective IgA deficiency, CVID, and diabetes. The prevalence of
experience postobstructive mucus trapping. Patients with local- selective IgA deficiency is different across different races/
ized but secondary CRS usually require surgery, but the interven- ethnicities and can be a clinical challenge to diagnose. These
tion needs both drainage and reventilation of the sinuses as well as patients may present with recurrent sinopulmonary infection,
the underlying pathologic condition to be treated for the CRS to allergic symptoms, and autoimmune illnesses. However, most
resolve. patients are asymptomatic. These patients will have a serum IgA
Diffuse disease due to mucociliary defects includes primary level less than 7 mg/dL (to convert to grams per liter, multiply by
ciliary dyskinesia and cystic fibrosis, which are caused by different 0.01) in individuals older than 4 years with otherwise normal
jamaotolaryngology.com (Reprinted) JAMA Otolaryngology–Head & Neck Surgery Published online July 9, 2020 E5
© 2020 American Medical Association. All rights reserved.
Downloaded From: https://jamanetwork.com/ by a Glasgow University Library User on 07/10/2020
Clinical Review & Education Review Contemporary Classification of Chronic Rhinosinusitis
Figure 4. Primary Diffuse Non–Type 2 Chronic Rhinosinusitis (CRS): Non-Eosinophilic CRS (eCRS)
A Polyp formation B Pansinus involvement
A, Nasal endoscopy demonstrating
that polyp formation occurs in
different inflammatory subtypes.
The secretions are distinctively
different from those in eCRS.
B, Coronal computed tomography
scan demonstrating the diffuse or
pansinus involvement. This patient
(age, >50 years) was poorly
responsive to corticosteroids with
disease in both upper and lower
airways but had complete remission
with long-term macrolide therapy.
Figure 5. Classification of Secondary Chronic Rhinosinusitis (CRS)
Anatomical Clinical examples and
distribution Mechanism phenotypes
Local pathologic Odontogenic
features Fungal ball tumor
Localized
(unilateral)
Mechanical PCD
(mucociliary) CF
Secondary CRS
Inflammatory GPA
(autoimmune) EGPA
Diffuse
(bilateral)
Selective IgA
Immunity deficiency
(immunodeficiency) CVID
Diabetes
Secondary CRS implies that another local or systemic pathologic condition or disease process is the factor associated with the sinus changes. The functional
anatomical compartments involved create the first level of separation into local and diffuse. These compartments are usually unilateral and bilateral in distribution.
Diffuse does not imply pansinusitis but simply that the disease is not confined to a known anatomical unit, taking into account whether local anatomical factors
are associated with pathogenesis. The mechanism for diffuse disease implies that another process is associated with the CRS and either a mechanical (mucociliary),
an inflammatory (usually autoimmune), or an immunodeficiency dysfunction is present. The phenotypes or clinical examples are CRS entities that have been
described and how they align with this system. CF indicates cystic fibrosis; CVID, common variable immunodeficiency; EGPA, eosinophilic granulomatosis with
polyangiitis; GPA, granulomatosis with polyangiitis; IgA, immunoglobulin A; and PCD, primary ciliary dyskinesia.
immunoglobulins and normal responses to vaccinations. Patients
may present with allergic symptoms including conjunctivitis, rhini- Conclusions
tis, urticaria, eczema, food allergies, and asthma. The IgE concen-
tration is often elevated in these patients.56 Common variable With scientific advances in the understanding of CRS, a working clas-
immunodeficiency is often not diagnosed until adulthood despite sification system for CRS should move away from the dichotomy of
being one of the most common primary immunodeficiencies. CRS with nasal polyps and CRS without nasal polyps to change clini-
Patients with CVID have a normal number of B immune cells; how- cal practice. The proposed classification has been left intentionally
ever, the cells do not mature appropriately. Patients with CVID simple (local or diffuse and type 2 or non–type 2) to give it the flex-
present with a broad range of symptoms including recurrent bac- ibility to incorporate future research and allow phenotypes to be de-
terial infections, autoimmunity, interstitial lung disease, and aller- fined. The proposal allows for current therapies, from balloons to
gic symptoms. Recurrent sinusitis is found in half the patients with macrolides to surgery and biologics, to be more appropriately di-
CVID.57 Some of these patients may have already undergone mul- rected. Although research continues to further define the sub-
tiple sinus operations, and their sinusitis may not have been well types of CRS into phenotypes and endotypes, the proposed classi-
controlled with conventional therapy; therefore, investigations fication system of primary CRS by anatomical distribution and
into immunodeficiencies should be considered. endotype dominance allows for a pathway forward.
E6 JAMA Otolaryngology–Head & Neck Surgery Published online July 9, 2020 (Reprinted) jamaotolaryngology.com
© 2020 American Medical Association. All rights reserved.
Downloaded From: https://jamanetwork.com/ by a Glasgow University Library User on 07/10/2020
Contemporary Classification of Chronic Rhinosinusitis Review Clinical Review & Education
ARTICLE INFORMATION heterogeneous disease. Lancet. 2008;372(9643): 24. Schubert MS. Allergic fungal sinusitis.
