Professional Documents
Culture Documents
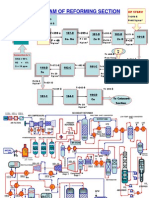
Biomass Gasification
Uploaded by
mysingaporeCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Biomass Gasification
Uploaded by
mysingaporeCopyright:
Available Formats
1/23/2013
Biomass Characterization and Gasification
CHEN 4470: Process Design Practice
Sushil Adhikari, Ph.D. Biosystems Engineering Department
January 24, 2013
Biomass Properties
Physical Properties
Density, size, shape, area
Chemical Properties
Heating value, proximate analysis, ultimate analysis
Biomass Constituents
Hemicellulose, cellulose and lignin
1/23/2013
Proximate Analysis
Proximate Analysis (weight percentage) Moisture Content (wet basis/dry basis) ASTM E871 Ash ContentASTM D1102 Volatile Matters-- ASTM E872 Fixed Carbon
Ultimate Analysis (contd.)
Ultimate Analysis (ASTM D 5373-02) Carbon (E 777) Hydrogen (E 777) Nitrogen (E 778) Oxygen Other elements-S, Cl.. Carbon, hydrogen and nitrogen are converted into carbon dioxide, water vapor, nitrogen, respectively for quantification. Usually, oxygen is calculated from the difference (100-C-H-N).
1/23/2013
Heating Value
Heating value represents the heat released when the chemical compound is stoichiometrically combusted. Heating value is expressed in terms of higher (gross) heating value (HHV) or lower (net) heating value (LHV). While measuring HHV, the products of combustion are cooled to the initial temperature of the compound. In LHV, the water produced during combustion is not condensed.
Table:Proximate,ultimateandheatingvalueanalyses(dryweightbasis)ofselectedbiomass feedstocks
Switchgrass Proximate Analysis FixedCarbon 14.34 VolatileMatter 76.69 Ash 8.97 Ultimate Analysis Carbon Hydrogen Nitrogen Oxygen Sulfur Chlorine Hybrid poplar 12.49 84.81 2.70 Pine woodchipsb 18.01 81.71 0.28 Sugar bagasse 11.95 85.61 2.44 cane Wyoming Elkol coal 51.4 44.4 4.2
46.68 5.82 0.77 37.38 0.19 0.19
50.18 6.06 0.60 40.43 0.02 0.01
49.33 5.03 0.53 44.70 0.13 0.003
48.64 5.87 0.16 42.82 0.04 0.03
71.5 5.3 1.2 16.9 0.9 n/a
HHV, MJ/kg
calculated
18.06
19.02
19.40
18.99
29.50
from difference. n/a= not available.
1/23/2013
Enthalpies of Formation
Enthalpies of formation is quite useful for thermodynamic calculations such as Gibbs free energy of minimization. The standard enthalpy of formation of a particular biomass sample is equal to the sum of heats of formation of the products on combustion minus the HHV. If you use the minus sign, then you
should use - for the HHV because of exothermicity. Otherwise, you can use plus sign without worrying any sign for the HHV.
It is assumed that ash is inert. Standard enthalpies of formation at 298 K of the combustion products are as follows: CO2 = -94.05; H2O =-68.37; NO2 =8.09; SO2 =70.95 in kcal/g-mol
Biomass Gasification
Biomass: Gasification: High Temperature (800-900oC) Products: Syngas: H2 CO CO2 CH4 CH1.44O0.66 Insufficient Oxidizing agent
(Air, O2, H2O and CO2) Small solid or liquid fractions
1/23/2013
Biomass Gasification
Partial oxidation of biomass to produce a low calorific-value fuel called syngas or producer gas. Main components of the producer gas are CO, H2, CO2, CH4, N2, and H2O. Chemical transformation can take place in fixed, moving, or fluidized bed or entrained flow gasifiers at temperatures of 1400 to 1800F with pressures from 1 to 30 atmospheres.
