Professional Documents
Culture Documents
A Rapid Method For Determining Compound Composition Cement Clinker: Application To Closed Loop Kiln Control
Uploaded by
qamhOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
A Rapid Method For Determining Compound Composition Cement Clinker: Application To Closed Loop Kiln Control
Uploaded by
qamhCopyright:
Available Formats
P. K.
Mehta"
M. J. Shah
A Rapid Method for Determining Compound
Composition of Cement Clinker: Application to
Closed Loop Kiln Control
Abstract: A procedure for determination of phase composition
of commercial cement clinker using quantitative x-ray powder diffraction analysis is described. An external standard and comparison of peak heights rather than the peak areas are used for rapid analysis. Several standard curves were prepared using diffraction patterns from known mixtures
of previously prepared pure components
which exist in cement clinker. For purposes of comparison, both the suggested external standard method and the internal standard
method which is generally accepted for quantitative X-ray diffraction analysis were used.
It was found that the faster external standard
method gives analysis of important clinker constituents with accuracy adequate for quality control in a cement plant. With the use of
suggested grinding procedure and an automatic sample changer attachment to standardX-ray diffraction equipment, it is possible to
obtain an on-line analysis of clinker in one half hour. This analysis can then be deployed in performing closed loop control
a cement
of
kiln with a proposed algorithm.
Introduction
The properties of cement are influenced by its compound
composition, that is, by the relative proportions of its
chemical constituents; however, no directmethod for
determiningcompoundcompositionis
in usebycommercial cement manufacturers at present. Rather, an indirectprocedureisfollowed
in which the product is
subjected to chemical analysisand compound composition
is computed from the analysis under the assumption of
equilibrium reaction conditions.
A problem that has plagued the cement industry is the
absence of a rapid method for chemical analysis of the
final product from the kiln-commonly known as clinker.
Conventional gravimetric methods for complete chemical
analysis of clinker require approximately 24 hours to test
the clinker quality. Because
of the high production rate of a
kiln, and because of the indirect procedurefor determining
compound composition,which frequently yields erroneous
results,' considerable loss in production or deterioration
of product quality may occur during the time required for
analysis.
While
several
have
been
made
to
use x-ray diffraction analysis for directly ascertaining the
compound composition of cement clinker, the procedures
employed by previousworkershavebeenmore
or less
106
University of California at Berkeley. Present address: Jaipur Udyog
Cement Works, Savaimadhavpur, Rajastan, India.
IBM JOURNAL
JANUARY
1967
useful only as research tools due to the long time (5-12
hours) required for analysis.
In this paper, an x-ray diffractionprocedure is described
for analysis of those cement compoundswhich significantly
affect product quality. Usingthis procedure in combination
with proposed methods for sample preparation and grinding and with the use of an automatic sample changer, it
should be possible to accomplish the desired analysis in
about one-half hour. Since the processing timefor material
in the kiln is of the order of 5 hours, the procedure offers
the possibility of on-linecomputer control of product
quality.
Scope of investigation
Present attempts to improve the performance of a cement
kiln through the use of digital and analog computers rely
mainly on:
(1) Optimum blendingof the raw materials using elemental
analysis of quarry materials byx-rayfluorescence.
(2) Control of burning-zone temperature in the kiln.
(3) Prediction of ring formation and other phenomena
which cause shutdowns so that proper corrective actions based on operator experiencemaybetaken.
(4) Attempts to minimize the effects of such external dis-
turbances as varying ambientconditions and inconsistent fuel quality.
Because the performance of a cement kiln is dependent
on so many parameters, any one or all of these steps will
not assure uniform clinker quality, even through it may
significantly improve kiln performance.For example, Step
1 providesonlyelementalanalysis,
and if the crystalline nature of SiO, in raw material changes from clay to
quartz, the burnability of clinker is drastically changed,
and even with control of burning zone temperature, the
clinkerquality may be seriouslyaffected.Ananalytical
tool which can provideproduct quality information rapidly
can be of invaluable assistance in kiln control. This information can beused as feedback control on several
directly or indirectly controllable variables (such as burning zone temperature, kiln speed, raw material blending,
etc.).