Accepted for Publication: May 5, 2020. 1107-1119. doi:10.1016/S0140-6736(08)61452-X Otolaryngol Clin North Am. 2004;37(2):301-326.
10. Akdis CA, Bachert C, Cingi C, et al. Endotypes doi:10.1016/S0030-6665(03)00152-X
Published Online: July 9, 2020.
doi:10.1001/jamaoto.2020.1453 and phenotypes of chronic rhinosinusitis: 25. Folker RJ, Marple BF, Mabry RL, Mabry CS.
a PRACTALL document of the European Academy Treatment of allergic fungal sinusitis: a comparison
Author Contributions: Drs Grayson and Harvey of Allergy and Clinical Immunology and the trial of postoperative immunotherapy with specific
had full access to all of the data in the study and American Academy of Allergy, Asthma & fungal antigens. Laryngoscope. 1998;108(11, pt 1):
take responsibility for the integrity of the data and Immunology. J Allergy Clin Immunol. 2013;131(6): 1623-1627. doi:10.1097/00005537-199811000-
the accuracy of the data analysis. 1479-1490. doi:10.1016/j.jaci.2013.02.036 00007
Concept and design: All authors.
Acquisition, analysis, or interpretation of data: 11. Bachert C, Akdis CA. Phenotypes and emerging 26. Shah A, Panchal N, Agarwal AK. Concomitant
Grayson, Senior, Harvey. endotypes of chronic rhinosinusitis. J Allergy Clin allergic bronchopulmonary aspergillosis and allergic
Drafting of the manuscript: Grayson. Immunol Pract. 2016;4(4):621-628. doi:10.1016/j. Aspergillus sinusitis: a review of an uncommon
Critical revision of the manuscript for important jaip.2016.05.004 association. Clin Exp Allergy. 2001;31(12):1896-1905.
intellectual content: All authors. 12. Khan A, Vandeplas G, Huynh TMT, et al. doi:10.1046/j.1365-2222.2001.01159.x
Administrative, technical, or material support: The Global Allergy and Asthma European Network 27. Barham HP, Osborn JL, Snidvongs K, Mrad N,
Grayson. (GALEN) rhinosinusitis cohort: a large European Sacks R, Harvey RJ. Remodeling changes of the
Supervision: Hopkins, Mori, Senior, Harvey. cross-sectional study of chronic rhinosinusitis upper airway with chronic rhinosinusitis. Int Forum
Conflict of Interest Disclosures: Dr Hopkins patients with and without nasal polyps. Rhinology. Allergy Rhinol. 2015;5(7):565-572. doi:10.1002/alr.
reported completing board work for Sanofi, 2019;57(1):32-42. doi:10.4193/Rhin17.255 21546
AstraZeneca, Smith & Nephew, and Olympus. 13. Fokkens WJ, Lund VJ, Hopkins C, et al. 28. DelGaudio JM, Loftus PA, Hamizan AW,
Dr Harvey reported serving as a consultant with European position paper on rhinosinusitis and nasal Harvey RJ, Wise SK. Central compartment atopic
Medtronic, Olympus, and NeilMed Pharmaceuticals polyps 2020. Rhinology. 2020;58(suppl S29):1-464. disease. Am J Rhinol Allergy. 2017;31(4):228-234.
and serving on the speakers bureau for doi:10.4193/Rhin20.600 doi:10.2500/ajra.2017.31.4443
GlaxoSmithKline, Seqiris, and AstraZeneca. 14. Stevens WW, Peters AT, Tan BK, et al. 29. Marcus S, Schertzer J, Roland LT, Wise SK,
No other disclosures were reported. Associations between inflammatory endotypes and Levy JM, DelGaudio JM. Central compartment
clinical presentations in chronic rhinosinusitis. atopic disease: prevalence of allergy and asthma
REFERENCES J Allergy Clin Immunol Pract. 2019;7(8):2812-2820.e3. compared with other subtypes of chronic
1. Fokkens W, Lund V, Mullol J; European Position doi:10.1016/j.jaip.2019.05.009 rhinosinusitis with nasal polyps. Int Forum Allergy
Paper on Rhinosinusitis and Nasal Polyps Group. 15. McHugh T, Snidvongs K, Xie M, Banglawala S, Rhinol. 2020;10(2):183-189. doi:10.1002/alr.22454
European position paper on rhinosinusitis and nasal Sommer D. High tissue eosinophilia as a marker to 30. Hamizan AW, Loftus PA, Alvarado R, et al.
polyps 2007. Rhinol Suppl. 2007;20:1-136. predict recurrence for eosinophilic chronic Allergic phenotype of chronic rhinosinusitis based
2. Fokkens WJ, Lund VJ, Mullol J, et al. European rhinosinusitis: a systematic review and on radiologic pattern of disease. Laryngoscope.