Syngas Potential
Source: Jenny B. Tennant. NETL Overview of DOEs Gasification Program
1/23/2013
Conversion of Syngas to Fuels
Power
Gasification Steps
1. Drying (>150 oC) 2. Pyrolysis or Devolatilization (150-700 oC) 3. Combustion (700-1500 oC) 4. Reduction (800-1100 oC) Processes 1, 2, and 4 absorb heat whereas step 3 releases heat.
Source: Prabir Basu, 2006. Combustion and Gasification in Fluidized Beds
1/23/2013
Drying
Every kg of moisture in the biomass takes away a minimum of 2260 kJ to vaporize water (Basu, 2010). Typical moisture content of freshly ranges from 30 to 60% and for some biomass it can exceed 90%. For the production of a fuel gas, most gasification system use dry biomass with a moisture content of 10 to 20%.
Pyrolysis
Complex physical and chemical processes occur during the pyrolysis process. It starts slowly at 350 oC, accelerating to an almost instantaneous rate above 700 oC. During pyrolysis process, large compounds are broken down and evaporate with other volatile components. Biomass + Heat Char Vapors/liquid (tar or PAHs) + Gases+
1/23/2013
Combustion
Oxidation or combustion is one of the most important reactions in the gasification. All the thermal energy needed for endothermic reactions are provided during this step. Oxygen supplied to the gasifier reacts with combustible products, resulting the formation of CO2 and H2O.
Gasification Chemistry
Biomass
Oxygen Syngas
Steam
Source: Jenny B. Tennant. NETL Overview of DOEs Gasification Program
1/23/2013
Reactions
Combustion Reactions Boudouard Reaction Water-Gas Reaction Methanation Reaction CO shift Reaction (Water-Gas Shift Reaction) Methane Steam Reforming Reaction
Source: Prabir Basu, 2006. Combustion and Gasification in Fluidized Beds
Reactions (cont.)
Combustion Reactions
C+1/2 O2 CO CO+1/2 O2 CO2 H2 + O2 H2O (H = -111 MJ/kmol) ( H = -283 MJ/kmol) ( H = -242 MJ/kmol)
Boudouard Reaction
C+CO2 2CO ( H = +172 MJ/kmol)
1/23/2013
Reactions (contd.)
Water-gas Reaction
C+H2O CO+H2 ( H = +131 MJ/kmol)
Methanation Reaction
C+2H2 CH4 ( H = -75 MJ/kmol)
Methane Steam Reforming Reaction
CH4+H2O CO + 3H2 ( H = +206 MJ/kmol)
Reactions (contd.)
Water-gas Shift Reaction
CO+H2O CO2 + H2 ( H = -41 MJ/kmol)
For real fuel, the overall reaction can be written as:
CnHmOp + ??O2 CO +CO2+H2+CH4+H2O+tar
10
1/23/2013
Heating Value of Syngas
The higher heating value of the syngas can be calculated by the volumetric fraction and the higher heating values of gas components, which is given by
Types of Gasifier
Updraft Gasifier
Source: Olofsson et al., 2005.
Downdraft Gasifier
Crossdraft Gasifier
11
1/23/2013
Mobile BIOMAX
Features
Field deployable. Self contained and doesnt need grid connection. 25 kWe generating capacity. 50 lbs biomass consumed per hour.
Mobile BIOMAX (contd.)
12
1/23/2013
Biomax control system
64 control points (temps, pressures, flows, motors, engine, generator, etc.) 30 auto alarms with text messaging or email. Auto remote start up and shut down. Full data logging downloadable. Remote trouble diagnosis / software upgrades. Manual on-site push button start-stop.
Fluidization Regimes
Source: Introduction to Fluidization Technology by Dr. Karl V. Jacob and Dr. Ray Cocco on April 13, 2011 at ChemE on Demand
13
1/23/2013
Types of Gasifier (cont.)
Bubbling Fluidized Bed Gasifier
Source: Olofsson et al., 2005.
Entrained Flow Gasifier
Fig.Gasconditioningsystem
Fig. AuburnUniversitysbubblingfluidizedbedgasifierandbiomassfeeder
14
1/23/2013
Advantages/ Disadvantages
Updraft Gasifier
Size, shape and moisture content of biomass particles are less critical than with a downdraft gasifier. Design is simple and results in a fairly high heating value of the gas. The quality of the syngas is generally quite low. High temperature near the reactor grate can cause blocking due to ash fusion
Source: Olofsson et al., 2005.
