Professional Documents
Culture Documents
Corossion Electrolytique
Uploaded by
Yacine Byacine0 ratings0% found this document useful (0 votes)
9 views1 pageThe document attempts to determine the best sequestering agent out of citric acid, ammonium citrate, and EDTA thickened with gelatin to remove iron corrosion from tinplated metals. Tests using atomic absorption spectroscopy, colorimetry and visual observation found that EDTA was most effective at removing iron corrosion without affecting the appearance of the tin.
Original Description:
Original Title
corossion electrolytique
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe document attempts to determine the best sequestering agent out of citric acid, ammonium citrate, and EDTA thickened with gelatin to remove iron corrosion from tinplated metals. Tests using atomic absorption spectroscopy, colorimetry and visual observation found that EDTA was most effective at removing iron corrosion without affecting the appearance of the tin.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
9 views1 pageCorossion Electrolytique
Uploaded by
Yacine ByacineThe document attempts to determine the best sequestering agent out of citric acid, ammonium citrate, and EDTA thickened with gelatin to remove iron corrosion from tinplated metals. Tests using atomic absorption spectroscopy, colorimetry and visual observation found that EDTA was most effective at removing iron corrosion without affecting the appearance of the tin.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 1
attempted to determine which of three sequestering agents (citric acid, ammonium citrate, and
ethylene diamino-tetra-acetic-acid or EDTA), thickened with a 5% gelatin solution, would better
remove iron corrosion from tinplated metals. The best of the three would be the one which most
effectively removed iron corrosion while leaving the appearance of the tin unaffected. Using atomic
absorption spectroscopy, the Macbeth colorimeter and visual observations, it was determined that
EDTA was the best choice. While not as effective in removing the iron corrosion, tests showed
You might also like
- EtchingDocument11 pagesEtchingOlgalycos100% (1)
- Complexometric TitrationDocument12 pagesComplexometric TitrationBwhzad HameedNo ratings yet
- Determination of Aluminium by Back TitrationDocument5 pagesDetermination of Aluminium by Back TitrationKojo Eghan75% (12)
- Faltermeier R AMT New Corrosion Inhibitor 1992Document50 pagesFaltermeier R AMT New Corrosion Inhibitor 1992Bruno DebenjakNo ratings yet
- Astm G69 PDFDocument1 pageAstm G69 PDFMechWell0% (1)
- 3 CT-2271Document7 pages3 CT-2271KnspeisNo ratings yet
- Hydration Mechanisms of Mineral Trioxide Aggregate: J. CamilleriDocument9 pagesHydration Mechanisms of Mineral Trioxide Aggregate: J. CamilleristarlordeNo ratings yet
- XRD and Electron Microscope Study of An As-Cast 26.6% Chromium White Iron MicrostructureDocument9 pagesXRD and Electron Microscope Study of An As-Cast 26.6% Chromium White Iron MicrostructureJMarce16No ratings yet
- Chelating AgentDocument4 pagesChelating AgentsyikinabukhoriNo ratings yet
- (Aplikasi Ekstraksi) Solid-Liquid Extraction With An Ammoniacal EDTA Solution The Separation of Traces of Copper From AlumuniumDocument3 pages(Aplikasi Ekstraksi) Solid-Liquid Extraction With An Ammoniacal EDTA Solution The Separation of Traces of Copper From AlumuniumDian Rahmat YuneriNo ratings yet
- Corrosion Control of Copper in Nitric AcDocument10 pagesCorrosion Control of Copper in Nitric Aclorenaov177No ratings yet
- Chelating AgentsDocument5 pagesChelating AgentsSalil MukundanNo ratings yet
