Professional Documents
Culture Documents
Difference Between Bioavailability and Bioequivalence
Uploaded by
prakash deshmukhOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Difference Between Bioavailability and Bioequivalence
Uploaded by
prakash deshmukhCopyright:
Available Formats
9/6/23, 6:15 PM Difference between Bioavailability and Bioequivalence
Menu
Difference between Bioavailability and
Bioequivalence
March 24, 2023
Bioavailability and bioequivalence studies are essential in the regulatory submission of
a new drug product or a generic drug product. Innovator drug product vs Generic drug
product. Studies on bioavailability and bioequivalence are necessary to verify
therapeutic equivalency. Frequently bioavailability and bioequivalence rely on
pharmacokinetics measures such as Area under the Curve (AUC) to assess the extent
of systemic exposure and Cmax and Tmax to assess the rate of systemic absorption. You
may read: Generic name of drugs, Brand name of Drugs, Generic drug/ medicine,
Brand name drug/medicine
Table of Contents
Definition of Bioavailability vs Bioequivalence
According to USFDA, Bioavailability is the rate and extent to which the active ingredient
or active moiety is absorbed from a drug product and becomes available at the site of
action [1].
According to EMA, The rate and extent to which a substance or its active moiety is
delivered from a pharmaceutical form and becomes available in the general circulation
(taking into consideration that the substance in the general circulation is in exchange
with the substance at the site of action).
Also, according to WHO, The rate and extent to which the active moiety is absorbed
from a pharmaceutical dosage form and becomes available at the site(s) of action.
Dose, Dosage, Dosage Form, and Dosage Regimen.
Whereas, according to USFDA, Bioequivalence means the absence of a significant
difference in the rate and extent to which the active ingredient or active moiety in
pharmaceutical equivalents or pharmaceutical alternatives becomes available at the
https://pharmaeducation.net/bioavailability-and-bioequivalence/#:~:text=Bioavailability is measured by using three main pharmacokinetic variable… 1/11
9/6/23, 6:15 PM Difference between Bioavailability and Bioequivalence
site of drug action when administered at the same molar dose under similar conditions
in an appropriately designed study [1].
A short expression of Bioavailability vs Bioequivalence
BA or F is a short expression of bioavailability. BE is a short expression of
bioequivalence.
Which application type requires a BA or BE?
Bioavailability (BA) information of a drug product is essential for Investigational New
Drug Application (IND), New Drug Application (NDA), and NDA supplements [2].
On the other hand, a bioequivalence study with pharmacokinetic endpoints of a drug
product is essential for Abbreviated New Drug Application (ANDA [3].
The Main focus of Bioavailability vs Bioequivalence
Studies on bioavailability concentrate on figuring out how and when a drug substance
escapes from the oral dosage form and reaches the site of action.
On the other hand, bioequivalence studies focus on the performance of the drug
product and usually involve comparisons of two drug products: T and R. Here T is the
test product. In the United States, R is termed the RLD. In the WHO document, R is
termed the “comparator pharmaceutical product” (CPP) [5].
Reference product for Bioavailability vs Bioequivalence
Generally, for the bioavailability study of a new drug product, the reference product
should be a solution, suspension, or intravenous (IV) dosage form of the new drug.
Conducting a BA study with an intravenous (IV) reference product enables assessment
of the impact of the route of administration on BA and defines the absolute BA of the
drug released from the drug product. Conducting a BA study comparing one
formulation to another enables an assessment of relative BA.
Whereas, for the bioequivalence study of a generic drug product, the reference product
should be the innovator drug product or reference listed drug. BE study is to measure
and compare formulation performance between two or more drug products. However,
https://pharmaeducation.net/bioavailability-and-bioequivalence/#:~:text=Bioavailability is measured by using three main pharmacokinetic variable… 2/11
9/6/23, 6:15 PM Difference between Bioavailability and Bioequivalence
in the case of bioequivalence studies of a new formulation of an innovator drug
product, the reference product should receive approval. For regulatory approval of a
generic drug product (test product, T) must be bioequivalent to the Reference listed
drug product R.
What are the objectives of Bioavailability vs Bioequivalence?
Objectives of Bioavailability [2, 6]
1. Bioavailability data provide an estimation of the fraction of the drug absorption,
distribution, metabolism, excretion, and the effects of food on the absorption of
the drug, dose proportionality, or linearity in the pharmacokinetics of the active
moieties and when appropriate inactive moieties.
2. BA provides useful information to establish dosage regimens and to support
drug labeling, such as distribution and elimination characteristics of the drug.