As far as on-line control is concerned, the analytical
procedure for determining product compositionneed
not be accurate in the sense of measuring exact composition. Oneis interested only in an accurate assessment of the
relative shift inweight fraction of one, two, or more compounds. For example, an increase in dicalcium silicate,"
either sudden or gradual, over a period of several hours,
may be an indication of lower burning zone temperature
due to an increase in feed rate to the burning zone, a decrease in fuel rate and/or calorific value of the fuel, an
incorrect ratio of limestone to silica, or one of several other
factors. Once the increase in C2S is detected, a computer
program may be used to identify those variables which
could causethe increase and corrective action may be taken
to change the control variables by an amount proportional
to the percent variation of C,S from its normal operating
value.
It is not the purpose of this paper to give an accurate
relationship between change in cement composition
and
the operating variables of a cement kiln, the subject being
extremely
complex as indicated by the
Rather,
it is intended to report a rapid analytical technique that is
regarded as a step toward the goal of closed-loop quality
control of cement clinker composition withthe assistance
of an x-ray diffraction techniquethat yields phase composition rapidly. In the followingsections, the theory of
quantitive x-ray powder diffraction analysis and the proposedexperimentalprocedure
are described. The final
section presents a discussion of results and illustrates how
one can use this
analytical tool for closed-loop control
of a cement kiln.
*
CIS
CzS
CsA
The notation of cement chemistry will be used hereafter; in it,
= 3 CaO.SiOz;
=2
=3
CaO.SiOs;
CaO .A1-03;
C A F = 4 Ca0.AlrOs.FesOs.
Theory
When a sample consisting of several compounds is subjected to x-ray powder diffraction, the quantitative determination of any compound, i, in the sample is theoretically
possible by the application of the following relationship,
which is the basic equation for Quantitative X-ray Diffraction Analysis' (QXDA).
where
Xi = Proportion by weight of compound i in the mixture.
Zi = Intensity of a selected diffraction peak due to compound i in the mixture.
I,' = Intensity of the selecteddiffractionpeakdue
to
compound i when a pure specimen of compound i
is x-rayed under identical test conditions.
p,,, = Mass absorption coefficient of the mixture.
pi = Mass absorption coefficient due to compound i
alone.
It is obvious from Eq. (1) that if p, and pi are not
significantlydifferent, the quantitative determination of
compound composition could be achieved by direct comparison of peak intensities, Zi and::Z
If the mass absorption coefficientof a compound is
significantly different from the mass absorption coefficient
of the mixture of which it is a constituent, the frequently
used procedure for QXDAconsists of intergrinding a
known proportion, Xs,of internal standard* with the
sample and determination of the unknown, Xi, by applying
the following relationship:
where
Z8 = Intensity of the internal standard peak in the mixture.
Iso = Intensity of the internal standard peak when a pure
specimen of the internal standard is x-rayed under
identical test conditions.
ps = Mass absorption coefficient due to the internal
standard.
K = A constant which can be evaluatedfrom the samples
containing known amount of Xi.
Eq. (3) presents a method whereby differences between
the mass absorption coefficientof a compound and the
* Any inert material, with a strong peak in the vicinity of peaks selected
for QXDA of compounds in the mixture, and with no overlapping of the
internalstandardpeak
by other peaks, can be used as internal standard.
1oz
RAPID MEASUREMENT OF CLINKER COMPOSITION
mixture containing it do not affect the results of QXDA
because determination of properties of Xiin the sample is
possible by direct comparisonof the peak intensitiesof the
compound and the internal standard.
The application of the internal standard method to
QXDA of compounds in portland cementhasbeen reported by several investigators, including Kantro et al,,
Sm~lczyk,~
Midgley et a1: and Berger et al.5The method
involves precisely weighing predetermined amounts of the
sample and the internal standard, intergrinding themin the
presence of a dispersing fluid for 2-3 hours, drying the
specimen by vacuum and heat treatment (1-2 hrs), subjecting the powder to x-ray analysis and, finally,determining the peak intensities by measuring area under the
peaks with a planimeter.* It is obvious that the procedure
describedabove is not amenable to on-lineanalysis of
portland cement clinker, especially due to the long time
required for the sample preparation.
Eq. (l), however, can be modified as follows:
X ,= I,!!&
ZioP<
or
Experimental procedure
x;- zi
-
"
IiOELt
E
L
n
I i I s 0 - K - -Ii
- "_
zsoziopi
Is0 '
(4)
where Iso canbemade
constant by using an external
standard which assures that the test conditions for x-ray
diffraction of the sample remain identicalto those usedfor
determination of K from a series of standard samples having known amounts of X,.Equation (4) then reduces to:
x' -
KZ,.
