Professional Documents
Culture Documents
Hammet Eqn
Uploaded by
Deepan KumarOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Hammet Eqn
Uploaded by
Deepan KumarCopyright:
Available Formats
Autumn 2004
Linear Free Energy Relationships
Linear free energy relationships are attempts to develop quantitative relationships between structure and activity. Consider a particular reaction between two substrates. We might carry out a series of reactions by varying one of the reactants slightly, for example by examining substituents with a range of electronegativities. We might expect that the reaction rate, or the position of the equilibrium between reactants and products, will change as we change the reactant in this way. If the same series of of changes in conditions affects a second reaction in exactly the same way as it affected the first reaction, we say that there exists a linear free energy relationship between the two sets of effects. Such relationships can be useful in helping to elucidate reaction mechanisms and in predicting rates or equilibria.
2
Autumn 2004
The Hammett Equation
One of the earliest examples of a LFER between:
the rate of base catalysed hydrolysis of a group of ethyl esters to form a series of carboxylic acids. the equilibrium position of the ionisation in water of ! the corresponding group of acids.
RCOOEt + OH " #k $ RCOO" + EtOH #
RCOOH + OH " #K $ # RCOO" + H 2O %# #
A direct relationship was found between these processes for a specific set ! of compounds, the p- and m-substituted benzoic acids (R=Ar).
log k = " logK + C
Caveats: Ortho isomers do not fall on the line.
Aliphatic acids do not fall on the line.
Autumn 2004
The Hammett Equation
Why do ortho isomers and aliphatic compounds not exhibit the straight line relationship? Steric considerations:
Crowding is increased in the tetrahedral transition state for o-isomers. Flexibility of aliphatic compounds means that correlation between transition state structure and equilibrium position may not be strong.
Autumn 2004
Derivation of the Hammett Equation
The relationship between the two reactions is given by:
log k = " logK + C
Considering the unsubstituted carboxylic acid as the base case reaction, we can subtract its value from both sides of the equation.
log k " log k H = # (logK " logK H )
These are simply the pKa values of the substituted and unsubstituted benzoic acids.
" k % log$ ' = ( ) pK a ( H ) * pK a = ( ) + # kH &
The term (pKa(H) - pKa) is given the symbol !m or !p for meta and para-substituted benzoic ! acids and is known as the substitution constant. This can be calculated for any substituted benzoic acid for which we can find (or measure) pKa.
Autumn 2004
Derivation of the Hammett Equation
Hammett found that many other reactions also showed straight-line correlations of their rate or equilibrium behavior for a series of substituents with the equilibrium behavior of benzoic acid (manifested as !).
or " K % log$ ' = ()* #KH &
NH2 + X O! O Cl k O N H X
" k % log$ ' = ( ) * # kH &
a
!
logk/ko or log K/Ko
benzene 25 C
a b
"= -2.69
"= 2.51
OEt k EtOH 25 C X X CH2COO ! K X H2O 25 C X COO! OH! k EtOH/H2O 25 C X
b c
EtI
CH2COOH
c d
0.47 -0.99
COOEt
d
X
-1
-0.8 -0.6 -0.4 -0.2
0 sigma
0.2
0.4
0.6
0.8
Autumn 2004
The Hammett Equation
The equation describing the straight line correlation between a series of reactions with substituted aromatics and the hydrolysis of benzoic acids with the same substituents is known as the Hammett Equation.
" k % log$ ' = ( ) * # kH &
or
" K % log$ ' = ()* #KH &
! = substituent constant
!Log of the ratio of
either the reaction rate constant or the equilibrium constant
" = reaction constant Proportionality constant between log of k (or K) values and !
A measure of the total polar effect exerted by substituent X (relative to no substituent) on the reaction centre. recall: ! = -(pKa # pKa(H)) #ve = electron-donating +ve = electron-withdrawing
7
Autumn 2004
Physical Meaning of ! and "
The substituent constant ! is a measure of the total polar effect exerted by substituent X (relative to no substituent) on the reaction centre.