3. Bioavailability of drug products aids in the FDA’s evaluation of the safety and
effectiveness of a product in IND, NDA, or NDA supplements.
Objectives of Bioequivalence [3, 6]
1. To measure and compare formulation performance between two or more drug
products.
2. Bioequivalence provides a link between pivotal and early clinical trial
formulation.
3. To determine the therapeutic equivalence between the pharmaceutical
equivalence generic drug product and a corresponding reference listed drug.
4. For an innovator drug product, bioequivalence studies establish the performance
of the new formulation product intended for marketing with an already approved
innovator drug product to evaluate the safety/efficacy of the new formulation
drug product. Efficacy vs Potency.
5. To provide information on product quality and performance when there are
changes in components, composition, and method of manufacture after approval
of the drug product.
Measurement of Bioavailability vs Bioequivalence
Measurement of Bioavailability
https://pharmaeducation.net/bioavailability-and-bioequivalence/#:~:text=Bioavailability is measured by using three main pharmacokinetic variable… 3/11
9/6/23, 6:15 PM Difference between Bioavailability and Bioequivalence
Bioavailability is an indirect or surrogate measure of the rate and extent to which the
drug substance or active moiety is absorbed from a drug product and becomes
available at its target sites of action. Bioavailability is measured by using three main
pharmacokinetic variables:
The area under the blood drug concentration versus time curve (AUC)
The maximum blood concentration (Cmax)
The time to reach maximum concentration (Tmax)
Measurement of Bioequivalence
On the other hand, Bioequivalence (BE) is a surrogate measure of in vivo drug product
performance, and dissolution profile comparisons as a measure of in vitro drug product
performance. BE has been established via bioavailability testing. Bioequivalence is
measured based on the relative bioavailability of the innovator drug product versus the
generic drug product.
It is important to note that bioequivalence studies are also performed for innovator drug
products in some situations such as: between early and late clinical trial formulations;
between formulations used in clinical trials and stability studies, if different; or between
clinical trial formulations and to-be-marketed drug products, if different; or between
equivalence of product strengths; between marketed innovator product and a new
formulation of marketed innovator product (change in one or more excipients).
https://pharmaeducation.net/bioavailability-and-bioequivalence/#:~:text=Bioavailability is measured by using three main pharmacokinetic variable… 4/11
9/6/23, 6:15 PM Difference between Bioavailability and Bioequivalence
Bioequivalence is based on a comparison of ratios where the ratio of generic to
innovator for each pharmacokinetic variable does not differ by more than 8:10, this is
how the range for the confidence intervals is defined: 8/10 = 0.80 gives the lower limit
and 10/8 = 1.25 gives the upper limit. The 90% confidence intervals for the ratios of
[4]
both Cmax and AUC should be contained within the limits of 0.80–1.25 . The FDA
considers two products bioequivalent if the 90% confidence intervals (CI) of the relative
mean Cmax, AUC (0–t), and AUC (0–∞) of the test (e.g. generic drug product) to RLD
(e.g. innovator drug product) should be within 80% to 125% in the fasting state. [1].
Nature of Study
Bioavailability studies are expletory. Whereas, bioequivalence studies are a
confirmatory or statutory term that requires ANDA applicants to demonstrate, among
other things, that the proposed generic product is bioequivalent to its reference listed
drug.
The interrelation between bioavailability and bioequivalence
Bioavailability is not been assessed via bioequivalence testing. Bioavailability is
assessed using three main pharmacokinetic variables
The area under the blood drug concentration versus time curve (AUC)
The maximum blood concentration (Cmax)
The time to reach maximum concentration (Tmax)
On the other hand, bioequivalence is assessed via bioavailability testing.
Example of Bioavailability vs bioequivalence
Examples of Bioavailability: after oral administration of 20 mg of Rabeprazole
Sodium Delayed-Release tablet, 52% of this drug is absorbed unchanged
systematically as compared to intravenous administration. So, the absolute
bioavailability of this drug product is 52% [7].
Examples of bioequivalence: two medicines are bioequivalent if there is no clinically
significant difference in their bioavailability. Based on the bioequivalence studies under
fasting and fed conditions, Idiazole, Rabeprazolnatrium, and Rodesa 20 mg tablets are
bioequivalent with Pariet 20 mg. Here, Idiazole, Rabeprazolnatrium, and Rodesa 20 mg
https://pharmaeducation.net/bioavailability-and-bioequivalence/#:~:text=Bioavailability is measured by using three main pharmacokinetic variable… 5/11
9/6/23, 6:15 PM Difference between Bioavailability and Bioequivalence
tablets are the generic drug product. Pariet is the RLD of Rabeprazole Sodium Delay
Release tablet in the UK and Australian markets and Aciphex is the RLD of
Rabeprazole Sodium Delay Release tablet for the US market.