position paper on rhinosinusitis and nasal polyps meta-analysis. Int Forum Allergy Rhinol. 2018;8(12): 2018;128(9):2015-2021. doi:10.1002/lary.27180
2012. Rhinol Suppl. 2012;23:3, 1-298. 1421-1429. doi:10.1002/alr.22194 31. Scadding G, Lund V. Investigative Rhinology.
3. Takabayashi T, Kato A, Peters AT, et al. Excessive 16. Snidvongs K, Lam M, Sacks R, et al. Structured Taylor & Francis; 2004. doi:10.3109/9780203504772
fibrin deposition in nasal polyps caused by histopathology profiling of chronic rhinosinusitis in 32. Mori E, Matsuwaki Y, Mitsuyama C, Okushi T,
fibrinolytic impairment through reduction of tissue routine practice. Int Forum Allergy Rhinol. 2012;2 Nakajima T, Moriyama H. Risk factors for olfactory
plasminogen activator expression. Am J Respir Crit (5):376-385. doi:10.1002/alr.21032 dysfunction in chronic rhinosinusitis. Auris Nasus
Care Med. 2013;187(1):49-57. doi:10.1164/rccm. 17. Ahern S, Cervin A. Inflammation and Larynx. 2013;40(5):465-469. doi:10.1016/j.anl.2012.
201207-1292OC endotyping in chronic rhinosinusitis—a paradigm 12.005
4. Cao PP, Wang ZC, Schleimer RP, Liu Z. shift. Medicina (Kaunas). 2019;55(4):E95. doi:10. 33. Ho J, Hamizan AW, Alvarado R, Rimmer J,
Pathophysiologic mechanisms of chronic 3390/medicina55040095 Sewell WA, Harvey RJ. Systemic predictors of
rhinosinusitis and their roles in emerging disease 18. Henry EK, Inclan-Rico JM, Siracusa MC. Type 2 eosinophilic chronic rhinosinusitis. Am J Rhinol
endotypes. Ann Allergy Asthma Immunol. 2019;122 cytokine responses: regulating immunity to Allergy. 2018;32(4):252-257. doi:10.1177/
(1):33-40. doi:10.1016/j.anai.2018.10.014 helminth parasites and allergic inflammation. Curr 1945892418779451
5. Turner JH, Chandra RK, Li P, Bonnet K, Pharmacol Rep. 2017;3(6):346-359. doi:10.1007/ 34. Grayson JW, Cavada M, Harvey RJ. Clinically
Schlundt DG. Identification of clinically relevant s40495-017-0114-1 relevant phenotypes in chronic rhinosinusitis.
chronic rhinosinusitis endotypes using cluster 19. Oakley GM, Christensen JM, Sacks R, Earls P, J Otolaryngol Head Neck Surg. 2019;48(1):23.
analysis of mucus cytokines. J Allergy Clin Immunol. Harvey RJ. Characteristics of macrolide responders doi:10.1186/s40463-019-0350-y
2018;141(5):1895-1897.e7. doi:10.1016/j.jaci.2018. in persistent post-surgical rhinosinusitis. Rhinology.
02.002 35. Morse JC, Li P, Ely KA, et al. Chronic
2018;56(2):111-117. doi:10.4193/Rhin17.049 rhinosinusitis in elderly patients is associated with
6. Wenzel SE. Asthma phenotypes: the evolution 20. Oakley GM, Harvey RJ, Lund VJ. The role of an exaggerated neutrophilic proinflammatory
from clinical to molecular approaches. Nat Med. macrolides in chronic rhinosinusitis (CRSsNP and response to pathogenic bacteria. J Allergy Clin
2012;18(5):716-725. doi:10.1038/nm.2678 CRSwNP). Curr Allergy Asthma Rep. 2017;17(5):30. Immunol. 2019;143(3):990-1002. doi:10.1016/j.jaci.
7. Ozdemir C, Kucuksezer UC, Akdis M, Akdis CA. doi:10.1007/s11882-017-0696-z 2018.10.056
The concepts of asthma endotypes and phenotypes 21. Commins SP, Borish L, Steinke JW. Immunologic 36. Patel NA, Ferguson BJ. Odontogenic sinusitis:
to guide current and novel treatment strategies. messenger molecules: cytokines, interferons, and an ancient but under-appreciated cause of maxillary
Expert Rev Respir Med. 2018;12(9):733-743. doi:10. chemokines. J Allergy Clin Immunol. 2010;125(2) sinusitis. Curr Opin Otolaryngol Head Neck Surg.
1080/17476348.2018.1505507 (suppl 2):S53-S72. doi:10.1016/j.jaci.2009.07.008 2012;20(1):24-28. doi:10.1097/MOO.