Electron-withdrawing m-NO2 (! = +0.71) increases stability of tetrahedral intermediate compared to electrondonating m-CH3 (! = -0.07).
Methoxy substituent can be electron-withdrawing due to inductive effects (meta, ! = +0.12) , or electron-donating (para, ! = -0.27) due to mesomeric effects.
8
Autumn 2004
Physical Meaning of ! and "
The reaction constant " is the slope of the line correlating log k or log K with the sigma values of the substituents. The sign of the slope tells whether a reaction rate is accelerated or suppressed by electron-donating vs. electron withdrawing substituents. Negative " is diagnostic of the development of positive charge at the reaction centre in the transition state of the rate-limiting step. rate will be suppressed by electron-withdrawing substituents. Positive " is diagnostic of the development of negative charge at the reaction centre in the transition state of the rate-limiting step. rate will be accelerated by electron-withdrawing substituents. The magnitude of " is a measure of how susceptible a reaction is to the electronic characteristics of the substituent.
Autumn 2004
Significance of "
Lets consider again two of the reaction examples we looked at previously:
NH2 O + X Cl k O N H X
benzene 25 C
a
logk/ko or log K/Ko
"= -2.69
"= 2.51
b
COOEt COO! OH! k EtOH/H2O 25 C X
d
d
+ X
-1
-0.8 -0.6 -0.4 -0.2
0 sigma
0.2
0.4
0.6
0.8
electrondonating
electronwithdrawing
10
Autumn 2004
A Closer Look at Substituent Constants
In many cases, we find that strongly electron-withdrawing or strongly electron-donating substituents dont fall on the line predicted by the Hammett correlation. Example: p-CN and p-NO2 are above the line; this suggests that compounds with these substituents act as stronger acids than we would have predicted from their ! values.
OH + H2 O X
O!
+ H3 O+ X
When electron-withdrawing due to to mesomeric effects can be extended to the reaction centre via through conjugation, the result is an even more stabilized species.
11
Autumn 2004
Modified Substituent Constants
!#
Scale
We can develop new ! values for these substrates by separating out these through-conjugation effects from inductive effects. Develop line with " value based on m-substituents only, which cannot exhibit mesomeric effects. The amount by which certain substituents deviate from the line can be added to their ! values to produce a new scale of !# value.
substituent CO2Et COMe CN CHO NO2
!p 0.45 0.50 0.66 0.43 0.78
!p# 0.68 0.84 0.88 1.03 1.27 12
Autumn 2004
Modified Substituent Constants !+ Scale
Similarly, in some cases,we find that strongly electron-donating substituents dont fall on the line predicted by the Hammett correlation. Example: SN1 solvolysis of p-substituted tertiary halides
p-OCH3 and p-CH3 are above the line; through-conjugation enhances their electron-donating ability.
!+ scale:
substituent C6H5 Me MeO NH2 NMe2 !p -0.01 -0.17 -0.27 -0.66 -0.83 !p+ -0.18 -0.31 -0.78 -1.30 -1.70
13
Autumn 2004
Uses of Hammett Plots
How do we make use of Hammett plots? Calculation of k or K for a specific reaction of a specific compound: If we know " for a particular reaction, then we k log x = " # $ x can calculate the rate (or equilibrium) constant kH for any substituent relative to that for the unsubstituted compound (because we also know ! for the substituent). ! To provide information about reaction pathways: Magnitude and sign of " tell about development of charge at reaction centre. If !+ or !- gives a better correlation than !, then we know we have a reaction where through conjugation is important. Deviations from linearity: arguably, the most mechanistically informative Hammett plots are ones that dont give straight lines!