Summary of the difference between bioavailability and
bioequivalence
Features Bioavailability Bioequivalence
Bioequivalence means the
absence of a significant difference
in the rate and extent to which the
Bioavailability is the rate and
active ingredient or active moiety in
extent to which the active
pharmaceutical equivalents or
ingredient or active moiety is
Definition pharmaceutical alternatives
absorbed from a drug product
becomes available at the site of
and becomes available at the
drug action when administered at
site of action.
the same molar dose under similar
conditions in an appropriately
designed study.
Short
BA or F BE
expression
Investigational New Drug
Application (IND), New Drug Abbreviated New Drug Application
Require for
Application (NDA), and NDA (ANDA
supplements.
On determining the process On the performance of the drug
and time frame by which a product and usually involve
Main focus drug substance is released comparisons of two drug products:
from the oral dosage form and T (test product) and (Innovator
moves to the site of action. drug product or RLD).
Reference A solution, suspension, or Innovator drug product or
product intravenous (IV) dosage form reference listed drug (RLD).
https://pharmaeducation.net/bioavailability-and-bioequivalence/#:~:text=Bioavailability is measured by using three main pharmacokinetic variable… 6/11
9/6/23, 6:15 PM Difference between Bioavailability and Bioequivalence
of the new drug.
A. To measure and compare
A. Bioavailability data provide formulation performance between
an estimation of the fraction of two or more drug products.
the drug absorption, B. Bioequivalence provides a link
distribution, metabolism, between the pivotal and early
excretion, and the effects of clinical trial formulation.
food on the absorption of the C. To determine the therapeutic
drug, dose proportionality, or equivalence between the
linearity in the pharmaceutical equivalence
pharmacokinetics of the active generic drug product and a
moieties and when corresponding reference listed
appropriate inactive moieties. drug.
D. For an innovator drug product,
Objectives
B. BA provides useful bioequivalence studies establish
information to establish the performance of the new
dosage regimens and to formulation product intended for
support drug labeling, such as marketing with an already
distribution and elimination approved innovator drug product to
characteristics of the drug. evaluate the safety/efficacy of the
C. Bioavailability of drug new formulation drug product.
products aids in the FDA’s E. To provide information on
evaluation of the safety and product quality and performance
effectiveness of a product in when there are changes in
IND, NDA, or NDA components, composition, and
supplements. method of manufacture after
approval of the drug product.
Measurement Bioavailability is an indirect or Bioequivalence (BE) is a surrogate
surrogate measure of the rate measure of in vivo drug product
and extent to which the drug performance and dissolution profile
substance or active moiety is comparisons as a measure of in
absorbed from a drug product vitro drug product performance. BE
and becomes available at its has been established via
target sites of action. bioavailability testing.
https://pharmaeducation.net/bioavailability-and-bioequivalence/#:~:text=Bioavailability is measured by using three main pharmacokinetic variable… 7/11
9/6/23, 6:15 PM Difference between Bioavailability and Bioequivalence
Bioavailability is measured by Bioequivalence is measured based
using three main on the relative bioavailability of
pharmacokinetic variables: the innovator drug
AUC, Cmax, Tmax. product versus the generic drug
product.
Nature of
Expletory study Confirmatory or statutory study
Study
Not assesse via
Interrelation Assess via bioavailability testing.
bioequivalence testing.
Based on the bioequivalence
studies under fasting and fed
After oral administration of 20
conditions, Idiazole,
mg of Rabeprazole Sodium
Rabeprazolnatrium, and Rodesa
Delayed-Release tablet, 52%
20 mg tablets are bioequivalent
of this drug is absorbed
with Pariet 20 mg. Here, Idiazole,
Examples unchanged systematically as
Rabeprazolnatrium, and Rodesa
compared to intravenous
20 mg tablets are the generic drug
administration. So, the
product. Pariet is the RLD of
absolute bioavailability of this
Rabeprazole Sodium Delayed-
drug product is 52%.
Release tablet in the UK and
Australia markets.
References
1. Center for Drug Evaluation and Research (2003). “Guidance for Industry:
Bioavailability and Bioequivalence Studies for Orally Administered Drug Products –
General Considerations”. United States Food and Drug Administration.
2. Center for Drug Evaluation and Research (2003). “Guidance for Industry:
Bioavailability Studies Submitted in NDAs or INDs — General Considerations”.
United States Food and Drug Administration.