8. Tomassen P, Vandeplas G, Van Zele T, et al. 22. Mucida D, Salek-Ardakani S. Regulation of TH17 0b013e32834e62ed
Inflammatory endotypes of chronic rhinosinusitis cells in the mucosal surfaces. J Allergy Clin Immunol. 37. Knisely A, Holmes T, Barham H, Sacks R,
based on cluster analysis of biomarkers. J Allergy 2009;123(5):997-1003. doi:10.1016/j.jaci.2009.03. Harvey R. Isolated sphenoid sinus opacification:
Clin Immunol. 2016;137(5):1449-1456.e4. doi:10. 016 a systematic review. Am J Otolaryngol. 2017;38(2):
1016/j.jaci.2015.12.1324 237-243. doi:10.1016/j.amjoto.2017.01.014
23. Kuhn FA, Swain R Jr. Allergic fungal sinusitis:
9. Anderson GP. Endotyping asthma: new insights diagnosis and treatment. Curr Opin Otolaryngol 38. McCormick JP, Weeks CG, Rivers NJ, et al.
into key pathogenic mechanisms in a complex, Head Neck Surg. 2003;11(1):1-5. doi:10.1097/ Prevalence of chronic rhinosinusitis in
00020840-200302000-00001 bronchiectasis patients suspected of ciliary
jamaotolaryngology.com (Reprinted) JAMA Otolaryngology–Head & Neck Surgery Published online July 9, 2020 E7
© 2020 American Medical Association. All rights reserved.
Downloaded From: https://jamanetwork.com/ by a Glasgow University Library User on 07/10/2020
Clinical Review & Education Review Contemporary Classification of Chronic Rhinosinusitis
dyskinesia. Int Forum Allergy Rhinol. 2019;9(12): 45. Park GY, Kim HY, Min JY, Dhong HJ, Chung SK. patients. Medicine (Baltimore). 1999;78(1):26-37.
1430-1435. doi:10.1002/alr.22414 Endodontic treatment: a significant risk factor for doi:10.1097/00005792-199901000-00003
39. Nakamaru Y, Takagi D, Suzuki M, et al. Otologic the development of maxillary fungal ball. Clin Exp 52. Beltrán Rodríguez-Cabo O, Reyes E,
and rhinologic manifestations of eosinophilic Otorhinolaryngol. 2010;3(3):136-140. doi:10.3342/ Rojas-Serrano J, Flores-Suárez LF. Increased
granulomatosis with polyangiitis. Audiol Neurootol. ceo.2010.3.3.136 histopathological yield for granulomatosis with
2016;21(1):45-53. doi:10.1159/000442040 46. Goutaki M, Meier AB, Halbeisen FS, et al. polyangiitis based on nasal endoscopy of suspected
40. Low CM, Keogh KA, Saba ES, et al. Chronic Clinical manifestations in primary ciliary dyskinesia: active lesions. Eur Arch Otorhinolaryngol. 2018;275
rhinosinusitis in eosinophilic granulomatosis with systematic review and meta-analysis. Eur Respir J. (2):425-429. doi:10.1007/s00405-017-4841-z
polyangiitis: clinical presentation and antineutrophil 2016;48(4):1081-1095. doi:10.1183/13993003. 53. Hoffman GS, Kerr GS, Leavitt RY, et al. Wegener
cytoplasmic antibodies. Int Forum Allergy Rhinol. 00736-2016 granulomatosis: an analysis of 158 patients. Ann
2020;10(2):217-222. doi:10.1002/alr.22503 47. Alanin MC, Johansen HK, Aanaes K, et al. Intern Med. 1992;116(6):488-498. doi:10.7326/
41. Mazza JM, Lin SY. Primary immunodeficiency Simultaneous sinus and lung infections in patients 0003-4819-116-6-488
and recalcitrant chronic sinusitis: a systematic with primary ciliary dyskinesia. Acta Otolaryngol. 54. Holle JU, Gross WL. Neurological involvement
review. Int Forum Allergy Rhinol. 2016;6(10): 2015;135(1):58-63. doi:10.3109/00016489.2014. in Wegener’s granulomatosis. Curr Opin Rheumatol.
1029-1033. doi:10.1002/alr.21789 962185 2011;23(1):7-11. doi:10.1097/BOR.0b013e32834115f9
42. Shin JM, Baek BJ, Byun JY, Jun YJ, Lee JY. 48. Laura CA, Lourdes P, Eduardo F, Fabricio I, 55. Florian A, Slavich M, Blockmans D,
Analysis of sinonasal anatomical variations Urquiola F. Nasal polyposis in patients with primary Dymarkowski S, Bogaert J. Cardiac involvement in
associated with maxillary sinus fungal balls. Auris ciliary dyskinesia. J Otolaryngol ENT Res. 2017;7 granulomatosis with polyangiitis (Wegener
Nasus Larynx. 2016;43(5):524-528. doi:10.1016/j. (4):00210. doi:10.15406/joentr.2017.07.00210 granulomatosis). Circulation. 2011;124(13):e342-e344.