"
(5)
Pm
Furthermore, if pCLm
does not changeappreciably for a
series of samples consisting of varying proportions of a
number of compounds, then:
Xi = K I i .
(6)
Equation (6) suggests that an easy way to perform online analysisof cement compounds isby direct comparison
of peak intensities obtained by x-ray diffraction of clinker
specimens from the rotary kiln.
C,S (tricalciumsilicate) and C,S(dicalciumsilicate)
constitute about 80 percent of normal portland cement
clinker. As the mass absorption coefficient of C,S is 102,
and that of C,S is 95, it is expected that minor variations
in their amounts would not influence the mass absorption
coefficientof clinker. The mass absorption coefficientof
108
* Berger, et al.6 use adigitaldata collection systemandacomputer
analyze the results recordedon punched paper tape.
P. K. MEHTA AND M. J . SHAH
C,A (tricalciumaluminate), whichmay constitute 5-15
percent of normal portland cement,is91,whereas,
the
mass absorption coefficientof the ferritephase, which
usually constitutes 7-10 percent of normal portland cement, and the chemical compositionof which approximates
the formula C,AF, is about 140. As the mass absorption
coefficient of the ferrite phaseissignificantlydifferent
from the other major constituents of portland cement
clinker, it is obvious that variations in the ferrite phase
content of clinker affect the validity of Eq. (6), which is
based on the assumption that the mass absorption of
samples containing different proportions of certain compounds remains fairlyconstant.
On the theoreticalconsiderations,therefore, it is obvious that while the internal standard technique is more
accurate, the external standard technique appears to be
rapid and readily adaptable to on-lineanalysis. In this
investigation we have used both the internal and external
standard techniques to prepare standard curves with the
purpose of showing that for on-lineanalysisthemore
rapidexternal
standard method gives adequate comparison.
to
In order to obtain the standard specimens of portland
cementclinker, the individual constituent compounds,
C,S, C,S, C3A, and C,AF, were made separately and then
blended in required proportions. In this manner, specimens containing a wide range of k n m compound compositions were prepared.Details of procedures on the
preparation of compounds, preparation of standard specimens and their QXDA will be now discussed.
Preparation of pure compounds
The preparation of pure compoundswas carried out at the
facilities of the Engineering Materials Laboratory of the
University of California, Berkeley. Stoichiometric preparations of analytical-reagent-grade calciumcarbonate, silicic
acid, aluminum hydroxide and iron oxidewereused to
prepare raw mixes for making the pure compounds. Each
raw mix was ground in a pebble mill, made into a paste
with distilled water,and dried to disk-shaped cakeswhich
were heat-treated repeatedly in a furnace until no impurities in the desiredcompound were detectable by x-ray
diffraction.
In the case of C3S, small amounts of magnesium oxide
were added to the raw m i x so that the chemical analysis of
the final product showed one percentMgO and one percent
Al,O,. This servedto stabilize the C3Sin monoclinic form,
which is the usual form of C,S* in commercial portland
cements. In the caseofC,S,
the commercialcements
normally contain the beta form of C2S. Aboutone percent
* Cas so substituted is called alite, but for the purpose of maintaining
uniform nomenclature, the given formulais used.
CaO was used as a stabilizer for beta C,S. In addition,
quickcooling of the product was required in order to
prevent its conversion to gamma C,S. No special problems
were encountered in the preparation of C3A andthe
ferrite phase.
Table 1 Compound composition of standard clinker samples
Sample
c3s
CB
CaA
ClAF
Ia
30%
30
30
50%
50
50
50
10%
5
0
15
10%
15
20
5
10
5
0
15
15
20
5
Ib
IC
Preparation of standard specimens
Commercial portland cement clinker may contain 30-70
percentC3S, 10-50 percent C,S, 0-15 percent C3A and
5-20 percent** ferrite phase, C,AF. Normally, the calcium
silicates amount to about 80 percent, whereas,the remaining 20 percent of clinker is constituted of the tricalcium
aluminate and the ferrite phase. Table 1 shows the data
on twenty standard specimens which were correspondingly
designed to cover a broad range of compound compositions. The pure compounds, ground to pass200-mesh
sieve,were proportioned in accordance with the plan
shown in Table 1. The mixes were contained in air-tight
plastic vials and were blended together thoroughly by a
mechanical blending device.