3. Center for Drug Evaluation and Research (2003). “Guidance for Industry:
Bioequivalence Studies with Pharmacokinetic Endpoints for Drugs Submitted Under
https://pharmaeducation.net/bioavailability-and-bioequivalence/#:~:text=Bioavailability is measured by using three main pharmacokinetic variable… 8/11
9/6/23, 6:15 PM Difference between Bioavailability and Bioequivalence
an ANDA — General Considerations”. United States Food and Drug Administration.
4. Birkett D. Generics – equal or not? Australian Prescriber, 2003(26):85-7.
5. WHO Guidance for organizations performing in vivo bioequivalence studies. WHO
Technical Report Series No. 996, 2016, Annex 9
6. United States Pharmacopeia and National Formulary (USP 43–NF 38). United
States Pharmacopeial Convention; 2020. Accessed January 01, 2021.
7. Highlights of prescribing information of ACIPHEX® (rabeprazole sodium) Delayed-
Release Tablets. Eisai Inc. Retrieved 27 February 2021.
Pharma Difference
Difference between Packing and Packaging
AUC in Pharmacokinetics
DIFFERENCES
Difference between Bioavailability and Bioequivalence
Difference between Packing and Packaging
Difference between Signs and Symptoms
Difference between Drug Design and Drug Development
IV Bolus vs IV Infusion of Parenteral Drug Product
Difference between Compression and Compaction of Tablets
https://pharmaeducation.net/bioavailability-and-bioequivalence/#:~:text=Bioavailability is measured by using three main pharmacokinetic variable… 9/11
9/6/23, 6:15 PM Difference between Bioavailability and Bioequivalence
......................................................................................................
TECHNOLOGY
Friability Test of Tablets, Granules, Spheroids
Quality control tests of tablets or Evaluation of tablets
Tablet Compression Machine: Types, Functional Parts
Excipients for Tablets with examples
Colouring Agents, Colorants, Color Additives in Pharmaceutical
Tablet Coating Defects and Remedies
Examples of suspension (Pharmacy, Chemistry, Food)
Tablet defects
................................................................................................................
CLARIFICATION
Generic medicines vs Brand name medicines
Difference between Ointment Cream Paste Gel Lotion Jelly
Agonist, Partial Agonist, Antagonist, and Inverse Agonist
Dose, Dosage, Dosage Form, Dosage Regimen
Enzyme, Coenzyme, Apoenzyme, Holoenzyme, and Cofactor
Prebiotics, Probiotics, Synbiotics, Psychobiotics
Vitamin, Pro-vitamin, Antivitamin
https://pharmaeducation.net/bioavailability-and-bioequivalence/#:~:text=Bioavailability is measured by using three main pharmacokinetic variabl… 10/11
9/6/23, 6:15 PM Difference between Bioavailability and Bioequivalence
Designed & Developed by Waled B. & Sohag M. S. U
Facebook Twitter LinkedIn
Patreon Mastodon Pinterest
Content, services, and products on our website are for informational purposes only.
Pharmaeducation.net does not provide medical advice, diagnosis, or treatment.
About Us Privacy Policy Contact Us
©2020-2023 PharmaEducation.Net. All rights reserved.
https://pharmaeducation.net/bioavailability-and-bioequivalence/#:~:text=Bioavailability is measured by using three main pharmacokinetic variabl… 11/11
You might also like
- Fast Facts: Biosimilars in Hematology and Oncology: Biologics and biosimilars - getting decisions rightFrom EverandFast Facts: Biosimilars in Hematology and Oncology: Biologics and biosimilars - getting decisions rightNo ratings yet
- Final ProjectDocument119 pagesFinal ProjectSaurabh KumarNo ratings yet
- Pkin08 Soal Biofar Pak BoesroDocument117 pagesPkin08 Soal Biofar Pak Boesroabu_59No ratings yet
- Drug Absorption PDFDocument117 pagesDrug Absorption PDFAndhia DhiyaNo ratings yet
- Hemanagadurga2019 Bioavailability StudiesDocument17 pagesHemanagadurga2019 Bioavailability Studiesraghuraj75No ratings yet