- E53-07 (Reapproved 2013) PDFDocument4 pagesE53-07 (Reapproved 2013) PDFearth metallurgical services pvt.ltd.No ratings yet
- Corrosion Characterization of AluminiumDocument4 pagesCorrosion Characterization of AluminiumHanzla ShahidNo ratings yet
- Complexometric Titrations: StructureDocument13 pagesComplexometric Titrations: StructureAnujNo ratings yet
- Copper Corrosion Inhibition by Azadirachta Indica Leaves Extract in 0.5 M Sulphuric AcidDocument4 pagesCopper Corrosion Inhibition by Azadirachta Indica Leaves Extract in 0.5 M Sulphuric AcidKArenNo ratings yet
- Very Helpful 3Document8 pagesVery Helpful 3abarenowell88No ratings yet
- 1,2,3-Triazole Derivatives As Corrosion Inhibitors For Mild Steel in Acidic Medium - Experimental and Computational Chemistry StudiesDocument11 pages1,2,3-Triazole Derivatives As Corrosion Inhibitors For Mild Steel in Acidic Medium - Experimental and Computational Chemistry StudiesDaniel GonzagaNo ratings yet
- Tetrahedron Letters: Juan Tian, Cuicui Yi, Huasheng Fang, Dayong Sang, Zhoujun He, Jiahui Wang, Yongjiang Gan, Qing AnDocument3 pagesTetrahedron Letters: Juan Tian, Cuicui Yi, Huasheng Fang, Dayong Sang, Zhoujun He, Jiahui Wang, Yongjiang Gan, Qing AnAntônio Neto MachadoNo ratings yet
- Adsorption Mechanism of Long-Chain Alkylamines On Quartz and AlbiteDocument9 pagesAdsorption Mechanism of Long-Chain Alkylamines On Quartz and AlbiteDiego VeneuNo ratings yet
- Microscopy: Rubber. Plastic. LatexDocument3 pagesMicroscopy: Rubber. Plastic. LatexSuresh MgNo ratings yet
- Chelating AgentsDocument5 pagesChelating AgentsrandatagNo ratings yet
- Waste Water ManagementDocument11 pagesWaste Water ManagementViknesh VikiNo ratings yet
- Extracting Aluminum From Dross Tailings: A.M. AmerDocument4 pagesExtracting Aluminum From Dross Tailings: A.M. Amermontie3No ratings yet
- Springnet Rusting CompatibleDocument24 pagesSpringnet Rusting CompatibleTeresa LaiNo ratings yet
- Finite Element Modelling and Validation of Thermomechanical Behaviour For Layered Aluminium Parts Made by Composite Metal Foil ManufacturingDocument18 pagesFinite Element Modelling and Validation of Thermomechanical Behaviour For Layered Aluminium Parts Made by Composite Metal Foil ManufacturingNigus TeklehaymanotNo ratings yet
- Obtaining New Components and Testing Physical-Mechanical Properties of Rubber-Metal Based Damper Device SystemsDocument7 pagesObtaining New Components and Testing Physical-Mechanical Properties of Rubber-Metal Based Damper Device SystemsCentral Asian StudiesNo ratings yet
- Extraction of Zinc and Copper-Zinc Mixtures From Ammoniacal Solutions Into Emulsion Liquid Membranes Using LIX 84I®Document8 pagesExtraction of Zinc and Copper-Zinc Mixtures From Ammoniacal Solutions Into Emulsion Liquid Membranes Using LIX 84I®ahmad syuhaibNo ratings yet
- Complexometric TitrationDocument8 pagesComplexometric TitrationYUNITA DWINo ratings yet
- 10.1016@S0013 46869600250 2 PDFDocument5 pages10.1016@S0013 46869600250 2 PDFAna LuizaNo ratings yet
- Molecular Simulation, Quantum Chemical Calculations and Electrochemical Studies For Inhibition of Mild Steel by TriazolesDocument9 pagesMolecular Simulation, Quantum Chemical Calculations and Electrochemical Studies For Inhibition of Mild Steel by TriazolesManal NaciriNo ratings yet
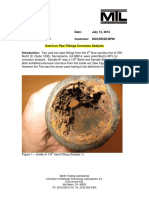
- Cast Iron Pipe Fittings Corrosion AnalysisDocument6 pagesCast Iron Pipe Fittings Corrosion AnalysisTeby RodoNo ratings yet
- An Improved Etchant For Copper and Copper Alloys: METALLOGRAPHY 9, 51-61 (1976)Document11 pagesAn Improved Etchant For Copper and Copper Alloys: METALLOGRAPHY 9, 51-61 (1976)Michael Vincent MirafuentesNo ratings yet
- Document 32Document17 pagesDocument 32rik.sengupta.08No ratings yet
