Professional Documents
Culture Documents
Lec09 03072007 W
Uploaded by
José Carlos RessoniOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Lec09 03072007 W
Uploaded by
José Carlos RessoniCopyright:
Available Formats
10.37 Chemical and Biological Reaction Engineering, Spring 2007 Prof. K.
Dane Wittrup Lecture 9: Reactor Size Comparisons for PFR and CSTR This lecture covers reactors in series and in parallel, and how the choice of reactor affects selectivity versus conversion.
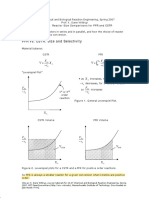
PFR vs. CSTR: Size and Selectivity
Material balance: CSTR PFR
V=
Levenspiel Plot
FAo XA rA
V=
XA
FAo dX A rA
as X A increases, C A decreases
FAo rA
1st or 2nd order reaction
rA decreases, for 1st and 2nd order, F so Ao increases rA
Figure 1. General Levenspiel Plot.
XA
CSTR Volume PFR Volume
FAo rA
FAo rA
VCSTR
VPFR
XA
XA
Figure 2. Levenspiel plots for a CSTR and a PFR for positive order reactions. So PFR is always a smaller reactor for a given conversion when kinetics are positive order.
Cite as: K. Dane Wittrup, course materials for 10.37 Chemical and Biological Reaction Engineering, Spring 2007. MIT OpenCourseWare (http://ocw.mit.edu), Massachusetts Institute of Technology. Downloaded on [DD Month YYYY].
Non-monotonically positive order kinetics arise: Autocatalytic reactions (e.g. cell growth) Adiabatic or non-isothermal exothermic reactions Product inhibited reactions (some enzymes)
Series of Reactors
Example: 2 CSTRs FAo
v1 x1
FA1
v2
FA2
V1 = FAo X1 rA1
x2
Figure 3. Schematic of two CSTRs in series.
0 2nd reactor: In + Out + Prod = Acc
FA1 FA2 + rA2 V2 = Steady state
10.37 Chemical and Biological Reaction Engineering, Spring 2007 Prof. K. Dane Wittrup
Lecture 9 Page 2 of 6
Cite as: K. Dane Wittrup, course materials for 10.37 Chemical and Biological Reaction Engineering, Spring 2007. MIT OpenCourseWare (http://ocw.mit.edu), Massachusetts Institute of Technology. Downloaded on [DD Month YYYY].
FA2 = FA0 X 2 FA0
V2 =
FA0 rA2
( X 2 X1 )
FAo rA
V1
V2
Figure 4. Reactor volumes for 2 CSTRs in series.
X1
X2
Multiple CSTRs begin to approximate a single PFR
FAo rA
X
Figure 5. Reactor volumes for multiple CSTRs in series.
10.37 Chemical and Biological Reaction Engineering, Spring 2007 Prof. K. Dane Wittrup
Lecture 9 Page 3 of 6
Cite as: K. Dane Wittrup, course materials for 10.37 Chemical and Biological Reaction Engineering, Spring 2007. MIT OpenCourseWare (http://ocw.mit.edu), Massachusetts Institute of Technology. Downloaded on [DD Month YYYY].
FAo rA
VPFR VCSTR
FAo rA
VCSTR VPFR
X
Designed final conversion
XA
Final X
Figure 6. Levenspiel plots comparing CSTR and PFR volumes for changing kinetics. Left: The CSTR has the smaller volume. Right: The PFR eventually has the smaller volume. Choice of PFR vs CSTR depends on conversion. Choose the reactor that has the smallest volume reduce cost. Reactors: CSTR
FAo rA
VCSTR VPFR
PFR
X
Final X Figure 7. To achieve the desired conversion with smaller reactor volumes, use a combination. In this case, use a CSTR then a PFR. By doing so, the reactor volume is less than the area underneath the curve. For competing parallel reactions, selectivity for desired product can dominate the choice. Example
A D A U
1 rD = kd C A
D = Desired, U = Undesired
rU = ku C A
10.37 Chemical and Biological Reaction Engineering, Spring 2007 Prof. K. Dane Wittrup
Lecture 9 Page 4 of 6
Cite as: K. Dane Wittrup, course materials for 10.37 Chemical and Biological Reaction Engineering, Spring 2007. MIT OpenCourseWare (http://ocw.mit.edu), Massachusetts Institute of Technology. Downloaded on [DD Month YYYY].