14
Autumn 2004
Deviations from Linearity in Hammett Plots
Concave Upwards deviation: Compare the Hammett plots for the hydrolysis of ArCO2R (R= Me and Et) carried out in 99.9% H2SO4. Me esters show well-behaved plot with " = -3.25 Et esters show well-behaved plot with " = -3.25 switching to " = + 2.0 Mechanism for Me esters:
CH3 OH O Ar
+ +
H O
OH2
O OMe Ar
slow
O
+
O Ar OH H+ Ar
OMe H+
Ar H2 O
OH
OH2
H2 O
Positive charge develops at reaction centre during rate-limiting step
15
Autumn 2004
Deviations from Linearity in Hammett Plots
Concave Upwards deviation: What happens for Et esters? Change in mechanism: positive charge near reaction centre is decreased in rate-limiting step, leading to a positive " value. Mechanism changes for Et esters but not Me esters because a stable carbocation +CH2Me can be formed in Et ester case. Mechanism for Et esters:
O Ar
+
O OCH2 Me Ar O H+
slow
O Ar OH
CH2 Me
CH2 Me
OH2
For electron-withdrawing substituents: Positive charge at reaction centre is decreased during rate-limiting step.
16
Autumn 2004
Deviations from Linearity in Hammett Plots
Concave Upwards deviation: Concave upwards deviation can usually be taken as evidence of a change in reaction mechanism. Any new pathway coming into play must be faster than the original pathway, or the original pathway would continue to dominate. A faster pathway gives an upward curving deviation.
100 90 80 70 60
new, faster pathway
ln k
50 40 30 20 10 0 0 0.2 0.4 0.6 0.8 1
sigma
17
Autumn 2004
Deviations from Linearity in Hammett Plots
Concave Downwards deviation: Concave downwards deviation can also be observed, as in this example of the cyclodehydration of substituted 2-phenyltriarylmethanol compounds:
Positive " for electrondonating X,Z substituents Negative " for electronwithdrawing X,Z substituents
18
Autumn 2004
Deviations from Linearity in Hammett Plots
Concave Downwards deviation: Reaction mechanism: E1 elimination of H2O to form a carbocation followed by an internal electrophilic substitution. Which step is rate-limiting, a) or b) ?
a)
+
! H2 O C Ar OH2 Ar C+ Ar
Ar
b)
C+ Ar Ar Ar
C+ Ar
+
H
19
Autumn 2004
Deviations from Linearity in Hammett Plots
Concave Downwards deviation: Which step is rate-limiting? In a), positive charge at the reaction centre is increasing (" = negative). This suggests that a) is rate-limiting for the right of the Hammett pot. In b), positive charge at the reaction centre is decreasing (" = positive). This suggests that b) is rate-limiting for the left of the Hammett plot.
b) r.l.s. a) r.l.s.
a)
+
! H2 O C Ar OH2 Ar C+ Ar
Ar
b)
C+ Ar Ar Ar
C+ Ar
+
H
20
Autumn 2004
Deviations from Linearity in Hammett Plots
Concave Upwards deviation: indicates change in reaction mechanism.
Concave Downards deviation: indicates same mechanism, change in rate-limiting step.
21
Autumn 2004
Thermodynamic Implications of Hammett Plots
We have mentioned several times that linear free energy relationships make a correlation between thermodynamic ($G) and transition state ($G) properties of the reaction, which is grounded on an empirical and not a theoretical basis.
Rate constant:
$ k ' "2.303RT # log& ) = (*H m " *H xm ) " T (*S m " *Sxm ) % kH (
RCOOEt + OH " #k $ RCOO" + EtOH #
log(k/kH)
Equilibrium constant:
! $ K ' o o o o "2.303RT # log& ) = (*H " *H x ) " T (*S " *Sx ) %KH (
RCOO" + H 2O
log(K/KH)
RCOOH + OH " #K $ # %# #
!
!