- Synergistic Inhibition Effects of Octadecylamine and Tetradecyl Trimethyl Ammonium Bromide On Carbon Steel Corrosion in The H2S and CO2 Brine SolutionDocument8 pagesSynergistic Inhibition Effects of Octadecylamine and Tetradecyl Trimethyl Ammonium Bromide On Carbon Steel Corrosion in The H2S and CO2 Brine SolutionMarcus BaltesNo ratings yet
- Dosis de Reactivo FentonDocument84 pagesDosis de Reactivo FentonGUILLERMO ALEJANDRO MARTINEZ LOPEZNo ratings yet
- ch1 The Analysis of Things As They AreDocument5 pagesch1 The Analysis of Things As They AreGazel Avery SantanaNo ratings yet
- Introduction To Sputtering, Targets and Utilization PDFDocument4 pagesIntroduction To Sputtering, Targets and Utilization PDFanon_876950641No ratings yet
- Bond Strength of Two Resin Cements To Titanium After Different Surface Conditioning MethodsDocument7 pagesBond Strength of Two Resin Cements To Titanium After Different Surface Conditioning MethodsFrontiersNo ratings yet
- Advantages Using This Method in Analyzing The Element (Metals) in Environment Sample Elements Can Be DeterminedDocument2 pagesAdvantages Using This Method in Analyzing The Element (Metals) in Environment Sample Elements Can Be Determinedzifa_89No ratings yet
- Stir Casting TechniqueDocument12 pagesStir Casting TechniquehafizalNo ratings yet
- Gamma AluminaDocument5 pagesGamma AluminaAssyakurNo ratings yet
- 1 s2.0 S1877705812028020 MainDocument8 pages1 s2.0 S1877705812028020 MainJuliano. PQMNo ratings yet
- ProposalDocument24 pagesProposalRonesa SayonNo ratings yet
- DCG Recording With Red LightDocument10 pagesDCG Recording With Red LightMaximoNo ratings yet
- Journal of Engineering Sciences and Innovation: Viscosity of Potassium Carbonate Solutions Promoted by New AminesDocument8 pagesJournal of Engineering Sciences and Innovation: Viscosity of Potassium Carbonate Solutions Promoted by New Amines18-033 Wahyu PratamaNo ratings yet
- Alpha and Beta Alumina by X RDDocument26 pagesAlpha and Beta Alumina by X RDnovernandoNo ratings yet
- Calcium Silicatebased Cements and Functional Impacts of Various ConstituentsDocument11 pagesCalcium Silicatebased Cements and Functional Impacts of Various Constituentsphucbm bmNo ratings yet
- Interaction of Serotonin and Melatonin With Sodium, Potassium, Calcium, Lithium and AluminiumDocument7 pagesInteraction of Serotonin and Melatonin With Sodium, Potassium, Calcium, Lithium and AluminiumminkaNo ratings yet
- The Effect of Particle Size Distribution On Physical Properties of CastablesDocument5 pagesThe Effect of Particle Size Distribution On Physical Properties of CastablesKhaled BOUALINo ratings yet
- Electrical Conductivity in MetalsDocument19 pagesElectrical Conductivity in MetalsNicole Anne BorromeoNo ratings yet
- Title: The Corrosion of Different Metals in Salt Water and Tap WaterDocument21 pagesTitle: The Corrosion of Different Metals in Salt Water and Tap WaterYi Ling GohNo ratings yet
- Jevremovi - Et Al-2016-Materials and CorrosionDocument13 pagesJevremovi - Et Al-2016-Materials and CorrosionPaulo JuniorNo ratings yet
- The of Supported Platinum Catalysts: StructureDocument5 pagesThe of Supported Platinum Catalysts: StructurejoseramonfachadoNo ratings yet
- CanMetallQuart EffectofrelativehumidityoncoppercorrosionbyaceticandformicDocument11 pagesCanMetallQuart EffectofrelativehumidityoncoppercorrosionbyaceticandformicRicardo TatamuesNo ratings yet
- Corrosion Inhibition of Copper in Sea Water Using Derivatives of Tetrazoles and ThiosemicarbazideDocument7 pagesCorrosion Inhibition of Copper in Sea Water Using Derivatives of Tetrazoles and ThiosemicarbazideEstudiante2346No ratings yet