Advantages (cont.)
Downdraft Gasifier
Produced gas is generally of relatively good quality and has low level of tars. Up to 99.9% of the formed tar is consumed minimizing tar cleanup. Syngas contains relatively high levels of CO2 since a large portion of the biomass is oxidised. Heating value is low. Size and shape and low moisture content of biomass particles must be controlled within close limits.
Source: Olofsson et al., 2005.
15
1/23/2013
Advantages/ Disadvantages (cont.)
Crossdraft Gasifier Design is simple. Quality of syngas is generally poor. Heating value of the syngas is low and the tar content is high.
Source: Olofsson et al., 2005.
Advantages/ Disadvantages (cont.)
Bubbling Fluidized Bed Gasifier
Reactor allows high rates of throughput, higher than fixed beds. Results in good mixing, optimized kinetics, particle/gas contact and heat transfer as well as long residence time. High carbon conversion rates and, consequently, high yields. Sand bed makes it possible to use in-bed catalytic processing. Syngas is rich in particulates
Source: Olofsson et al., 2005.
16
1/23/2013
Advantages/ Disadvantages (cont.)
Entrained Flow Gasifier
Almost tar free syngas Leach-resistant molten slag A high percentage of energy is converted into sensible heat. Production of biomass powder is an extra cost.
Source: Olofsson et al., 2005.
Composition of Gas Yield
Fuel Composition Gasifying Medium Operating Pressure Temperature Moisture Content of the Fuel Mode of Bringing the Reactants into Contact
Source: Prabir Basu, 2006. Combustion and Gasification in Fluidized Beds
17
1/23/2013
Gas Composition (cont.)
Component Nitrogen CO CO2 H2 CH4 Heating Value, kJ/m3 Composition, % 50-54 17-22 9-15 12-20 2-3 5000-5900
Gas composition presented here is from downdraft gasifier operated at 20% MC.
Source: Wood gas as engine fuel. FAO 1986. pp.19
Effect of Operating Parameters
Temperature Pressure Feed Characteristics
Fuel Reactivity Volatile Matters Ash Moisture Content
Source: Prabir Basu, 2006. Combustion and Gasification in Fluidized Beds
18
1/23/2013
Volatile Matter
Fuels with high volatile matter content are easier to gasify. Also, char produced from gasification process is more porous and easier to gasify. Biomass has high volatile matters and produces high tar content. High tar content makes gas clean-up process difficult.
Source: Prabir Basu, 2006. Combustion and Gasification in Fluidized Beds
Ash Content
Ash content does not have direct influence on the gas composition. However, it affects the practical operation of gasifier. Ash can be removed either in solid or liquid form. In fixed and fluidizing beds, ash is removed in solid form. If the ash is removed in the solid form, feedstocks should have high ashmelting/softening temperatures and the gasifier should be operated at well below melting temperature.
19
1/23/2013
Ash Content (cont.)
The relationship between ash melting temperature and composition is a complicated. It mainly depends on SiO2-Al2O3-Cao-FeO. High in silica and alumina will result high in ash-melting temperature. But, the ratio of silica/alumina is also equally important. It is reduced by the presence of CaO and FeO. Ash-melting temperature of coal is more than 1200 oC but biomass can have significantly lower than 950 oC.
Ash Content (cont.)
20
1/23/2013
Syngas Composition from Different Feedstocks
Constituents Fraction (N2 balance)
25.0 Peanut hulls Saw dust Poultry Litter Wood chips
Higher Heating Value
6.0
20.0
5.0
15.0 %vol. MJ/m3 O2 CO CO2 CH4 H2
4.0
3.0
10.0
2.0 5.0 1.0
0.0
0.0 Peanut Saw dust Poultry hulls Litter Wood chips
Gautam et al. (2009), ASABE Annual International Meeting. June 21-June 24, 2009, Reno, NV
Gasification Processes and Methanol Production
Process Condition Circulating fluidized bed Feedstock (wood), t/d Steam, t/t dry feed Oxygen, t/t dry feed Air, t/t dry feed Gas. Temp., oC Gas. Press., psi Exit gas (dry) H2 (vol.%) CO (vol.%) CH4 (vol.%) CO2 (vol.%) H2/CO
Source: Klass (1998).