22
Autumn 2004
Thermodynamic Implications of Hammett Plots
Why do these relationships work? The implicit meaning of a linear Hammett plot is that one or more of the following three conditions is satisfied in each series of reactions: . $H is linearly related to $S for the series . $H is constant for the series . $S is constant for the series
$ k ' m m "2.303RT # log& ) = (*H m " *H H ) " T (*S m " *S H ) kH ( % $ K ' o o o o "2.303RT # log& ) = (*H " *H H ) " T (*S " *S H ) KH ( %
23
Rate constant:
Equilibrium constant: !
You might also like
- Protecting Group HandoutDocument5 pagesProtecting Group HandoutRafaelle Sanvictores SilongNo ratings yet
- Vehicle Suspension DesignDocument63 pagesVehicle Suspension DesignVenkatesh ChegireddyNo ratings yet
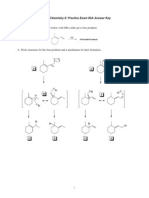
- Organic Chemistry II Practice Exam #3A Answer KeyDocument8 pagesOrganic Chemistry II Practice Exam #3A Answer Keyhiep237No ratings yet
- Organic Chemistry From Retrosynthesis To Asymmetric Synthesis (PDFDrive)Document221 pagesOrganic Chemistry From Retrosynthesis To Asymmetric Synthesis (PDFDrive)I CbergNo ratings yet
- Name Reactions-II PDFDocument32 pagesName Reactions-II PDFEstanislao Amadeo Avogadro100% (1)
- LT Loads&SwitchgearDocument48 pagesLT Loads&SwitchgearsreekanthbammidiNo ratings yet
- Tamil Nadu Leave RulesDocument26 pagesTamil Nadu Leave RulesDr.Sagindar85% (27)
- Organic Chemistry - Top 500 Question Bank For JEE Main by MathonGo PDFDocument65 pagesOrganic Chemistry - Top 500 Question Bank For JEE Main by MathonGo PDFDeepak BinwalNo ratings yet
- 01.coordination Chemistry Class Notes Part I-1 PDFDocument86 pages01.coordination Chemistry Class Notes Part I-1 PDFShadrack Peter100% (1)
- 4.0L EngineDocument347 pages4.0L EngineCapssa Oscar100% (2)
- MSCCH07Document385 pagesMSCCH07capdesuro100% (1)
- E1 and E2 ReactionsDocument30 pagesE1 and E2 ReactionsVidhu Pandey100% (1)
- Engineering Chemistry by SS Dara PDFDocument272 pagesEngineering Chemistry by SS Dara PDFAnkit57% (14)
- Synthetic ReagentsDocument75 pagesSynthetic ReagentsBapu Thorat100% (1)
- Ball Valve FCTDocument16 pagesBall Valve FCTAnggi SujiwoNo ratings yet
- Freno Motor ISMDocument6 pagesFreno Motor ISMFabiiam Kiimtero100% (2)
- 10 - Introduction To PROFIBUS DP and The HMI Operator PanelDocument31 pages10 - Introduction To PROFIBUS DP and The HMI Operator PanelMichele BacocchiaNo ratings yet
- Asymmetric SynthesisDocument55 pagesAsymmetric Synthesisevsgoud_goud0% (1)
- Biggest Chemistry Collection: Csir Ugc Net JRF Chemistry M.SC Chemistry Entrance IIT-JAM Chemistry NEET ChemistryDocument823 pagesBiggest Chemistry Collection: Csir Ugc Net JRF Chemistry M.SC Chemistry Entrance IIT-JAM Chemistry NEET Chemistrysahil popli100% (1)
- Resonance and Inductive Effects PresentationDocument36 pagesResonance and Inductive Effects Presentationeagl33yeNo ratings yet
- Organic Chemistry Midterm 1 Dir+eff++keyDocument1 pageOrganic Chemistry Midterm 1 Dir+eff++keyNorma Leticia RamosNo ratings yet