- Review Article: Barbara Davit, April C. Braddy, Dale P. Conner, and Lawrence X. YuDocument17 pagesReview Article: Barbara Davit, April C. Braddy, Dale P. Conner, and Lawrence X. Yulhthang1990No ratings yet
- Bioavailability and BioequivalenceDocument28 pagesBioavailability and Bioequivalencekulbhushan singhNo ratings yet
- Bioequivalence: Faculty of Pharmacy, Nursing and Health Professions Master in Industrial Pharmaceutical TechnologyDocument15 pagesBioequivalence: Faculty of Pharmacy, Nursing and Health Professions Master in Industrial Pharmaceutical TechnologyMayson Bali100% (1)
- Bioequivalence: Faculty of Pharmacy, Nursing and Health Professions Master in Industrial Pharmaceutical TechnologyDocument16 pagesBioequivalence: Faculty of Pharmacy, Nursing and Health Professions Master in Industrial Pharmaceutical TechnologyMayson BaliNo ratings yet
- Guideline On Bioavailability and BioequivalanceDocument26 pagesGuideline On Bioavailability and BioequivalanceGima Amezia SariNo ratings yet
- BIOEQUI2Document5 pagesBIOEQUI2Laiba KhalidNo ratings yet
- Bioavailability and Bieq Review ArticleDocument15 pagesBioavailability and Bieq Review ArticlevinayNo ratings yet
- Ba-Be PDFDocument30 pagesBa-Be PDFUswatun Hasanah7201No ratings yet
- 24 13v3i2 5Document9 pages24 13v3i2 5gopalNo ratings yet
- Under Guidance of Mr. Mali K.K. (Assistant Professor) : 1 Yspm, YtcDocument41 pagesUnder Guidance of Mr. Mali K.K. (Assistant Professor) : 1 Yspm, YtcSabiruddin Mirza DipuNo ratings yet
- Bio Availability and Bioequivalane-LastDocument13 pagesBio Availability and Bioequivalane-LastAhmedothman62100% (1)
- Thailand BABE GuidelineDocument27 pagesThailand BABE GuidelineDianeNo ratings yet
- 8 Ba Be FdaDocument36 pages8 Ba Be Fdaraju narayana padalaNo ratings yet
- Asean Guidelines For: Final Draft: 21 July 2004Document29 pagesAsean Guidelines For: Final Draft: 21 July 2004Jone YingNo ratings yet
- What Is Biopharmaceutics?: Brand NameDocument10 pagesWhat Is Biopharmaceutics?: Brand NameAhmad Jamal HashmiNo ratings yet
- Nomenclature terms for therapeutic equivalenceDocument4 pagesNomenclature terms for therapeutic equivalencekurutalaNo ratings yet
- Guidance for Oral Extended Release Generic DrugsDocument14 pagesGuidance for Oral Extended Release Generic DrugsAnonymous Se5IdneSpNo ratings yet
- In Vitro - in Vivo Correlation From Theory To ApplicationsDocument21 pagesIn Vitro - in Vivo Correlation From Theory To ApplicationsahlawamarhabaNo ratings yet
- 2 Amrutaobjectivesandconsiderationsinbioavailabilitystudy 130211000517 Phpapp01 PDFDocument31 pages2 Amrutaobjectivesandconsiderationsinbioavailabilitystudy 130211000517 Phpapp01 PDFsaurabh chaturvediNo ratings yet
- Comparing Generic and Innovator Drugs A Review ofDocument16 pagesComparing Generic and Innovator Drugs A Review ofPrachi PandeyNo ratings yet
- Bioavailability and BioequivalenceDocument6 pagesBioavailability and BioequivalenceDharmesh PatelNo ratings yet
- Bioavailability and Bioequivalence: Understanding BA and BE StudiesDocument53 pagesBioavailability and Bioequivalence: Understanding BA and BE StudiesWalaa abo foolNo ratings yet
- Bioavailability and Bioequivalence (BABE)Document4 pagesBioavailability and Bioequivalence (BABE)Wingielyn Honculada BaldozaNo ratings yet
- Note For Guidance On The Investigation of Bioavailability & BioequivalenceDocument19 pagesNote For Guidance On The Investigation of Bioavailability & BioequivalenceAhmed AliNo ratings yet
- Bioavailability and BioequivalenceDocument56 pagesBioavailability and Bioequivalenceنور الهدى100% (1)
- Bioequevalance StudiesDocument55 pagesBioequevalance StudiesMubammad MursaleenNo ratings yet