Define selectivity
SD /U =
rD kd (1 2 ) = CA rU ku
If
1 > 2 , as C A
increases, S D / U increases
-Favors PFR because C A starts at C Ao then drops whereas CSTR concentrations are always at lower C A . If
1 < 2 , as C A 1 = 2
then
increases, S D / U decreases
-CSTR favored If
S D /U =
kd , no dependence on C A ku
-Therefore no CSTR/PFR preference. Define a fractional yield
1 kd C dCD A = 2 1 dC A kd C A + ku C A
Overall fractional yield
All D produced All A consumed
For a CSTR:
= xit C
0
C A = C A C A
For a PFR:
1 C A
C At
CA 0
dC A
If
1 = 2
C A
C Af
C A0
CA
Figure 8. Fractional yield versus concentration. Selectivity does not depend on CA.
10.37 Chemical and Biological Reaction Engineering, Spring 2007 Prof. K. Dane Wittrup
Lecture 9 Page 5 of 6
Cite as: K. Dane Wittrup, course materials for 10.37 Chemical and Biological Reaction Engineering, Spring 2007. MIT OpenCourseWare (http://ocw.mit.edu), Massachusetts Institute of Technology. Downloaded on [DD Month YYYY].
If
1 > 2
CA
Figure 9. Fractional yield versus concentration when 1 > 2. CSTR PFR
CSTR C A
C Af
PFR C A
C Af
C A0 CA
C A0 CA
Figure 10. Comparison of overall fractional yield for a CSTR and a PFR when 1 > 2. PFR is preferred because PFR > CSTR , therefore the yield of D per mol A consumed is higher. If
1 < 2
CSTR C A
PFR C A
C Af
C A0
CA
C Af
C A0 C A
CSTR PFR Figure 11. Comparsion of overall fractional yield for a CSTR and a PFR when 1 > 2.
PFR < CSTR
10.37 Chemical and Biological Reaction Engineering, Spring 2007 Prof. K. Dane Wittrup Lecture 9 Page 6 of 6
Cite as: K. Dane Wittrup, course materials for 10.37 Chemical and Biological Reaction Engineering, Spring 2007. MIT OpenCourseWare (http://ocw.mit.edu), Massachusetts Institute of Technology. Downloaded on [DD Month YYYY].
You might also like
- Ancient Skies of Northern Europe: Stars, Constellations, and The Moon in Nordic MythologyDocument12 pagesAncient Skies of Northern Europe: Stars, Constellations, and The Moon in Nordic MythologyoldenglishblogNo ratings yet
- Littelfuse EVI EV Charging Infrastructure Presentation PDFDocument19 pagesLittelfuse EVI EV Charging Infrastructure Presentation PDFRaka Pratindy100% (1)
- Hall Effect Sensor Placement For Permanent Magnet Brushless DC MotorsDocument4 pagesHall Effect Sensor Placement For Permanent Magnet Brushless DC Motorsmarco.dado2700No ratings yet
- Batching Plant ReportDocument89 pagesBatching Plant ReportShahzad Tabassum0% (3)
- LATOUR, Bruno - On InterobjectivityDocument19 pagesLATOUR, Bruno - On InterobjectivityjoaofmaNo ratings yet
- Newton Papers Letter Nat Phil Cohen EdDocument512 pagesNewton Papers Letter Nat Phil Cohen EdFernando ProtoNo ratings yet
- CardiomyopathyDocument1 pageCardiomyopathyTrisha VergaraNo ratings yet
- Thermodynamics - by WWW - LearnEngineering.inDocument38 pagesThermodynamics - by WWW - LearnEngineering.inTamal KumbhakarNo ratings yet
- CHFEN 3553 Chemical Reaction Engineering: April 28, 2003 1:00 PM - 3:00 PM Answer All QuestionsDocument4 pagesCHFEN 3553 Chemical Reaction Engineering: April 28, 2003 1:00 PM - 3:00 PM Answer All QuestionsJiahui TanNo ratings yet