- VSL Technical Report - PT ExternalDocument36 pagesVSL Technical Report - PT ExternalTran Nguyen KhiemNo ratings yet
- FMO LectureDocument14 pagesFMO Lecturebooks4free23No ratings yet
- An Introduction to Co-Ordination Chemistry: International Series of Monographs in Inorganic ChemistryFrom EverandAn Introduction to Co-Ordination Chemistry: International Series of Monographs in Inorganic ChemistryRating: 3.5 out of 5 stars3.5/5 (2)
- Settling Tank DecantDocument23 pagesSettling Tank Decantjvan migvelNo ratings yet
- Aromaticity NotesDocument10 pagesAromaticity NotesVirendra Singh Rajput100% (1)
- Reaction MechanismDocument68 pagesReaction MechanismSiddarth Singh73% (11)
- Coordination TheoryDocument38 pagesCoordination TheorySLEVINPJOY100% (11)
- Absorption Spectra and Chemical Bonding in ComplexesFrom EverandAbsorption Spectra and Chemical Bonding in ComplexesRating: 2.5 out of 5 stars2.5/5 (2)
- Leah4Sci OrgChem Cheat Sheet PDFDocument32 pagesLeah4Sci OrgChem Cheat Sheet PDFhNo ratings yet
- Org Chem Sem 3 Paper 2Document15 pagesOrg Chem Sem 3 Paper 2Rohit DeshmukhNo ratings yet
- AromaticityDocument12 pagesAromaticityV G Viju KumarNo ratings yet
- PMR Spectroscopy: Solved Problems Volume : IIFrom EverandPMR Spectroscopy: Solved Problems Volume : IIRating: 5 out of 5 stars5/5 (3)
- Retro Workbook For Organic SynthesisDocument15 pagesRetro Workbook For Organic SynthesisUmmaraRabbani100% (1)
- Unit 10 Thermogravimetric AnalysisDocument24 pagesUnit 10 Thermogravimetric Analysismaidhily83% (6)
- Problems On Named ReactionsDocument103 pagesProblems On Named ReactionsBapu ThoratNo ratings yet
- Csir Model QuestionsDocument15 pagesCsir Model QuestionsbaluNo ratings yet
- Introduction of Organic Chemistry by Eyes of Ajnish Kumar Gupta (AKG)Document24 pagesIntroduction of Organic Chemistry by Eyes of Ajnish Kumar Gupta (AKG)ajju_208180% (5)
- Hammett Parameters: Eric Amerling & Christine Nervig University of UtahDocument8 pagesHammett Parameters: Eric Amerling & Christine Nervig University of UtahThanh LêNo ratings yet
- SET-NET Pericyclic ReactionsDocument61 pagesSET-NET Pericyclic ReactionsBapu ThoratNo ratings yet
- Organic Chemistry 2021Document76 pagesOrganic Chemistry 2021Arah Mae BonillaNo ratings yet
- Bio Inorganic 1 PPT ChemistryDocument57 pagesBio Inorganic 1 PPT ChemistryShantanu MawaskarNo ratings yet
- Hetero-Cyclic CompoundsDocument69 pagesHetero-Cyclic CompoundsNaveed SajidNo ratings yet
- Aromaticity CompleteDocument104 pagesAromaticity Completewahidalwahdi100% (1)
- OrganicChemistryChapter5 PDFDocument19 pagesOrganicChemistryChapter5 PDFJuliet Tatiana CumbeNo ratings yet
- MOT Inorganic ChemistryDocument3 pagesMOT Inorganic ChemistryLohith Loli100% (1)
- Exams Organic Chemistry MITDocument333 pagesExams Organic Chemistry MITn2h_spNo ratings yet
- Grignard Reagents Review MeetingDocument28 pagesGrignard Reagents Review MeetingShivali SharmaNo ratings yet
- Module8 PDFDocument40 pagesModule8 PDFFaizan AhmadNo ratings yet
- CH2203 - Spectroscopy of Inorganic CompoundsDocument6 pagesCH2203 - Spectroscopy of Inorganic CompoundsJohnNo ratings yet