- Bioequivalence - An Overview of Statistical ConceptsDocument8 pagesBioequivalence - An Overview of Statistical ConceptsAhmad Abdullah Najjar100% (2)
- Generic Medicines: Interchangeability of WHO-prequalified GenericsDocument6 pagesGeneric Medicines: Interchangeability of WHO-prequalified GenericsGuide LPNo ratings yet
- In Vitro in Vivo Correlation Ivivc A Strategic Tool in Drug Development Jbb.S3 001Document12 pagesIn Vitro in Vivo Correlation Ivivc A Strategic Tool in Drug Development Jbb.S3 001Jerome K GeorgeNo ratings yet
- Bioavailability and Bioequivalence GuideDocument59 pagesBioavailability and Bioequivalence GuideAbdi JifarNo ratings yet
- ASEAN BE Guidelines-Amended According To1st BABE TWG15th PPWGDocument29 pagesASEAN BE Guidelines-Amended According To1st BABE TWG15th PPWGTarikNo ratings yet
- Regulatory Perspectives On Strength-Dependent Dissolution ProfilesDocument11 pagesRegulatory Perspectives On Strength-Dependent Dissolution ProfilesJminsNo ratings yet
- Pre 66222Document18 pagesPre 66222Md. JewelNo ratings yet
- Guidelines For Bioavailability and Bioequivalence Studies: A ReviewDocument6 pagesGuidelines For Bioavailability and Bioequivalence Studies: A ReviewNagabhushanam MaddiNo ratings yet
- Importance of Bioavailability and Bioequivalence Studies (BABEDocument9 pagesImportance of Bioavailability and Bioequivalence Studies (BABEJessie ChuNo ratings yet
- Drug Product Performance GuideDocument119 pagesDrug Product Performance GuideSachin BagewadiNo ratings yet
- IVIVCDocument21 pagesIVIVCashpharma007No ratings yet
- Bioavailability and BioequivalenceDocument39 pagesBioavailability and Bioequivalenceنور الهدىNo ratings yet
- BASIC PHARMACOKINETICS - CHAPTER 8: BioavailabilityDocument111 pagesBASIC PHARMACOKINETICS - CHAPTER 8: BioavailabilityDrHeba100% (10)
- Importance of in Vitro in Vivo Studies IDocument23 pagesImportance of in Vitro in Vivo Studies IDan Martínez Silva100% (1)
- MPP205 Exp No 24 - 25Document2 pagesMPP205 Exp No 24 - 25KishoreNo ratings yet
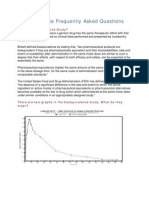
- Bioequivalence Frequently Asked Questions: What Is Bioequivalence Study?Document4 pagesBioequivalence Frequently Asked Questions: What Is Bioequivalence Study?Ahmed HasanNo ratings yet
- Regulatory Requirement of Similar Biologic For Marketing AuthorizationDocument19 pagesRegulatory Requirement of Similar Biologic For Marketing AuthorizationSatheesh SachinNo ratings yet
- Biowaiver: An Alternative To in Vivo Pharmacokinetic Bioequivalence StudiesDocument8 pagesBiowaiver: An Alternative To in Vivo Pharmacokinetic Bioequivalence StudiesVinz AlvarezNo ratings yet
- Bioavailability: by Abera JDocument59 pagesBioavailability: by Abera JAbera JamboNo ratings yet
- Definition of BiopharmaceuticsDocument3 pagesDefinition of BiopharmaceuticsvafaashkNo ratings yet
- Drug Delivery Inhalation BioequivalenceDocument12 pagesDrug Delivery Inhalation Bioequivalenceparinaferns0% (1)
- Bioavailability and BioequivalenceDocument81 pagesBioavailability and BioequivalenceNiharika ModiNo ratings yet
- In-Vitro-In-Vivo Correlation Definitions and Regu Ce: Latory GuidanDocument11 pagesIn-Vitro-In-Vivo Correlation Definitions and Regu Ce: Latory GuidankripenderNo ratings yet
- BiopharmaceuticsDocument21 pagesBiopharmaceuticsSilvy100% (1)
- Loshieni Shri - Drug Product PerformanceDocument10 pagesLoshieni Shri - Drug Product PerformanceloshieniNo ratings yet
- Biological Aspects of FormulationsDocument8 pagesBiological Aspects of FormulationsKabirNo ratings yet
- Bioavailability and BioequivalenceDocument47 pagesBioavailability and BioequivalenceGiovanne BuendiaNo ratings yet
- Guided Equivalence StudyDocument30 pagesGuided Equivalence StudyTejas PatelNo ratings yet
- Considerations For Migrating A Drug Product Presentation From Vial To Pre-Filled SyringeDocument4 pagesConsiderations For Migrating A Drug Product Presentation From Vial To Pre-Filled Syringeprakash deshmukhNo ratings yet