- Prob 01Document12 pagesProb 01Ahmad SayyedahmadNo ratings yet
- L1-5 Energy BalancesDocument40 pagesL1-5 Energy BalancesHemavathy Rt100% (1)
- CHE1401 LabManual JULY2015Document76 pagesCHE1401 LabManual JULY2015Adrian Prashantha WeerakkodyNo ratings yet
- Economic Cost Analysis of Hybrid Renewable Energy System Using HOMERDocument6 pagesEconomic Cost Analysis of Hybrid Renewable Energy System Using HOMERAndiHilmySyahirNo ratings yet
- Separation Processes HandoutDocument10 pagesSeparation Processes Handoutkiran367No ratings yet
- Lab 05 - Extended SurfacesDocument12 pagesLab 05 - Extended SurfacesMuhammad FarhanNo ratings yet
- Internal Forced ConvectionDocument18 pagesInternal Forced ConvectionMohd Jamal Mohd MoktarNo ratings yet
- Environmental Essay On Nuclear Waste DisposalDocument10 pagesEnvironmental Essay On Nuclear Waste Disposalkay-No ratings yet
- GRanD Technical DocumentationDocument12 pagesGRanD Technical DocumentationHari Ram UpadhayayNo ratings yet
- MIT6 041F10 FinalDocument20 pagesMIT6 041F10 FinalGokce TuncerNo ratings yet
- PR 1-5Document18 pagesPR 1-5Febryan CaesarNo ratings yet
- Chapter 2 - Data InterpretationDocument24 pagesChapter 2 - Data InterpretationPHƯƠNG ĐẶNG YẾNNo ratings yet
- Physics Exam QuestionsDocument8 pagesPhysics Exam Questionspetesqueeler7052No ratings yet
- Mass Transfer Lab ManualDocument29 pagesMass Transfer Lab ManualPrajwal ChavanNo ratings yet
- Worksheet For Class 10Document9 pagesWorksheet For Class 10rishit allamNo ratings yet
- Topic 2 - Neutron Chain Fission ReactorDocument45 pagesTopic 2 - Neutron Chain Fission ReactorSit LucasNo ratings yet
- Opa1 Impacto MuscularDocument23 pagesOpa1 Impacto MuscularRafael GermanoNo ratings yet
- UO 4 Solid Handling UnitDocument17 pagesUO 4 Solid Handling UnitNoor FadzleenaNo ratings yet
- Advance Fluid Lab ManualDocument55 pagesAdvance Fluid Lab ManualchristianNo ratings yet
- Fluid Mechanics: (2nd Edition)Document6 pagesFluid Mechanics: (2nd Edition)Ehtisham ZiaNo ratings yet
- Kneader Mixer Lab Report by Group E (SECTION A)Document16 pagesKneader Mixer Lab Report by Group E (SECTION A)Maryam Fatima100% (1)
- P4E2: Kinetics of Homogeneous Reaction in Batch and Continuous Stirred-Tank Reactor at Two Different TemperatureDocument7 pagesP4E2: Kinetics of Homogeneous Reaction in Batch and Continuous Stirred-Tank Reactor at Two Different TemperaturejayaprinaNo ratings yet
- Chemical Engineering Mass Transfer NotesDocument36 pagesChemical Engineering Mass Transfer NotesLebohang Czar Nku50% (2)
- Units and DimensionDocument27 pagesUnits and DimensionIfiok UsoroNo ratings yet
- First Order SystemDocument21 pagesFirst Order SystemNiranjan BeheraNo ratings yet
- 7405Document8 pages7405Ebby OnyekweNo ratings yet
- Chapter 1 IntroductionDocument9 pagesChapter 1 IntroductionNikko ManaleseNo ratings yet
- Dew Point - Thermo 2 Lab Report B1 (DEW POINT)Document5 pagesDew Point - Thermo 2 Lab Report B1 (DEW POINT)Taqqi HaiderNo ratings yet
- MASS TRANSFER Diffusion LectureDocument11 pagesMASS TRANSFER Diffusion LectureRashmi Walvekar SiddiquiNo ratings yet