Id
30
IIa
40
40
10 40
40
IId
40
40
40
40
30IIIa
30IIIb
30IIIC
30IIId
50
50
50
50
IVa
60
60
IIb
IIC
Vb
vc
Vd
Selections of peaks for QXDA
Table 2 shows the peakswhichhavebeenselected
for
quantitative representation of the variouscompounds.
Kantro et a1' use 41.0' 20 and 51.8' 20 peaks in conjunction with a set of mathematicalequations for determination
of C,S and C,S content in cement. They state that their
choice of thesepeaksisgovernedby
the consideration
that the diffraction pattern of C3S does not contain any
strong peaks which are not overlapped by C2S peaks and
vice versa. Onthe other hand, other investigators, including
Smolczyk3 and M i d g l e ~ believe
,~
that 30.1' 20 and 51.8'
20 peaks in C3S pattern are not overlapped by any C,S
peak and are adequate for quantitative determination of
C3S. Similarly, the 31.1 ' peak in the clinker pattern is due
solely to beta C,S and the 33.2' and 33.8' peaks are due
solely to C3A and C,AF, respectively.
Equipment for x-ray diffraction analysis
The equipment used for x-ray diffraction analyses consisted
of an x-ray generator having a Cu target and a Ni filter, a
diffractometercoupledwith a scintillation counter, and
an x-y recorder. This is a standard type of equipment
available in x-rayanalytical laboratories. The samples were
x-rayed under the followingconditions:
Voltage =
Current =
Receiving slit =
Base line =
Pulse height =
Time constant =
40kV
35 mA
0.0006 in
9V
19.8 V
4.
* * Except white cement which, being low in FenOa, has a very small amount
of ferrite phase.
5
0
15
60
60
1020
20
20
20
10 70
70
70
15 70
10
10
10
10
IVb
IVC
IVd
10 Va
10
10
15
20
5
10
5
15
20
15
5
0
15
20
5
Table 2 Peaks selected for QXDA analysis
Compound
~~~
Degrees 20 (Cu, Ka)
d, A
cas2.97
beta C2S
C3A2.70
CdAF
2.93
background
30.1
31.1
33.2
33.8
30.5
2.87
2.65
Theoretically, the peak intensities are better represented
so in the internal
by peakareas rather than by peak heights,
standard method, step counting at 0.02 degrees 20 at
3,200 counts per second was adopted.
From the analysis of diffraction patterns it was, however,established that considerable overlappingbetween
the peaks made the determination of area under a particular peakdifficult.Hence, practical considerations of
on-linecementanalysis made peak-heightmeasurement
the preferred method for representingpeakintensities.
Measurement of peak heights made it possible to use the
faster procedure (external standard method) which involved scanning the specimens in 29-34 degrees 20 range
at 1/4 degree 20 per minute and 100 counts per second
counting rate. A silicondiskwasused
as the external
standard.
109
RAPID MEASUREMENT OF CLINKER COMPOSITION
30
"
50 -
25
40 -
30
WN
20 -
xa
y - 1
IO-
'
IRatio of peak heights
Ratio of peak heights
+ A
Ratio of peak heights
IRatio of peak heights
Figure 1 Calibration curves for internal standard method. Internal standard is the
36.1" peak of TiO,. Computed ratios compareheights of (a) 30.1" CSS peak, (b) 31.1" C2Speak, (c) 33.2" CSA peak, (d) 33.8" CAF peak,tothatstandard.
Figure 2 Calibrationcurves for externalstandardmethod.Externalstandardissilicon
disk. Directcomparison
of peak
heights with standard is given for (a) 30.1" Caspeak, (b) 31.1" C,S peak, ( c ) 33.2" CSA peak, (d) 33.8" CaAFpeak.
ax
IPeak height in counts
15
10
15
I Peak height in counts
20
25
Preparation of sample for x-ray diffraction analysis
110
In the internal standard method, 20 percentTiOz was
used as internal standard. Two gramsof precisely weighed
sample of a standard cement and 0.4 grams of precisely
weighed TiOz were ground for one hour in a mechanical
25 ml of trichloroethylene as a disAlundum grinder, using
persing agent. The ground paste was dried in a vacuum
drying oven for about an hour in order to drive off the
dispersingagent. In the external standard method, the
incorporation ofTiO,was omitted; hence, no weighing
of the standard sample was necessary. In order to obtain a
finely ground specimen, however, the same grinding and
dryingprocedure, as usedabove for internal standard
method, was adopted.
