Professional Documents
Culture Documents
Capacity Evaluation Conclusion
Uploaded by
عارف حسین0 ratings0% found this document useful (0 votes)
2 views1 pageThis document provides data on pending applications and registered products for veterinary oral dry powder, oral liquid, and overall manufacturing capacity utilization and availability. It shows that for oral dry powder there are 14 total registrations, 65 pending applications, no contract product registrations pending, 17 products pending application, an average capacity utilization of 31.96%, and an average available capacity of 68.04%. For oral liquid it lists 15 total registrations, 60 pending applications, no contract product registrations pending, 19 products pending application, 23.70% average capacity utilization and 76.30% average available capacity. The conclusion summarizes that the document outlines production and quality control capacity utilized and available.
Original Description:
Original Title
Capacity Evaluation Conclusion.docx
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document provides data on pending applications and registered products for veterinary oral dry powder, oral liquid, and overall manufacturing capacity utilization and availability. It shows that for oral dry powder there are 14 total registrations, 65 pending applications, no contract product registrations pending, 17 products pending application, an average capacity utilization of 31.96%, and an average available capacity of 68.04%. For oral liquid it lists 15 total registrations, 60 pending applications, no contract product registrations pending, 19 products pending application, 23.70% average capacity utilization and 76.30% average available capacity. The conclusion summarizes that the document outlines production and quality control capacity utilized and available.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
2 views1 pageCapacity Evaluation Conclusion
Uploaded by
عارف حسینThis document provides data on pending applications and registered products for veterinary oral dry powder, oral liquid, and overall manufacturing capacity utilization and availability. It shows that for oral dry powder there are 14 total registrations, 65 pending applications, no contract product registrations pending, 17 products pending application, an average capacity utilization of 31.96%, and an average available capacity of 68.04%. For oral liquid it lists 15 total registrations, 60 pending applications, no contract product registrations pending, 19 products pending application, 23.70% average capacity utilization and 76.30% average available capacity. The conclusion summarizes that the document outlines production and quality control capacity utilized and available.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 1
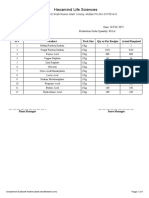
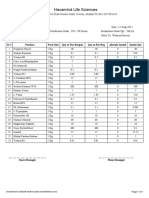
Section Name Manufacturer Pending Contract Contract Manufacturer Manufacturer
Total Application Product Products Capacity Capacity
Registration for Registration Pending Utilized Available
registration Application (Average) (Average)
Oral Dry Powder(Vet) 14 65 Nil 17 31.96 68.04
Oral Liquid(Vet) 15 60 Nil 19 23.70 76.30
CONCLUSION:
Production and QC capacity Utilized and Available is summarized as follow
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (344)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (120)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (399)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (73)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- Report TDPP Asi-Florfencol 20%Document23 pagesReport TDPP Asi-Florfencol 20%عارف حسینNo ratings yet
- Report TDPP Asi-Enrofloxacin 100 PDFDocument23 pagesReport TDPP Asi-Enrofloxacin 100 PDFعارف حسینNo ratings yet
- Butyricid & Organo New Devoloped FormulationDocument1 pageButyricid & Organo New Devoloped Formulationعارف حسینNo ratings yet
- Sop TĐPP Asi-Florfenicol 20%Document6 pagesSop TĐPP Asi-Florfenicol 20%عارف حسینNo ratings yet
- Certificate of Analysis Hexamind Life SciencesDocument1 pageCertificate of Analysis Hexamind Life Sciencesعارف حسینNo ratings yet
- Sop TĐPP Asi-Linco 4.4%Document6 pagesSop TĐPP Asi-Linco 4.4%عارف حسینNo ratings yet
- Report TDPP Asi-Linco 4.4%Document24 pagesReport TDPP Asi-Linco 4.4%عارف حسینNo ratings yet
- Sop TĐPP Asi-Florfenicol 20%Document6 pagesSop TĐPP Asi-Florfenicol 20%عارف حسینNo ratings yet
- Butyricid 2000 LitDocument1 pageButyricid 2000 Litعارف حسینNo ratings yet
- 5-List of Technical StaffDocument1 page5-List of Technical Staffعارف حسینNo ratings yet
- Batch DetailDocument7 pagesBatch Detailعارف حسینNo ratings yet
- Pulok KMukherjeeDocument21 pagesPulok KMukherjeeعارف حسینNo ratings yet
- Ar Engineering PDFDocument1 pageAr Engineering PDFعارف حسینNo ratings yet
- Quality PolicyDocument1 pageQuality Policyعارف حسینNo ratings yet
- Zorex 1000 lITDocument1 pageZorex 1000 lITعارف حسینNo ratings yet
- Process ModelDocument1 pageProcess Modelعارف حسینNo ratings yet
- Appointment Letter: Permanent EmploymentDocument3 pagesAppointment Letter: Permanent Employmentعارف حسینNo ratings yet
- Appointment Letter: Permanent EmploymentDocument3 pagesAppointment Letter: Permanent Employmentعارف حسینNo ratings yet
- Appointment LetterDocument2 pagesAppointment Letterعارف حسینNo ratings yet
- 16-Letter From Manufacturer Being Ready For InspectionDocument1 page16-Letter From Manufacturer Being Ready For Inspectionعارف حسینNo ratings yet
- Oranizalation Flow ChartDocument1 pageOranizalation Flow Chartعارف حسینNo ratings yet
- 16-Letter From Manufacturer Being Ready For InspectionDocument1 page16-Letter From Manufacturer Being Ready For Inspectionعارف حسینNo ratings yet
- 16-Letter From Manufacturer Being Ready For InspectionDocument1 page16-Letter From Manufacturer Being Ready For Inspectionعارف حسینNo ratings yet
- Appointment LetterDocument2 pagesAppointment Letterعارف حسینNo ratings yet
- Affidavit For The ProprietorDocument1 pageAffidavit For The Proprietorعارف حسینNo ratings yet
- Affidavit For The ProprietorDocument1 pageAffidavit For The Proprietorعارف حسینNo ratings yet
- Appointment Letter: Fatima RababDocument1 pageAppointment Letter: Fatima Rababعارف حسینNo ratings yet
- Appointment Letter: Fatima RababDocument1 pageAppointment Letter: Fatima Rababعارف حسینNo ratings yet
- Affidavit For The ProprietorDocument1 pageAffidavit For The Proprietorعارف حسینNo ratings yet