anl.2015.12.013 49. Storm van’s Gravesande K, Omran H. Primary doi:10.1161/CIRCULATIONAHA.111.030809
43. Jun YJ, Shin JM, Lee JY, Baek BJ. Bony changes ciliary dyskinesia: clinical presentation, diagnosis 56. Yazdani R, Azizi G, Abolhassani H,
in a unilateral maxillary sinus fungal ball. J Craniofac and genetics. Ann Med. 2005;37(6):439-449. Aghamohammadi A. Selective IgA deficiency:
Surg. 2018;29(1):e44-e47. doi:10.1097/SCS. doi:10.1080/07853890510011985 epidemiology, pathogenesis, clinical phenotype,
0000000000004010 50. Schraven SP, Wehrmann M, Wagner W, diagnosis, prognosis and management. Scand J
44. Yoon YH, Xu J, Park SK, Heo JH, Kim YM, Blumenstock G, Koitschev A. Prevalence and Immunol. 2017;85(1):3-12. doi:10.1111/sji.12499
Rha KS. A retrospective analysis of 538 sinonasal histopathology of chronic polypoid sinusitis in 57. Yazdani R, Habibi S, Sharifi L, et al. Common
fungus ball cases treated at a single tertiary medical pediatric patients with cystic fibrosis. J Cyst Fibros. variable immunodeficiency: epidemiology,
center in Korea (1996-2015). Int Forum Allergy Rhinol. 2011;10(3):181-186. doi:10.1016/j.jcf.2011.01.003 pathogenesis, clinical manifestations, diagnosis,
2017;7(11):1070-1075. doi:10.1002/alr.22007 51. Guillevin L, Cohen P, Gayraud M, Lhote F, classification, and management. J Investig Allergol
Jarrousse B, Casassus P. Churg-Strauss syndrome: Clin Immunol. 2020;30(1):14-34. doi:10.18176/
clinical study and long-term follow-up of 96 jiaci.0388
E8 JAMA Otolaryngology–Head & Neck Surgery Published online July 9, 2020 (Reprinted) jamaotolaryngology.com
© 2020 American Medical Association. All rights reserved.
Downloaded From: https://jamanetwork.com/ by a Glasgow University Library User on 07/10/2020
You might also like
- Pathoma 2017 PDFDocument405 pagesPathoma 2017 PDFswastikNo ratings yet
- Uworld - PEDIATRICSDocument50 pagesUworld - PEDIATRICSNikxy100% (1)
- ImmunodeficiencyDocument30 pagesImmunodeficiencyDan PantanoNo ratings yet
- Advanced Medicine Recall-A Must For MRCPDocument712 pagesAdvanced Medicine Recall-A Must For MRCPDr Sumant Sharma80% (5)
- DrRichard K Bernstein 37 Tips Tricks and Secrets To Defy DiabetesDocument38 pagesDrRichard K Bernstein 37 Tips Tricks and Secrets To Defy Diabetesbewesa100% (7)
- Primary Immunodeficiency DiseasesDocument24 pagesPrimary Immunodeficiency DiseasesClaire Henderson100% (1)
- MKSAP 16 - Infectious DiseaseDocument340 pagesMKSAP 16 - Infectious DiseaseBacanator75% (4)
- Multiple Sclerosis Lancet 2018Document15 pagesMultiple Sclerosis Lancet 2018Sarah Miryam CoffanoNo ratings yet
- Advanced Medicine Recall A Must For MRCP PDFDocument712 pagesAdvanced Medicine Recall A Must For MRCP PDFKai Xin100% (2)
- Nursing Care of Child With LIFE THREATENING CONDITIONSDocument70 pagesNursing Care of Child With LIFE THREATENING CONDITIONSinno so qtNo ratings yet
- Immunodeficiency DisordersDocument112 pagesImmunodeficiency Disorderswhoosh2008100% (1)
- Adult Chronic Rhinosinusitis: Nature Reviews Disease Primers December 2020Document20 pagesAdult Chronic Rhinosinusitis: Nature Reviews Disease Primers December 2020dayuNo ratings yet
- Greyson 2019Document10 pagesGreyson 2019Ziha Zia Leonita FauziNo ratings yet
- CRS 1Document26 pagesCRS 1Anita AmaliaNo ratings yet
- Fenotipe Rinosinusitis KronisDocument4 pagesFenotipe Rinosinusitis KronisAndika Anjani AgustinNo ratings yet
- Jurnal 3 Abses Leher DalamDocument6 pagesJurnal 3 Abses Leher DalamRohayu AsnawiNo ratings yet
- ARDS SubphenotypesDocument11 pagesARDS Subphenotypesvanes08fretNo ratings yet
- Acute Respiratory Distress Syndrome PhenotypesDocument12 pagesAcute Respiratory Distress Syndrome PhenotypesLeo AcNo ratings yet
- Koch 2009Document9 pagesKoch 2009Intan Kartika NursyahbaniNo ratings yet
- Pierre Robin Sequence: An Evidence-Based Treatment ProposalDocument7 pagesPierre Robin Sequence: An Evidence-Based Treatment ProposalAdrian UmbriaNo ratings yet
- ARDS Subphenotypes: Understanding A Heterogeneous Syndrome: Review Open AccessDocument8 pagesARDS Subphenotypes: Understanding A Heterogeneous Syndrome: Review Open AccessAlex CroNo ratings yet