In addition to finegrinding,prevention of preferred
orientation in the sample is necessary for quantitative
x-ray
diffraction
work.
In the present
investigation, this
P. K. MEHTA AND M. J. SHAH
$Lkihkk
eak height in counts
1 Peak height in counts
wasachievedbyplacing
an aluminumsampleholder,
open at both ends, on top of a clean glass slide, filling the
cavity lightly with the sample and sealing off the front
opening of the holder with another glass slide fixed to it
firmly with transparent tape. The surface of the specimen
on the back side of the holder was now readyfor exposure
to the x-rays. In a few preliminary trials on a standard
specimen, reproducible peak heights were obtained, thus
assuring a minimum of preferred orientation when the
samples were prepared in the above manner.
Results and discussion
Table 3 shows the data on ratio of heights of peaks selected
to represent the given compoundsquantitatively to the
height of the internal standard peak. These data have been
used to plot standard calibration curves for Cas, C,S,
C,A and C,AF, which are shown in Figs. la, b, c and d
respectively. The samples were x-rayed only once, so that
the resultscould not berechecked as required by the
normal recommended
practice
for quantitative x-ray
diffraction analysis.Hence, about 10 percent of the recordedpoints, whichwere abnormal, havebeendisregarded in the construction of standard curves.
Table 4 shows the data on direct comparison of peak
heights by the external standard method. The data is
plotted to yield standard calibration curves for Cas, C2S,
C,A and C,AF in Figs. 2a, b, c, and d, respectively.
By comparing the standard curvesbased on internal
standard method with those basedon the external standard
method, it can be observed that both the methods have
yielded comparable straight line calibration curves. Table 4
shows that by the external standard method, good reproducibility of 30.1' Cas peak and 31.1' C2S peak is possible,*whilefairreproducibility
for 33.2O C3Apeak and
33.8" C,AF peak is obtained. Some of the abnormal values
for C3A and CIAF phases could perhaps have been rectified if the samples had been x-rayed againto check for the
existence of preferred orientation.
Considering that the internal standard method requires
preciseweighingofboth
the sample and the internal
standard, that it is necessary to intergrind them in a dispersant for 2-3 hours and then to vacuum-dry the slurry
for 1-2 hours, and that the x-ray diffraction (step scanning) operational time is about 12 hours per sample,
the resultsobtainedwith
the rapid external standard
method appear to be very encouraging, Berger et al6 have,
however,recentlydeveloped
a fast (one to two hours
exclusiveof sample preparation time) internal standard
procedure of QXDA for routine mineralogical analysisof
portland cement in the laboratory. This method utilized a
computer to analyze the resultsrecorded on punched
papertape. In the presentinvestigationemploying the
external standard procedure, the operational period for
x-ray diffraction analysis
at 1/4' 20 scanning speedfor the
required 29-34 degrees 20 range is only 20 minutes. Although the time spent in the laboratory for fine grinding
of samples was about one to two hours, it is possible to
reduce it to about two minutes by using a small air-swept
grinding mill fed with a sample of clinker drawn from a
cement kiln by an automatic sampling device.Weighing
of the sample is not required because it is not necessary
to use any internal standard, as discussed above.
An automatic samplechanger for x-raydiffraction
purposes has alreadybeen de~eloped.~
If an automatic
sampleloadingdevicecouldbecombinedwithsuch
a
sample changer, it should be possible to complete an onlineanalysis of clinkercompounds from cementkilns
within one-half hour.
* Peak heights within each of the major sample groups, i.e., Group I, 11,
111, IV and V, which contained the same proportion of CSSand C B in a group,
have clearly reproduced themselves again and again.
Peak height ratios in standard samples using
TiOz internal standard
Table 3
Sample
No. 30.1/36.1"31.1"/36.1"33.2"/36.1"33.8"/36.1"
lay
IbY
ICY
Idy
18
20
19
18
28
28
30
27
107
92
49
182
34
54
72
20
IIay
IIby
IIcy
IIdy
26
27
28
25
23
25
28 *
23
130
103
33 *
192
34
57
81*
23
IIIay
JIIby
IIIcy
IIIdy
31
35*
31
29
17
19
17
18
119
83
42
208
34
61 *
13
21
Nay
IVby
IVcy
IVdy
32*
38
37
36
10
11
11
11
121
77
44
208
35
52
70
22
VaY
Vby
VCY
Vdy
41
43
41
39
7
7
7
7
126
72
57 *
184
37
43 *
72
21
* These values are considered abnormal and have, therefore, been
disregarded in plottingthecalibrationcurves.