- 333 Manual 11 T2 Armfield ManualDocument45 pages333 Manual 11 T2 Armfield ManualSyamim AsyrafNo ratings yet
- CP302 MassTransfer 02 OKDocument38 pagesCP302 MassTransfer 02 OKDeneshVijayNo ratings yet
- PT316 - Topic 1 - Single Particles in Fluids PDFDocument30 pagesPT316 - Topic 1 - Single Particles in Fluids PDFChemEngGirl89No ratings yet
- Homework 1Document9 pagesHomework 1AgithaNo ratings yet
- Gas UnitDocument4 pagesGas UnitZalina SamsuddinNo ratings yet
- Single Slit Diffraction ExpDocument5 pagesSingle Slit Diffraction ExpNaresh KumarNo ratings yet
- School of Civil, Environmental & Chemical EngineeringDocument38 pagesSchool of Civil, Environmental & Chemical EngineeringJair BarruetaNo ratings yet
- Unit 4 Chemical KineticsDocument8 pagesUnit 4 Chemical KineticsRahgul M.S.No ratings yet
- I. Principles of Wind EnergyDocument3 pagesI. Principles of Wind EnergyJohn TauloNo ratings yet
- Experiment 7Document7 pagesExperiment 7Ariadna AgotoNo ratings yet
- Center of Pressure and Hydrostatic Force On A Submerged Body-RevDocument7 pagesCenter of Pressure and Hydrostatic Force On A Submerged Body-RevAnkit MittalNo ratings yet
- Lab Report 5 DistillationDocument6 pagesLab Report 5 DistillationMyeeka Hammond100% (1)
- 24 Ijecsmay PDFDocument11 pages24 Ijecsmay PDFHarshaNo ratings yet
- B 4 Stress ManagementDocument10 pagesB 4 Stress ManagementSourabh GargNo ratings yet
- Collection and Analysis of Rate DataDocument24 pagesCollection and Analysis of Rate DataAfs IkhlasNo ratings yet
- Chemical ThermodynamicsDocument33 pagesChemical ThermodynamicsAkash Ghosh0% (1)
- Isothermal Reactor Design: 1. Batch OperationDocument3 pagesIsothermal Reactor Design: 1. Batch Operationنزار الدهاميNo ratings yet
- Why Democracies Ban Political PartiesDocument31 pagesWhy Democracies Ban Political PartiesVasile CioriciNo ratings yet
- PSHE - Lesson Plan 1Document1 pagePSHE - Lesson Plan 1Sowole JideNo ratings yet
- ECE 3040 Lecture 22: Numerical Solution of Differential EquationsDocument34 pagesECE 3040 Lecture 22: Numerical Solution of Differential EquationsAqe KitamNo ratings yet
- UNIT II: Fuels: SyllabusDocument26 pagesUNIT II: Fuels: SyllabusMAYUR BHOSALE100% (1)
- Massachusetts Institute of Technology v. VizioDocument17 pagesMassachusetts Institute of Technology v. VizioPriorSmartNo ratings yet
- Catalyst Characterization TechniquesDocument8 pagesCatalyst Characterization TechniquesDaniel DadzieNo ratings yet
- Radiation Safety: ἄτομος or átomos meaning "indivisible") is the smallest particle of aDocument10 pagesRadiation Safety: ἄτομος or átomos meaning "indivisible") is the smallest particle of adypietNo ratings yet
- Experimental Thermodynamics: Experimental Thermodynamics of Non-Reacting FluidsFrom EverandExperimental Thermodynamics: Experimental Thermodynamics of Non-Reacting FluidsB. Le NeindreNo ratings yet
- A Tale of Two Cities (World Classics, Unabridged)From EverandA Tale of Two Cities (World Classics, Unabridged)Rating: 4 out of 5 stars4/5 (6889)