- 01 StereochemistryDocument6 pages01 StereochemistryGundum Bodyz100% (1)
- Sharpless Asymmetric EpoxidationDocument19 pagesSharpless Asymmetric Epoxidationjaanabhenchod100% (2)
- RetrosynthesisDocument20 pagesRetrosynthesistamilsam19860% (1)
- Carbocation - A General ViewDocument6 pagesCarbocation - A General ViewUsama AsifNo ratings yet
- Organic Chemistry IIDocument7 pagesOrganic Chemistry IIRoberto SIlvaNo ratings yet
- Pericyclics-2014 Handout PDFDocument79 pagesPericyclics-2014 Handout PDFnavchemNo ratings yet
- Book Solution FoyesDocument15 pagesBook Solution Foyesnorma ireneNo ratings yet
- Foundations of Organic ChemistryDocument4 pagesFoundations of Organic Chemistryeager18No ratings yet
- 15321581482018-19 M.sc. Organic Chemistry Part-IIDocument17 pages15321581482018-19 M.sc. Organic Chemistry Part-IIArnab ChakrabortyNo ratings yet
- Ugi ReactionsDocument36 pagesUgi ReactionsHarjinder Singh Bhatia100% (1)
- Chapter 3 Coordination ChemistryDocument41 pagesChapter 3 Coordination Chemistrytarun ratnaNo ratings yet
- Csir Ugc JRF Net Chemistry Paper 1 (Part B) Series - 1Document22 pagesCsir Ugc JRF Net Chemistry Paper 1 (Part B) Series - 1polamrajuNo ratings yet
- Aromaticity 2019Document65 pagesAromaticity 2019Shreya PrakashNo ratings yet
- Isolobal AnalogyDocument4 pagesIsolobal Analogyindu priyaNo ratings yet
- Transition MetalDocument45 pagesTransition MetalNurul NadiaNo ratings yet
- Molecular RearrangementsDocument9 pagesMolecular RearrangementsDhanaswamy Ilangeswaran67% (3)
- Notes On Coordination CompoundsDocument12 pagesNotes On Coordination CompoundsRojo JohnNo ratings yet
- A Concise Text-Book of Organic Chemistry: The Commonwealth and International Library: Chemistry DivisionFrom EverandA Concise Text-Book of Organic Chemistry: The Commonwealth and International Library: Chemistry DivisionRating: 5 out of 5 stars5/5 (2)
- 12 Chemistry ChapterTests Chapter 7 Level 1 Test 1Document1 page12 Chemistry ChapterTests Chapter 7 Level 1 Test 1Deepan KumarNo ratings yet
- SET15-Chemical Sciences - P III-A West Bengal 2015Document16 pagesSET15-Chemical Sciences - P III-A West Bengal 2015Deepan KumarNo ratings yet
- Ce20100200009 36898265Document2 pagesCe20100200009 36898265dhavaleshNo ratings yet
- L Scheme Iyr Syll BookDocument202 pagesL Scheme Iyr Syll BookDeepan KumarNo ratings yet
- Aromaticity: 1 Hückel Rule (Review)Document5 pagesAromaticity: 1 Hückel Rule (Review)Deepan KumarNo ratings yet
- 2013 SP 12 Chemistry 02Document6 pages2013 SP 12 Chemistry 02dhriti1996singhNo ratings yet
- 2014 SP 12 Chemistry 06Document6 pages2014 SP 12 Chemistry 06Deepan KumarNo ratings yet
- Chemistry HotsDocument48 pagesChemistry HotsleesaaanNo ratings yet
- Organic SpectrosDocument7 pagesOrganic SpectrosDeepan KumarNo ratings yet
- Short Course On Heterocyclic Chemistry: and Reactions of Heteroaromatic Rings With ElectrophilesDocument40 pagesShort Course On Heterocyclic Chemistry: and Reactions of Heteroaromatic Rings With ElectrophilesSaman FatimaNo ratings yet
