Professional Documents
Culture Documents
Cal Sheet
Uploaded by
alialnuimy960 ratings0% found this document useful (0 votes)
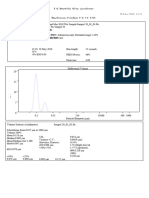
3 views1 pageThe document summarizes analytical data from quality control testing of a pharmaceutical product. It reports the product name and strength, batch number, manufacturing and expiration dates. Assay and dissolution testing results are provided, including weight and concentration measurements, average area counts, percentage of assay, and passing results within specified ranges for both tests.
Original Description:
Original Title
cal sheet
Copyright
© © All Rights Reserved
Available Formats
XLS, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe document summarizes analytical data from quality control testing of a pharmaceutical product. It reports the product name and strength, batch number, manufacturing and expiration dates. Assay and dissolution testing results are provided, including weight and concentration measurements, average area counts, percentage of assay, and passing results within specified ranges for both tests.
Copyright:
© All Rights Reserved
Available Formats
Download as XLS, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
3 views1 pageCal Sheet
Uploaded by
alialnuimy96The document summarizes analytical data from quality control testing of a pharmaceutical product. It reports the product name and strength, batch number, manufacturing and expiration dates. Assay and dissolution testing results are provided, including weight and concentration measurements, average area counts, percentage of assay, and passing results within specified ranges for both tests.
Copyright:
© All Rights Reserved
Available Formats
Download as XLS, PDF, TXT or read online from Scribd
You are on page 1of 1
Product Name
Strength 50 MG /ML
Batch No. : 700 Mfg. date : 12/22 Exp date: 12/24
Assay of API 90-120 %
Std. Data
Std. Wt mg Std. Conc. Av.Std. Area Std Potency Std. B. No STD Area
56.20 0.0490 84200 98 86446
Sample Data 86331
Sample Wt/mg Sample Conc. Sample Area % Assay Average 86679
50 0.0500 94531 107.72 77345
50 0.0500 94549 107.74
50 0.0500 92957 105.92
106.76
50 0.0500 92736 105.67 AV 84200
SD 4572.4589573518
RSD 5.4305
Diss of API NLT 80%
Std. Data
Std. Wt mg Std. Conc. Av.Std. Area Std Potency Std. B. No STD Area
57.80 0.1007 2322454 98 2332441
Sample Data 2301849
Sample Wt/mg Sample Conc. Sample Area % Assay Average 2333071
200 0.0444 1085937 103.82
200 0.0444 1085751 103.80
200 0.0444 1129174 107.95
200 0.0444 1129128 107.94
106.48
200 0.0444 1113158 106.42
200 0.0444 1139551 108.94 AV 2322454
SD 17846.9448739366
RSD 0.7685
You might also like
- 1-Particale Size Distribution Updated For All TestsDocument18 pages1-Particale Size Distribution Updated For All TestsAmmar AliNo ratings yet
- Rodsim:: Open Circuit Grinding SimulatorDocument38 pagesRodsim:: Open Circuit Grinding SimulatorJunior Romero ChavezNo ratings yet
- Lincolnville Schoold District Bus Data AliDocument23 pagesLincolnville Schoold District Bus Data AlialiNo ratings yet
- Al5 50Document3 pagesAl5 50Socrates LaskaridisNo ratings yet
- Table Statistics: Database: Database - BX Table: AssayDocument1 pageTable Statistics: Database: Database - BX Table: AssayAgung HidayantoNo ratings yet
- Ava7 50Document3 pagesAva7 50Socrates LaskaridisNo ratings yet
- Hasil Jihan BucinDocument4 pagesHasil Jihan BucinRiniNo ratings yet
- Al7 50Document3 pagesAl7 50Socrates LaskaridisNo ratings yet
- HJ5RPV-50 Product SpecificationsDocument4 pagesHJ5RPV-50 Product SpecificationsTo VT3No ratings yet
- EMES Pvt. LTD.: Natural Moisture ContentDocument5 pagesEMES Pvt. LTD.: Natural Moisture ContentShivsagar yadavNo ratings yet
- Ava5rn 50Document3 pagesAva5rn 50Socrates LaskaridisNo ratings yet
- Product Specifications: CharacteristicsDocument3 pagesProduct Specifications: Characteristicseslam salmonyNo ratings yet
- Cu EstaDocument2 pagesCu EstaNando YarangaNo ratings yet
- RegressionDocument6 pagesRegressionian92193No ratings yet
- Eviews KennyDocument3 pagesEviews KennyVeyrazzNo ratings yet
- MyfileDocument2 pagesMyfileoliviawang011231No ratings yet
- T1C1Document3 pagesT1C1arofanmuntok12No ratings yet
- Hydrometer - Lot-2, Chancho, 1.50Document1 pageHydrometer - Lot-2, Chancho, 1.50ALEX BNo ratings yet