Gasifier Type Bubbling fluidized bed 1650 0.3 0.3 0 982 507 Entrained 1650 0.03 0.5 0 1045 357
1650 0.31 0 1.46 927 14.8
21.1 46.8 14.9 11.3 0.45
30.7 22.2 12.0 35.2 1.38
33.9 50.7 0.2 14.9 0.66
21
1/23/2013
Design Consideration
Gasifier Efficiency
Cold gas efficiency Hot gas efficiency
Carbon Conversion Equivalence Ratio
Cold gas efficiency = (Heating value of product gas/Heating value of feedstocks)x100 % It is important to specify whether the heating values are on higher heating value or lower heating value basis.
Design Consideration (contd.)
The gas is not cooled before combustion and the sensible heat is also useful. Therefore, sometimes, hot gas efficiency is also used for such applications. Hot gas efficiency = (Heating value of product gas + Hsensible /Heating value of feedstocks) x 100 %
22
1/23/2013
Design Consideration (contd.)
Carbon conversion = {1 Carbon in gasification residue/Carbon in feedstocks} x 100 %
or
{Carbon in gas composition/Carbon in feedstocks} x 100 %
Care is required to interpret the data. Higher methane concentration could result in higher cold efficiency and good for power application but it is not the optimum choice for a synthesis gas applications to produce fuels and chemicals.
Design Consideration (contd.)
Equivalence Ratio (ER): = (A/F)actual/(A/F)stoichiometric The quality of syngas depends upon the value of ER. A low value of ER (<0.2) results in several problems including excessive char formation. A high value of ER (>0.4) results in excessive formation of CO2 and H2O. Typical range of ER is ~0.2 -0.4.
23
You might also like
- Single Disc Clutch DesignDocument32 pagesSingle Disc Clutch DesignWeins GemerlapNo ratings yet
- Lilin Downhole MotorDocument35 pagesLilin Downhole MotorIAN.SEMUT100% (2)
- Advanced Biomass Gasification: New Concepts for Efficiency Increase and Product FlexibilityFrom EverandAdvanced Biomass Gasification: New Concepts for Efficiency Increase and Product FlexibilityRating: 3 out of 5 stars3/5 (2)
- IEA The Status of Large Scale Biomass FiringDocument88 pagesIEA The Status of Large Scale Biomass Firinggakguk100% (1)
- 14 Gravity Separator Design - PowerDocument22 pages14 Gravity Separator Design - Powermatrix69100% (1)
- Biomass Gasification Tech UKDocument130 pagesBiomass Gasification Tech UKGuna Skrodere100% (2)
- F3 Maths 2012 1stexam Paper1Document3 pagesF3 Maths 2012 1stexam Paper1YiuhangLeung100% (2)
- Biomass Gasification and Pyrolysis Formatted Full ReportDocument41 pagesBiomass Gasification and Pyrolysis Formatted Full Reportanon_477439625100% (1)
- A Technical and Economic Assessment of CO2 Capture Technology For IGCCDocument293 pagesA Technical and Economic Assessment of CO2 Capture Technology For IGCCapi-3799861100% (1)
- Chapter No. 1: Catalytic Steam Gasification of MSWDocument79 pagesChapter No. 1: Catalytic Steam Gasification of MSWMUh Ammar100% (1)
- Mercury Gemini Program Design Survey. NASA ERC Design Criteria Program Stability, Guidance and ControlDocument217 pagesMercury Gemini Program Design Survey. NASA ERC Design Criteria Program Stability, Guidance and ControlBob Andrepont100% (1)
- Energy Conversion and ManagementDocument7 pagesEnergy Conversion and ManagementhusseinhshNo ratings yet