- Pro.753687 en EU R4Document14 pagesPro.753687 en EU R4Rakshit SharmaNo ratings yet
- Z4 Multi-Function Button Retrofit KitDocument12 pagesZ4 Multi-Function Button Retrofit KitgioioioiNo ratings yet
- Epoxylite TSA 220 - TDS - 2008Document3 pagesEpoxylite TSA 220 - TDS - 2008Anonymous sAmJfcVNo ratings yet
- ADPDocument165 pagesADPmohhizbarNo ratings yet
- Design CriteriaDocument6 pagesDesign CriteriaAllyssa Jane ErcillaNo ratings yet
- WAHLER Imagebroschuere SERIE enDocument16 pagesWAHLER Imagebroschuere SERIE ensagarpatel337No ratings yet
- Open IVDocument84 pagesOpen IVSokr AbdoNo ratings yet
- NE5521Document9 pagesNE5521Carlos TibussiNo ratings yet
- ITW Rust Inhibitive Primer 7CZ Product Data 1469103Document2 pagesITW Rust Inhibitive Primer 7CZ Product Data 1469103rihtak123No ratings yet
- 1964 US Army Vietnam War Military Police Traffic Control 203pDocument202 pages1964 US Army Vietnam War Military Police Traffic Control 203pwwwsurvivalebookscom100% (1)
- AMZ PresenceDocument6 pagesAMZ Presencemarginwalker77No ratings yet
- Materials Science and Engineering.: Session 9 - The Eutectic Phase DiagramsDocument34 pagesMaterials Science and Engineering.: Session 9 - The Eutectic Phase DiagramsfiestapaganaNo ratings yet
- 01-09-04MIL On, DTCs P1545, P0121, P0123, P0221 or P0222 Stored in ECM Fault MemoryDocument7 pages01-09-04MIL On, DTCs P1545, P0121, P0123, P0221 or P0222 Stored in ECM Fault MemoryKilobyte202005No ratings yet
- 6 Energy Calculation: UDEC Version 4.0Document28 pages6 Energy Calculation: UDEC Version 4.0Rakshit MishraNo ratings yet
- Parts Catalog: MF9280Cdn/9220Cdn/9170/ 9150/9130/8450 SeriesDocument94 pagesParts Catalog: MF9280Cdn/9220Cdn/9170/ 9150/9130/8450 SeriesAdorjan Sandor ZoltanNo ratings yet
- Lincoln PowerMaster III 2011Document10 pagesLincoln PowerMaster III 2011Manuel Aguirre PiconNo ratings yet
- A Biometric Model For Examination Screening and Attendance Monitoring in Yaba College of TechnologyDocument6 pagesA Biometric Model For Examination Screening and Attendance Monitoring in Yaba College of TechnologyWorld of Computer Science and Information Technology JournalNo ratings yet
- Group 14 : Strainers, Traps, Fine-Mesh Filters Filling Device For Methanol Drain DeviceDocument3 pagesGroup 14 : Strainers, Traps, Fine-Mesh Filters Filling Device For Methanol Drain DeviceLPG Equipment Consulting and ServicesNo ratings yet
- General Description: Dual Retriggerable Precision Monostable MultivibratorDocument17 pagesGeneral Description: Dual Retriggerable Precision Monostable Multivibratorsajad hejaziNo ratings yet
- Marking and Ordering Code System (Capacitors)Document6 pagesMarking and Ordering Code System (Capacitors)José TurinNo ratings yet
- A Time To Build: People's Housing in Asia: Photograph By: Yann Arthus-BertrandDocument14 pagesA Time To Build: People's Housing in Asia: Photograph By: Yann Arthus-BertrandarchitectsforumNo ratings yet
- Banana ChipsDocument5 pagesBanana Chipsbikram limbuNo ratings yet