- An Purge Validation 2022Document4 pagesAn Purge Validation 2022prakash deshmukhNo ratings yet
- Difference Between Recording and Monitoring - WikiDiffDocument2 pagesDifference Between Recording and Monitoring - WikiDiffprakash deshmukhNo ratings yet
- Difference Between Document and RecordDocument13 pagesDifference Between Document and Recordprakash deshmukhNo ratings yet
- Introduction To MRNA ManufacturingDocument3 pagesIntroduction To MRNA Manufacturingprakash deshmukhNo ratings yet
- 5 Key Components of A Compliant Disinfectant Efficacy Testing Package (Part 2)Document4 pages5 Key Components of A Compliant Disinfectant Efficacy Testing Package (Part 2)prakash deshmukhNo ratings yet
- Overcome The Challenges of Small Batch Production While Maximizing YieldDocument2 pagesOvercome The Challenges of Small Batch Production While Maximizing Yieldprakash deshmukhNo ratings yet
- 23 03 Po Annex1 EbookDocument23 pages23 03 Po Annex1 EbookDincer SELENNo ratings yet
- M RNA ManufacturingDocument1 pageM RNA Manufacturingprakash deshmukhNo ratings yet
- Environmental Monitoring - Live Online Training - ECA Academy May 2023Document5 pagesEnvironmental Monitoring - Live Online Training - ECA Academy May 2023prakash deshmukhNo ratings yet
- Real-Time Viable Particle Detection For Definitive Root Cause InvestigationDocument3 pagesReal-Time Viable Particle Detection For Definitive Root Cause Investigationprakash deshmukhNo ratings yet
- ISO 17025 Clause Wise ChangesDocument10 pagesISO 17025 Clause Wise ChangesaasNo ratings yet
- How To Establish An Aseptic Gowning Qualification ProgramDocument3 pagesHow To Establish An Aseptic Gowning Qualification Programprakash deshmukhNo ratings yet
- Aqueous Cleaning - BenefitsDocument10 pagesAqueous Cleaning - Benefitsprakash deshmukhNo ratings yet
- VHP BI From Mesa LabDocument1 pageVHP BI From Mesa Labprakash deshmukhNo ratings yet
- An Introduction To Environmental Monitoring Cleaning For Aseptic EnvironmentsDocument5 pagesAn Introduction To Environmental Monitoring Cleaning For Aseptic Environmentsprakash deshmukhNo ratings yet
- Google XRay LinkedIn 2022 821254898-7Document166 pagesGoogle XRay LinkedIn 2022 821254898-7prakash deshmukhNo ratings yet
- Quality Assurance Quality Control and Quality Management Systems Clarifying ConfusionDocument2 pagesQuality Assurance Quality Control and Quality Management Systems Clarifying Confusionprakash deshmukhNo ratings yet
- 5.redundant Sterilizing Filtration - MR - SomDocument34 pages5.redundant Sterilizing Filtration - MR - SomTaNo ratings yet
- Article Gas Replace Dye Ingress 2022Document4 pagesArticle Gas Replace Dye Ingress 2022prakash deshmukhNo ratings yet
- mRNA MFGDocument4 pagesmRNA MFGprakash deshmukhNo ratings yet
- Google XRay LinkedIn 2022 821254898-7Document166 pagesGoogle XRay LinkedIn 2022 821254898-7prakash deshmukhNo ratings yet
- Cleanroom Microbiology 101 Identifying Controlling Sources of ContaminationDocument4 pagesCleanroom Microbiology 101 Identifying Controlling Sources of Contaminationprakash deshmukhNo ratings yet
- Ebook Pharma 2018Document39 pagesEbook Pharma 2018prakash deshmukhNo ratings yet
- Validation Book, PDFDocument216 pagesValidation Book, PDFQA-KKV IndofarmaNo ratings yet
- GMP-IsO Quality Audit Manual For Healthcare Manufacturers and Their Suppliers InterpharmCRC Sixth Edition VOLUME 1Document459 pagesGMP-IsO Quality Audit Manual For Healthcare Manufacturers and Their Suppliers InterpharmCRC Sixth Edition VOLUME 1Shivanand Bharti80% (15)
- An Purge Validation 2022Document4 pagesAn Purge Validation 2022prakash deshmukhNo ratings yet
- Leadership 101Document112 pagesLeadership 101prakash deshmukhNo ratings yet