- Gas Yields Table Provides Approximate Biogas DataDocument3 pagesGas Yields Table Provides Approximate Biogas DataramiraliNo ratings yet
- Simulations and Modeling of Biomass Gasification Processes - MITDocument173 pagesSimulations and Modeling of Biomass Gasification Processes - MITGXGGXG100% (1)
- Ammonia and Urea ProductionDocument10 pagesAmmonia and Urea Productionwaheed_bhattiNo ratings yet
- Designing Process VesselsDocument4 pagesDesigning Process VesselsJanaki Devi ParratNo ratings yet
- What Is PyrolysisDocument3 pagesWhat Is PyrolysisHazim Calixto LimmayogNo ratings yet
- Biomass Pyrolysis: Salman ZafarDocument5 pagesBiomass Pyrolysis: Salman ZafarSalman Zafar100% (2)
- Dynamic Soil Structure Interaction - 01 - Chapter 1 - NaganoDocument42 pagesDynamic Soil Structure Interaction - 01 - Chapter 1 - Naganonevam21100% (1)
- TM - 11-5855-214-23&p - (N04596) PDFDocument65 pagesTM - 11-5855-214-23&p - (N04596) PDFtyra24No ratings yet
- Naphtha Steam Reforming For Hydrogen ProductionDocument9 pagesNaphtha Steam Reforming For Hydrogen ProductionsatishchemengNo ratings yet
- Biomass GasificationDocument10 pagesBiomass GasificationKashif Ahmed100% (2)
- Biomass Gasifier CalculationDocument10 pagesBiomass Gasifier CalculationH.J.Prabhu100% (3)
- Basic Design of a Fluidized Bed Gasifier for Rice HuskDocument8 pagesBasic Design of a Fluidized Bed Gasifier for Rice HuskAnna Yunita SitompulNo ratings yet
- 2014 - CO2 Capture Using Biochar Produced From Sugarcane Bagasse and Hickory WoodDocument27 pages2014 - CO2 Capture Using Biochar Produced From Sugarcane Bagasse and Hickory WoodpreemeeNo ratings yet
- Gasification of Municipal Solid WastesDocument8 pagesGasification of Municipal Solid WastesSalman Zafar100% (2)
- Aitkin County Plasma Gasification Assessment FINAL2 PDFDocument32 pagesAitkin County Plasma Gasification Assessment FINAL2 PDFpijusmagnificus54No ratings yet
- Thermal Gasification For Power and FuelsDocument105 pagesThermal Gasification For Power and FuelsRodolfo Barbosa YoungNo ratings yet
- ANAEROBIC DIGESTION, GASIFICATION AND PYROLYSIS FOR MUNICIPAL SOLID WASTEDocument22 pagesANAEROBIC DIGESTION, GASIFICATION AND PYROLYSIS FOR MUNICIPAL SOLID WASTEJoao Minho100% (1)
- CO2 Methanation for Renewable Energy Storage in Gas GridsDocument6 pagesCO2 Methanation for Renewable Energy Storage in Gas GridszirimiaNo ratings yet
- Bio Oil PDFDocument87 pagesBio Oil PDFNaga Sai BNo ratings yet
- Main Routes For The Thermo-Conversion of Biomass Into Fuels and Chemicals.Document11 pagesMain Routes For The Thermo-Conversion of Biomass Into Fuels and Chemicals.Julio Cesar Jimenez BautistaNo ratings yet
- PyrolysisDocument19 pagesPyrolysiskk_joshi13No ratings yet
- Pyrolisis and GasificationDocument704 pagesPyrolisis and Gasificationfsijest75% (4)
- Thesis Report FullDocument104 pagesThesis Report FullStory SungNo ratings yet
- Fast Pyrolysis Development - Venderbosch Et Al. 2010Document31 pagesFast Pyrolysis Development - Venderbosch Et Al. 2010Reginaldojoaquim100% (1)
- Biomass Technology ReviewDocument44 pagesBiomass Technology Reviewdavid.spiderweb396191% (11)