- Laryngeal DysplasiaDocument7 pagesLaryngeal DysplasiakarimahihdaNo ratings yet
- Postgrad Med J 2005 Pope 309 14Document14 pagesPostgrad Med J 2005 Pope 309 14Mas Teguh WijayantoNo ratings yet
- Modern Management of Osteoradionecrosis: ReviewDocument6 pagesModern Management of Osteoradionecrosis: Reviewita mustainahNo ratings yet
- Manejo de La Tos CronicaDocument6 pagesManejo de La Tos CronicaThomasMáximoMancinelliRinaldoNo ratings yet
- Martin Harris Et Al 2021 The Modified Barium Swallow Study For Oropharyngeal Dysphagia Recommendations From AnDocument10 pagesMartin Harris Et Al 2021 The Modified Barium Swallow Study For Oropharyngeal Dysphagia Recommendations From AnCARLA ARACELI GÓMEZNo ratings yet
- Pediatric Chronic Sinusitis Diagnosis and ManagementDocument10 pagesPediatric Chronic Sinusitis Diagnosis and ManagementcaromoradNo ratings yet
- Int Forum Allergy Rhinol - 2018 - Mattos - Establishing The Minimal Clinically Important Difference For The QuestionnaireDocument6 pagesInt Forum Allergy Rhinol - 2018 - Mattos - Establishing The Minimal Clinically Important Difference For The QuestionnaireSid TNo ratings yet
- Allergy - 2019 - Xu - Novel Findings in Immunopathophysiology of Chronic Rhinosinusitis and Their Role in A Model ofDocument12 pagesAllergy - 2019 - Xu - Novel Findings in Immunopathophysiology of Chronic Rhinosinusitis and Their Role in A Model ofronaldyohanesfNo ratings yet
- Jurnal Mata UNJADocument9 pagesJurnal Mata UNJAHerpian NugrahadilNo ratings yet
- JCM 12 03605 v2Document14 pagesJCM 12 03605 v2IgnacioCortésFuentesNo ratings yet
- Management of GranulomaDocument7 pagesManagement of Granulomamajid mirzaeeNo ratings yet
- Sarcoidose JamaDocument13 pagesSarcoidose JamafilipemateusoliveiraNo ratings yet
- Clinical Practice Guideline (Update) : Adult SinusitisDocument39 pagesClinical Practice Guideline (Update) : Adult SinusitisEmma FitrianaNo ratings yet
- 11 Acute Respiratory Distress Syndrome NEJM Thompson2017Document11 pages11 Acute Respiratory Distress Syndrome NEJM Thompson2017Camila Andrea PeñaNo ratings yet
- Paraneoplastic Syndromes Associated With LaryngealDocument15 pagesParaneoplastic Syndromes Associated With Laryngealandre halimNo ratings yet
- Tto Crup NatureDocument7 pagesTto Crup NatureCarolina Mora RuedaNo ratings yet
- Influence of Remoteness of Residence On Timeliness of Diagnosis and Treatment of Oral Cavity and Oropharynx Cancer: A Retrospective Cohort StudyDocument10 pagesInfluence of Remoteness of Residence On Timeliness of Diagnosis and Treatment of Oral Cavity and Oropharynx Cancer: A Retrospective Cohort StudyVitória LeiteNo ratings yet
- Bahan Ards 2Document52 pagesBahan Ards 2Raqi QatulawanisNo ratings yet
- (Ajomr)Document10 pages(Ajomr)MuhasbirNo ratings yet
- Systemic Diseases Affecting Osseointegration TherapyDocument7 pagesSystemic Diseases Affecting Osseointegration TherapyAlfred OrozcoNo ratings yet
- Kumar Et Al. - 2016 - International Myeloma Working Group Consensus CritDocument19 pagesKumar Et Al. - 2016 - International Myeloma Working Group Consensus CritCarina MoitaNo ratings yet
- Nejm SdraDocument11 pagesNejm SdraCHDanielNo ratings yet
- Guideline Chronic Rhinosinusitis in Children 2019Document7 pagesGuideline Chronic Rhinosinusitis in Children 2019sjhfksglakdjalfsfNo ratings yet
- Estenosis Sub 5Document7 pagesEstenosis Sub 5Nicolás HenaoNo ratings yet
- Expert Consensus Statement Management of Dysphagia in Head and Neck CancerDocument22 pagesExpert Consensus Statement Management of Dysphagia in Head and Neck CancerThomasMáximoMancinelliRinaldoNo ratings yet
- EPOC, Lancet 2015Document10 pagesEPOC, Lancet 2015Sebastian BurgosNo ratings yet
- Soni 2010Document4 pagesSoni 2010AlejandroVeraVargasNo ratings yet
- 0 2023 Piis0091674923000350Document21 pages0 2023 Piis0091674923000350emanuella_ema6860No ratings yet
- A Clinicopathological Study of Supraglottic Malignancy at A Tertiary Care Centre in Central India - May - 2023 - 1448516048 - 6309584Document3 pagesA Clinicopathological Study of Supraglottic Malignancy at A Tertiary Care Centre in Central India - May - 2023 - 1448516048 - 6309584surgeonforlifeNo ratings yet