- (Oklahoma Notes) Pamela S. Miles M.D., William F. Rayburn M.D., J. Christopher Carey M.D. (Auth.) - Obstetrics and Gynecology (1994, Springer US)Document224 pages(Oklahoma Notes) Pamela S. Miles M.D., William F. Rayburn M.D., J. Christopher Carey M.D. (Auth.) - Obstetrics and Gynecology (1994, Springer US)Muskan DasNo ratings yet
- 5 - Patients' Rights and ResponsibilitiesDocument12 pages5 - Patients' Rights and ResponsibilitiesMohammed HussienNo ratings yet
- The Long and Short of The Issue of Counsellors - The Star OnlineDocument2 pagesThe Long and Short of The Issue of Counsellors - The Star OnlinefareedhashimNo ratings yet
- Clinico-Aetiological Profile of Pancytopenia in Paediatric PracticeDocument4 pagesClinico-Aetiological Profile of Pancytopenia in Paediatric PracticeDesi AdiyatiNo ratings yet
- Medical Student CV Free PDFDocument4 pagesMedical Student CV Free PDFAngelo SalvadorNo ratings yet
- Cancer Epidemiology Assignment Charity NewDocument8 pagesCancer Epidemiology Assignment Charity NewSummer LarinNo ratings yet
- NCLEX Review Question 101-200Document14 pagesNCLEX Review Question 101-200Felimon BugtongNo ratings yet
- Paramedic Inter Facility Transfer TrainingDocument168 pagesParamedic Inter Facility Transfer TrainingnikunjhirparaNo ratings yet
- Occupational Therapy Initial EvaluationDocument10 pagesOccupational Therapy Initial EvaluationNerissa BanguiNo ratings yet
- Drug Study On Certain DrugsDocument10 pagesDrug Study On Certain DrugsMelvin D. RamosNo ratings yet
- 9 Top Benefits of KarakkayaDocument4 pages9 Top Benefits of KarakkayaAcharla SatyanarayanaNo ratings yet
- Maxillectomy A ReviewDocument17 pagesMaxillectomy A ReviewDr. T. Balasubramanian100% (3)
- Manual urinalysis guideDocument37 pagesManual urinalysis guideAdhya TiaraNo ratings yet
- Mask LTR 6.16 PDF No HLDocument14 pagesMask LTR 6.16 PDF No HLJoshua LingNo ratings yet
- Pressuring of Acupoints As A Complement To The - 2019 - Journal of AcupunctureDocument9 pagesPressuring of Acupoints As A Complement To The - 2019 - Journal of AcupunctureMatias DamiánNo ratings yet
- Hospital Standards 2021 - GAHARDocument314 pagesHospital Standards 2021 - GAHARKhalid ElwakilNo ratings yet
- Open - Oregonstate.education-Foundations of EpidemiologyDocument11 pagesOpen - Oregonstate.education-Foundations of EpidemiologyAMANE COTTAGE HOSPITALNo ratings yet
- Principal Letter Opposed ImmunizationDocument2 pagesPrincipal Letter Opposed Immunizationmpnaidu13111998100% (1)
- 26 Oct DRAFT Conference Program - Concurrent ProgramDocument25 pages26 Oct DRAFT Conference Program - Concurrent ProgramLucy MedinaNo ratings yet
- ITOM Group Assignment Issues at SCHDocument12 pagesITOM Group Assignment Issues at SCHzeeraNo ratings yet
- CDFDocument76 pagesCDFmishra.nagendra7311No ratings yet
- Otc Template Pi PholcodineDocument3 pagesOtc Template Pi PholcodineLucinda RuckleyNo ratings yet
- Hand and Upper ExtremityDocument722 pagesHand and Upper ExtremityPaul Radulescu - Fizioterapeut88% (8)
- 2016 ProgramDocument30 pages2016 ProgramctyvteNo ratings yet
- Femto-Lasik laser eye surgery explainedDocument4 pagesFemto-Lasik laser eye surgery explainedvrlftrNo ratings yet
- Iwq 0 K J98 V 8 KFPV 5 W 4 ZH 5 Z Q08 NJ Cy 30 PPBAo MXX 4 LDocument243 pagesIwq 0 K J98 V 8 KFPV 5 W 4 ZH 5 Z Q08 NJ Cy 30 PPBAo MXX 4 Lmaleknour2000No ratings yet
- Unidad 2. Tarea 2. EscrituraDocument5 pagesUnidad 2. Tarea 2. EscrituraAndrea ChavarroNo ratings yet
- Ellison - Hidden Truth About Cholesterol-Lowering Drugs (2006) PDFDocument42 pagesEllison - Hidden Truth About Cholesterol-Lowering Drugs (2006) PDFridwansurono100% (2)
- Steps in Outbreak InvestigationDocument34 pagesSteps in Outbreak Investigationsharmaine peroNo ratings yet
- Psychiatric MisadventuresDocument10 pagesPsychiatric MisadventuresRob_212No ratings yet