- Biogas Plant: Biogas Digester Design, Gas Production and PurificationFrom EverandBiogas Plant: Biogas Digester Design, Gas Production and PurificationNo ratings yet
- Biomass - Power PlantDocument16 pagesBiomass - Power PlantVinay MishraNo ratings yet
- ICED OJK Overview of Biogas Power 160926 BK FinDocument57 pagesICED OJK Overview of Biogas Power 160926 BK FinDigdo Setyaji100% (1)
- Danish Centralised Biogas PlantsDocument28 pagesDanish Centralised Biogas PlantsXazolNo ratings yet
- Waste IncinerationDocument111 pagesWaste Incinerationramirali100% (2)
- Biogas Generation A Study: By: Madhav Mohan Bsc. Physics (Hons.) Ii Year Roll Number: 1815052Document17 pagesBiogas Generation A Study: By: Madhav Mohan Bsc. Physics (Hons.) Ii Year Roll Number: 1815052Madhav Mohan100% (1)
- Gassification For Practical ApplicationsDocument350 pagesGassification For Practical ApplicationshugogalindoNo ratings yet
- BIOMASS GASIFICATION FOR TRANSPORTDocument72 pagesBIOMASS GASIFICATION FOR TRANSPORTBruno NavarroNo ratings yet
- FLASH PYROLYSIS FOR BIO-OIL PRODUCTIONDocument19 pagesFLASH PYROLYSIS FOR BIO-OIL PRODUCTIONerkiruthiraj100% (1)
- Characterization of Biochar from Fast Pyrolysis and Gasification SystemsDocument11 pagesCharacterization of Biochar from Fast Pyrolysis and Gasification SystemsJean Carlos Gonzalez HernandezNo ratings yet
- Biomass Gasification Overview Presentation)Document48 pagesBiomass Gasification Overview Presentation)api-3799861100% (3)
- Physical Conversion TechnologiesDocument89 pagesPhysical Conversion TechnologiesIttihad KhanNo ratings yet
- Generate Power from SyngasDocument4 pagesGenerate Power from SyngasConstantin StanNo ratings yet
- Kinetic Model of Biomass GasificationDocument7 pagesKinetic Model of Biomass GasificationjuaanxpoonceNo ratings yet
- Project Biomass Wood BrazilDocument34 pagesProject Biomass Wood Brazilmar4478100% (2)
- Co-firing of Coal and RDF in SuspensionDocument19 pagesCo-firing of Coal and RDF in SuspensionmjbotelhoNo ratings yet
- 2011 UK Waste WoodDocument31 pages2011 UK Waste WoodStuart JonesNo ratings yet
- Biomass EnergyDocument1 pageBiomass EnergyrapNo ratings yet
- Anaerobic Digesters: Frequently Asked Questions: What Is The Anaerobic Digestion Process?Document4 pagesAnaerobic Digesters: Frequently Asked Questions: What Is The Anaerobic Digestion Process?Kate MayerNo ratings yet
- Husk Power Systems (Introduction Updated)Document14 pagesHusk Power Systems (Introduction Updated)prabhat_praveenNo ratings yet
- Gasification of Municipal Solid Waste in The Plasma Gasification Melting ProcessDocument23 pagesGasification of Municipal Solid Waste in The Plasma Gasification Melting ProcessArjuncv100% (3)
- Hydrogen As Energy Source PDFDocument3 pagesHydrogen As Energy Source PDFAlfredo Merizalde AvilesNo ratings yet
- Prefeasibility Report Rajasthan Ganganagar As Per MoEF GuidelinesDocument25 pagesPrefeasibility Report Rajasthan Ganganagar As Per MoEF GuidelinesAmol DeshmukhNo ratings yet
- Pyrolysis of Municipal Plastic Wastes For Recovery of Gasline-Range HydrocarbonsDocument6 pagesPyrolysis of Municipal Plastic Wastes For Recovery of Gasline-Range HydrocarbonsElizabeth ThompsonNo ratings yet
- Plasma GasificationDocument6 pagesPlasma GasificationIntan Permata LaksmiNo ratings yet