- Regression Analysis: Source SS DF MS F P-ValueDocument8 pagesRegression Analysis: Source SS DF MS F P-ValueAlberto Andre Barboza VeraNo ratings yet
- Laxmi R7 90degDocument3 pagesLaxmi R7 90degAamir KhanNo ratings yet
- Product Specifications: AVA5-50FXDocument3 pagesProduct Specifications: AVA5-50FXhassan329100% (1)
- Oup 9Document26 pagesOup 9TAMIZHAN ANo ratings yet
- Excel PraktikumDocument11 pagesExcel PraktikumAndi Azzahra RatuNo ratings yet
- LS Particle Size Analyzer Beckman Coulter LS 13 320Document3 pagesLS Particle Size Analyzer Beckman Coulter LS 13 320hariNo ratings yet
- Schedule 40 PVC Pipe Dimensions & Pressure RatingsDocument1 pageSchedule 40 PVC Pipe Dimensions & Pressure RatingspicottNo ratings yet
- Schedule 40 PVC Pipe Dimensions & Pressure RatingsDocument1 pageSchedule 40 PVC Pipe Dimensions & Pressure RatingsprathapNo ratings yet
- Nama: Bernadetha Daud S NIM: B13.336 Prodi/jurusan: S1/AkuntansiDocument8 pagesNama: Bernadetha Daud S NIM: B13.336 Prodi/jurusan: S1/AkuntansiBernadetha Paula Daud SaputriNo ratings yet
- FXL 1480Document3 pagesFXL 1480KikafundaNo ratings yet
- Hydrometer - Lot-2, Chancho, 2.70Document1 pageHydrometer - Lot-2, Chancho, 2.70ALEX BNo ratings yet
- LognormalDocument2 pagesLognormalYves-donald MakoumbouNo ratings yet
- Gain & Loss FY EQUITY Report FNR352S106Document3 pagesGain & Loss FY EQUITY Report FNR352S106PALLAVI SHARMANo ratings yet
- Department of Civil Engineering - Materials Testing LaboratoryDocument24 pagesDepartment of Civil Engineering - Materials Testing LaboratoryChris JosephNo ratings yet
- HJ9-50 Product SpecificationsDocument3 pagesHJ9-50 Product SpecificationsTo VT3No ratings yet
- Syauqi - Nano Silika - Sampel 5 - 1273.nsz Measurement ResultsDocument3 pagesSyauqi - Nano Silika - Sampel 5 - 1273.nsz Measurement ResultsAhmad FarhanNo ratings yet
- Practical Report On: Concrete and MasonryDocument15 pagesPractical Report On: Concrete and MasonrySubodh Kumar KamalNo ratings yet
- Oup 8Document36 pagesOup 8TAMIZHAN ANo ratings yet
- Lampiran Glukosa DarahDocument4 pagesLampiran Glukosa DarahefendiNo ratings yet
- RCT7-WBC-4A-RNA Product SpecificationsDocument4 pagesRCT7-WBC-4A-RNA Product SpecificationsTo VT3No ratings yet
- Variogram Calculation: GeoviaDocument7 pagesVariogram Calculation: GeoviaIvanLzNo ratings yet
- Lab 2 WriteupDocument8 pagesLab 2 Writeupapi-702743009No ratings yet
- Samsung PN59D8000 CNET Review Calibration ResultsDocument7 pagesSamsung PN59D8000 CNET Review Calibration ResultsDavid KatzmaierNo ratings yet
- Heat Coil CalculationDocument2 pagesHeat Coil CalculationZhao XuanNo ratings yet
- Data Set 1aDocument6 pagesData Set 1aSufyan AshrafNo ratings yet
- AVA5-50FX Product SpecificationsDocument5 pagesAVA5-50FX Product SpecificationsPhi FeiNo ratings yet
- Mar 18, 2019 Report No.: Test DateDocument1 pageMar 18, 2019 Report No.: Test DateLeonardo MoraNo ratings yet
- Tugas - Heterogenitas - Raka Fajar NugrohoDocument4 pagesTugas - Heterogenitas - Raka Fajar NugrohoRaka NugrohoNo ratings yet
- Latihan 5 Frequencies Frequencies: StatisticsDocument19 pagesLatihan 5 Frequencies Frequencies: StatisticsuukumaediNo ratings yet
- Lab Report E11Document9 pagesLab Report E11rbaldwin8No ratings yet
- Comp ApplDocument10 pagesComp Applapi-3805289No ratings yet
- Schedule 80 PVC Pipe Dimensions & Pressure RatingsDocument1 pageSchedule 80 PVC Pipe Dimensions & Pressure RatingsprathapNo ratings yet
- Beckman Coulter LS - Sampel 20 - 01 - 03Document2 pagesBeckman Coulter LS - Sampel 20 - 01 - 03EriskaAgustinNo ratings yet
- Andrew LDF4 5-50Document4 pagesAndrew LDF4 5-50Hollmandavid VasquezNo ratings yet
- Viscosity Standard n4000 5181005110Document1 pageViscosity Standard n4000 5181005110Rachel McArdleNo ratings yet
- AnovaDocument2 pagesAnovaAthea TuboNo ratings yet
- Death RateDocument7 pagesDeath RateDahagam SaumithNo ratings yet
- Koreksi 2 - Excel Konsolidasi Mektan 2 - Kel 09Document15 pagesKoreksi 2 - Excel Konsolidasi Mektan 2 - Kel 09Malika KaylaniNo ratings yet
- Simple Multiple Regression (Taking All Scale Variable) : DescriptivesDocument26 pagesSimple Multiple Regression (Taking All Scale Variable) : Descriptivesrao saadNo ratings yet
- Hypothesis Testing Session 16Document28 pagesHypothesis Testing Session 16Mohit VermaNo ratings yet
- Estimation HVAC Work 1Document3 pagesEstimation HVAC Work 1Skylink Estimation TeamNo ratings yet