- 10 1002@lary 29336Document6 pages10 1002@lary 29336Novia KurniantiNo ratings yet
- 699-Article Text-1085-1-10-20181101Document6 pages699-Article Text-1085-1-10-20181101Shahid HussainNo ratings yet
- Chaaban 2013Document6 pagesChaaban 2013Gede Brian Nugraha DenaNo ratings yet
- Post Op Care Following FESS-Rudmik2011Document14 pagesPost Op Care Following FESS-Rudmik2011Muhammad Fakhri AltyanNo ratings yet
- Evaluation and Management of Laryngopharyngeal Reflux Disease: State of The Art ReviewDocument21 pagesEvaluation and Management of Laryngopharyngeal Reflux Disease: State of The Art ReviewDEVNo ratings yet
- CRPS 1Document18 pagesCRPS 1mozi neyNo ratings yet
- PARS Reader's Digest - Jan 2013Document9 pagesPARS Reader's Digest - Jan 2013info8673No ratings yet
- Chronic Rhinosinusitis SteroidDocument9 pagesChronic Rhinosinusitis SteroidHan's ChristianNo ratings yet
- Systemic Inflammatory Response Syndrome The CurrenDocument6 pagesSystemic Inflammatory Response Syndrome The CurrenberlianNo ratings yet
- Lumbar Spinal Stenosis: Syndrome, Diagnostics and Treatment: Nature Reviews Neurology August 2009Document13 pagesLumbar Spinal Stenosis: Syndrome, Diagnostics and Treatment: Nature Reviews Neurology August 2009Surya PangeranNo ratings yet
- Efficacy of Sinusitis UltrasoundDocument7 pagesEfficacy of Sinusitis UltrasoundYazid MultazamNo ratings yet
- Am J Otolaryngol: Derek K. Kong, Amanda M. Kong, Isaac Wasserman, Dillan F. Villavisanis, Alyssa M. HackettDocument6 pagesAm J Otolaryngol: Derek K. Kong, Amanda M. Kong, Isaac Wasserman, Dillan F. Villavisanis, Alyssa M. HackettYusi nursiamNo ratings yet
- Clinical Features of Intracranial Vestibular SchwannomasDocument6 pagesClinical Features of Intracranial Vestibular SchwannomasIndra PrimaNo ratings yet
- Int Forum Allergy Rhinol - 2017 - Levy - Defining The Minimal Clinically Important Difference For Olfactory Outcomes in TheDocument6 pagesInt Forum Allergy Rhinol - 2017 - Levy - Defining The Minimal Clinically Important Difference For Olfactory Outcomes in TheSid TNo ratings yet
- Argiris 2008Document15 pagesArgiris 2008Lisbeth TurraNo ratings yet
- Clinical Study of Headache in Relation To Sinusitis and Its ManagementDocument8 pagesClinical Study of Headache in Relation To Sinusitis and Its ManagementchintyaNo ratings yet
- Cavernous Sinus Syndrome: A Prospective Study of 73 Cases at A Tertiary Care Centre in Northern IndiaDocument7 pagesCavernous Sinus Syndrome: A Prospective Study of 73 Cases at A Tertiary Care Centre in Northern IndiaPriscila VerduzcoNo ratings yet
- Modern Management of Nasal Hemangiomas: JAMA Facial Plastic Surgery - Special CommunicationDocument6 pagesModern Management of Nasal Hemangiomas: JAMA Facial Plastic Surgery - Special Communicationcity22No ratings yet
- Decade of ClinicalDocument8 pagesDecade of ClinicalZha Zha Nurul ZahraNo ratings yet
- Husni Referensi Tesis SpesialisDocument7 pagesHusni Referensi Tesis SpesialisflorensiaNo ratings yet
- Referensi Tesis Spesialis DrumondDocument8 pagesReferensi Tesis Spesialis DrumondflorensiaNo ratings yet
- Referensi Tesis Spesialis StaudacherDocument20 pagesReferensi Tesis Spesialis StaudacherflorensiaNo ratings yet
- Tesis Spesialis Rino Referensi KatoDocument22 pagesTesis Spesialis Rino Referensi KatoflorensiaNo ratings yet
- Referensi Tesis Spesialis ChoDocument6 pagesReferensi Tesis Spesialis ChoflorensiaNo ratings yet
- 73 285 1 SMDocument15 pages73 285 1 SMflorensiaNo ratings yet
- Referensi Tesis Rino SononeDocument4 pagesReferensi Tesis Rino SononeflorensiaNo ratings yet
- Referensi Tesis Spesialis KhalidDocument7 pagesReferensi Tesis Spesialis KhalidflorensiaNo ratings yet
- El-Anwar2020 Article RadiologicalMiddleTurbinateVarDocument5 pagesEl-Anwar2020 Article RadiologicalMiddleTurbinateVarflorensiaNo ratings yet
- Auditory Pathways: Anatomy and Physiology: Handbook of Clinical Neurology March 2015Document24 pagesAuditory Pathways: Anatomy and Physiology: Handbook of Clinical Neurology March 2015florensiaNo ratings yet