- WCECS2012 pp1154-1158 PDFDocument5 pagesWCECS2012 pp1154-1158 PDFmysingaporeNo ratings yet
- AmmoniaDocument2 pagesAmmoniamysingaporeNo ratings yet
- Design of Vapor-Liquid Separator (Knockout Drum, Flash Drum)Document2 pagesDesign of Vapor-Liquid Separator (Knockout Drum, Flash Drum)KNNo ratings yet
- 4460 OptimizationDocument2 pages4460 OptimizationmysingaporeNo ratings yet
- Astm F1717-21Document11 pagesAstm F1717-21wenhsiaochuanNo ratings yet
- The Second Term Exam of EnglishDocument2 pagesThe Second Term Exam of Englishsof chimiste100% (1)
- Induction Heating - VerDocument2 pagesInduction Heating - Verdenivaldo2009No ratings yet
- Noise Margin Definition ExplainedDocument10 pagesNoise Margin Definition ExplainedAnil BhardwajNo ratings yet
- Lab Report 5Document6 pagesLab Report 5Sarwar Hosen SimonNo ratings yet
- Under The Aegis Of: WWW - Ucd.ie/cigrDocument430 pagesUnder The Aegis Of: WWW - Ucd.ie/cigrErin Walker100% (1)
- Aminpro FK TestDocument9 pagesAminpro FK TestpeilinlanNo ratings yet
- Adrian Stan MFQMCourseHsL2006Document60 pagesAdrian Stan MFQMCourseHsL2006禿公No ratings yet
- Determining Dielectric Constants Using A Parallel Plate CapacitorDocument5 pagesDetermining Dielectric Constants Using A Parallel Plate CapacitorAhmAd GhAziNo ratings yet
- Pivot Interactives Motion Graphing A Dry Ice Puck On A RampDocument2 pagesPivot Interactives Motion Graphing A Dry Ice Puck On A RampSophia0% (1)
- LM 08Document38 pagesLM 08Tôi Là Đăng TèoNo ratings yet
- Thermal Engineering PDFDocument76 pagesThermal Engineering PDFKartik KuriNo ratings yet
- Midas FEADocument2 pagesMidas FEACristian Camilo Londoño PiedrahítaNo ratings yet
- Wiring DiagramDocument24 pagesWiring DiagramReji Raju0% (1)
- Mousavi, Aliha, Imani - 2020 - On The Use of Edge Cracked Short Bend Beam Specimen For PMMA Fracture Toughness Testing Under Mixed-ModeDocument1 pageMousavi, Aliha, Imani - 2020 - On The Use of Edge Cracked Short Bend Beam Specimen For PMMA Fracture Toughness Testing Under Mixed-ModeMorteza AtaeiNo ratings yet
- Numerical Exploration of The Dang Van High Cycle FDocument18 pagesNumerical Exploration of The Dang Van High Cycle FHsan HaddarNo ratings yet
- Curriculum-Of Mathematics Government College Women University, SialkotDocument119 pagesCurriculum-Of Mathematics Government College Women University, SialkotHuzaifa GurmaniNo ratings yet
- Final PPT 3rd SemDocument16 pagesFinal PPT 3rd SemmonuNo ratings yet
- E1213-14 Práctica Estándar para La Diferencia Mínima de Temperatura Resoluble para Sistemas de Imágenes TérmicasDocument3 pagesE1213-14 Práctica Estándar para La Diferencia Mínima de Temperatura Resoluble para Sistemas de Imágenes Térmicasfredy lopezNo ratings yet
- Electric Current and Charge RelationshipDocument9 pagesElectric Current and Charge RelationshipLokman HakimNo ratings yet
- Ordinary Differential EquationDocument20 pagesOrdinary Differential EquationRadeanindaNo ratings yet
- Dosing Pump Innovata Drive ConceptDocument5 pagesDosing Pump Innovata Drive ConceptgarpNo ratings yet
- A Brief Overview of The Holographic TechnologyDocument5 pagesA Brief Overview of The Holographic TechnologyAltiel Ltd.No ratings yet
- ChemDocument2 pagesChemBaliuag Guia100% (4)