- PFR VS CSTRDocument6 pagesPFR VS CSTRSHIVAM CHATURVEDI IET Lucknow StudentNo ratings yet
- Continuous Stirred Tank Reactors (CSTRS) : Mole Balance On Component ADocument4 pagesContinuous Stirred Tank Reactors (CSTRS) : Mole Balance On Component AamalinaishahNo ratings yet
- Construction and Building Materials: Paolo ForaboschiDocument21 pagesConstruction and Building Materials: Paolo ForaboschiMarco CamposNo ratings yet
- CW9 Identifying Functions 5Document2 pagesCW9 Identifying Functions 5Xavier BurrusNo ratings yet
- Catchment CharacterisationDocument48 pagesCatchment CharacterisationSherlock BaileyNo ratings yet
- MAIN Electrical Parts List: Design LOC Sec-Code DescriptionDocument12 pagesMAIN Electrical Parts List: Design LOC Sec-Code DescriptionAndroid Schematics and CircuitsNo ratings yet
- 2023.01.25 Plan Pecatu Villa - FinishDocument3 pages2023.01.25 Plan Pecatu Villa - FinishTika AgungNo ratings yet
- SVE Event GuideDocument22 pagesSVE Event GuideMadalina MarinacheNo ratings yet
- Alfa Laval Energy Balance Casestory enDocument2 pagesAlfa Laval Energy Balance Casestory enHélder FernandoNo ratings yet
- Unit P1, P1.1: The Transfer of Energy by Heating ProcessesDocument9 pagesUnit P1, P1.1: The Transfer of Energy by Heating ProcessesTemilola OwolabiNo ratings yet
- Apacer SV250 CFast BiCS5 AA2 XX5XXX XXXEX Spec v1 - 3107344Document25 pagesApacer SV250 CFast BiCS5 AA2 XX5XXX XXXEX Spec v1 - 3107344ManunoghiNo ratings yet
- A 388 - A 388M - 03 - Qtm4oc9bmzg4tqDocument8 pagesA 388 - A 388M - 03 - Qtm4oc9bmzg4tqRod RoperNo ratings yet
- LUXEON Altilon SMDDocument25 pagesLUXEON Altilon SMDBertram PredikNo ratings yet
- Freight ForwardingDocument43 pagesFreight ForwardingEuroline NtombouNo ratings yet
- Training Schedule April 2021 To March 22 - 0Document18 pagesTraining Schedule April 2021 To March 22 - 0sanrexiNo ratings yet
- Hasnain A KhanDocument2 pagesHasnain A KhanFatima ButtNo ratings yet
- Seajet 118 Ultra-Build Epoxy PrimerDocument2 pagesSeajet 118 Ultra-Build Epoxy PrimerRai Hasnain Ahmad DahaNo ratings yet
- The Mole ConceptDocument12 pagesThe Mole ConceptVictor OkosunNo ratings yet
- 22.anaerobes (Part II)Document91 pages22.anaerobes (Part II)sigrid jaramilloNo ratings yet
- Westbury High School: Science Department Lesson PlanDocument3 pagesWestbury High School: Science Department Lesson PlanChrs TomNo ratings yet
- Managing Housekeeping Inventory: Ihm MumbaiDocument5 pagesManaging Housekeeping Inventory: Ihm MumbaiAbhishek ChowdhuryNo ratings yet
- 7383 - AdmissionTech - Aiub 19 SummerDocument5 pages7383 - AdmissionTech - Aiub 19 SummerdeshidhongbdNo ratings yet
- GFB V2 - VNT Boost Controller: (Part # 3009)Document2 pagesGFB V2 - VNT Boost Controller: (Part # 3009)blumng100% (1)
- ISKRA Letak MC3xx 2016 Ver.4.0 2016.compressedDocument6 pagesISKRA Letak MC3xx 2016 Ver.4.0 2016.compressedmelanitisNo ratings yet
- Cytokines and Interferons Types and FunctionsDocument25 pagesCytokines and Interferons Types and FunctionsSereen Abd El-rahmanNo ratings yet
- Herpes Simplex KeratitisDocument20 pagesHerpes Simplex KeratitisriskhapangestikaNo ratings yet