- Revista Brasileira DE Reumatologia: Cyclophosphamide Administration Routine in Autoimmune Rheumatic Diseases: A ReviewDocument9 pagesRevista Brasileira DE Reumatologia: Cyclophosphamide Administration Routine in Autoimmune Rheumatic Diseases: A ReviewflorensiaNo ratings yet
- 347 FullDocument8 pages347 FullflorensiaNo ratings yet
- Noise Induces Oxidative Stress in RatDocument5 pagesNoise Induces Oxidative Stress in RatflorensiaNo ratings yet
- Fiber-Optic Endoscopic Evaluation of Swallowing To Assess Swallowing Outcomes As A Function of Head Position in A Normal PopulationDocument6 pagesFiber-Optic Endoscopic Evaluation of Swallowing To Assess Swallowing Outcomes As A Function of Head Position in A Normal PopulationflorensiaNo ratings yet
- Hearing Research: Arwa Kurabi, Elizabeth M. Keithley, Gary D. Housley, Allen F. Ryan, Ann C.-Y. WongDocument9 pagesHearing Research: Arwa Kurabi, Elizabeth M. Keithley, Gary D. Housley, Allen F. Ryan, Ann C.-Y. WongflorensiaNo ratings yet
- Food Sensitivity and Laryngopharyngeal Reflux: Preliminary ObservationsDocument3 pagesFood Sensitivity and Laryngopharyngeal Reflux: Preliminary ObservationsflorensiaNo ratings yet
- 10 1016@j Otoeng 2012 05 002Document8 pages10 1016@j Otoeng 2012 05 002florensiaNo ratings yet
- Jurnal OSADocument10 pagesJurnal OSAflorensiaNo ratings yet
- Soal SoalDocument17 pagesSoal SoalflorensiaNo ratings yet
- 5 AnsDocument6 pages5 AnsflorensiaNo ratings yet
- Ministry of Education and Science of Ukraine V.N. Karazin Kharkiv National UniversityDocument44 pagesMinistry of Education and Science of Ukraine V.N. Karazin Kharkiv National UniversityDrTushar GoswamiNo ratings yet
- Consenso Britanico IDCVDocument17 pagesConsenso Britanico IDCVInmunologia DurandNo ratings yet
- EcgDocument433 pagesEcgihtisham1No ratings yet
- ABSTRACTS FROM NAPCON 2013 - Pancreatic Tuberculosis Presenting As A MassDocument26 pagesABSTRACTS FROM NAPCON 2013 - Pancreatic Tuberculosis Presenting As A MassMikmik bay BayNo ratings yet
- Bronchiectasis 2Document19 pagesBronchiectasis 2Liza FathiarianiNo ratings yet
- PID in ChildrenDocument8 pagesPID in ChildrenRoberto SoehartonoNo ratings yet
- Recognition of Immunodeficiency in The Newborn Period - UpToDateDocument21 pagesRecognition of Immunodeficiency in The Newborn Period - UpToDateAdela AlmadaNo ratings yet
- Autoimmunity in Common Variable Immunodeficiency A Systematic Review and Meta-AnalysisDocument27 pagesAutoimmunity in Common Variable Immunodeficiency A Systematic Review and Meta-AnalysisPanagiotisStoikosNo ratings yet
- Cytomegalovirus in Primary ImmunodeficiencyDocument9 pagesCytomegalovirus in Primary ImmunodeficiencyAle Pushoa UlloaNo ratings yet
- Flow Cytometry: Surface Markers and Beyond: Methods of Allergy and ImmunologyDocument10 pagesFlow Cytometry: Surface Markers and Beyond: Methods of Allergy and ImmunologyMunir AliNo ratings yet
- Primary Immune DeficiencyDocument3 pagesPrimary Immune Deficiencyrazedinblack2No ratings yet
- Arduino Sketches and TutorialsDocument38 pagesArduino Sketches and TutorialsShaya NirenbergNo ratings yet
- Curcumin Role in ParaproteinemiaDocument18 pagesCurcumin Role in ParaproteinemiaINDIRAKALYANINo ratings yet
- Approach To The Child With Recurrent Infections - UpToDate PDFDocument24 pagesApproach To The Child With Recurrent Infections - UpToDate PDFSasirekha SakkarapaniNo ratings yet
- 2016 CIS Annual Meeting Immune Deficiency & DysregulationDocument100 pages2016 CIS Annual Meeting Immune Deficiency & DysregulationMetallum MarionetteNo ratings yet
- Diseases of The Immune System: OBC 231 General PathologyDocument91 pagesDiseases of The Immune System: OBC 231 General PathologyRana MohammadNo ratings yet
- Hypogammaglobulinemia and ArthritisDocument36 pagesHypogammaglobulinemia and ArthritisNilesh NolkhaNo ratings yet
- Ijms 21 05223 PDFDocument42 pagesIjms 21 05223 PDFrohailNo ratings yet
- Immunodeficiency DisorderDocument14 pagesImmunodeficiency DisorderAaryan PatelNo ratings yet
- Immunodeficiency Disorders: DR - Kumaran GanesanDocument58 pagesImmunodeficiency Disorders: DR - Kumaran Ganesanvivek chavhanNo ratings yet