Directcomparison
of peak heights in standard
samples using silicon external standard
Table 4
Sample No.
33.8"
33.2"
31.1"
30.1"
Ia
Ib
IC
Id
16
16
16
16
16
17
16
19
78 *
69 *
36
140*
31
50
74 *
23
IIa
19
21 *
18
18
14
15
98
74 *
37
loo*
30
41
54
21
IIIb
IIIC
IIId
24
22
22
22
10
12
12
12
96
54
34
1
58 *
49
57
17
IVa
9
IVb
IVC
IVd
28
27
37 *
26
12*
90
52
20 *
126
31
43
55
10*
Va
30
31
34 *
29
7
6
7
98
54
26
114
36
36 *
57
17
IIb
IIC15
IId15
IJJa
Vb
vc
7
Vd
9
10
20
* These values are considered abnormal and have, therefore, been
disregarded in plottmgthecalibrationcurves.
111
RAPID MEASUREMENT OF CLINKER COMPOSITION
Start
CheckC3S
-I--?
sample
c3s
Check if
and
C2Sdecreasrd
Check raw mix
Changed
Check
C,A
Yes
cantly
No significant
change
C,S decreased
increased
signifir
and
composition
chemical
correct
C
@
.
Analyze
another
rau'
Check
mix
composition
CaO/SiO,
for
Changed
Improper
blending,
correct Composition
of raw mix
No changc
Figure 3 Strategy for closed-loop control using quantitative
x-ray diffraction analysis.
water
Correct
physical
Check
properties of
raw mix such as
fineness, % water
Chnnged
content
and
grinding cyclc
for properfineness
1 No
Application of theanalysisto
on-line control
If the ultimate goal underlying control of raw materials
and processing variables is to produce a uniform quality
of clinker in termsof compound composition, it is obvious
that one should measure the latter and then control the
former if necessary. In a hypothetical case, it is possible
that changes in two variables may cancel each other and
may not affect the compound composition of clinker and,
therefore, should not necessitateanyaction
to control
these variables. Thus, if one were to control, for example,
burning zone temperature at a fixed value in face of raw
material composition change, this may produce a change
in clinker composition as a result of offsetting the automatic cancelling effect which may have taken place
without
the burning zone temperature control.
In any case, once one has established
a routine procedure
and specific hardware for on-line QXDA analysis of the
cement clinker at approximately one-half hour intervals,
the next question is howto utilize the analysis for on-line
kiln control. As stated earlier, a quantitative relationship
between the cement clinker composition and the controllablevariables in a cementkiln,suchas
burning zone
temperature, feed rate, moisture content of raw slurry,
etc., is not now available. However, any trend of change
in the composition of the clinker from the desired point
can be used asa qualitative indicationof possible shifts in
values of kiln variables. This indication can then be used
in conjunctionwithmathematicalmodels
of kiln perf~rmance,'l-~
as well as operator experience to arrive at an
algorithm for a control computer to print out moves or
steps to be taken by the operator to correct the cement
clinker composition.On the other hand, if a reliable model
correlating the shift in clinkercomposition to the controllable variables is arrived at using regression analysis,
112
the modelinconjunctionwith
P. K. MEHTA AND M. J. SHAH
our QXDA analysis method
change
rate feedfeed
slurryraw
No change
Check
burning
ring
Check
zone
for
temperature
i
Change
Check for variables
affecting burning
zone temperature
1Change
&
Go to main control
Checktemperature
of compensating
secondary
air changes
Change program for
to correct burning
zone temperature
Check
Change
draft
at feed end
No change
quality
No change
of fuel
Table 5
Effect of variation in oxide content on clinker
composition
Perce,tt o/ Oxide (Ignited Basis)
Raw Mix
Oxides
Cu.v
Si0
AI 2
03
FezO3
CaO
MgO
Clinker
Conlpounds
c3s
c2s
CIA
C,AF
22.2
5.8
3.1
64.9
4.0
Case I1
Case 111
23.2
5.8
3.1
63.9
4.0
22.2
6.8
2.1
64.9
4.0
Percent of Compound
Composition)
(Potential
52.0
24.5
10.0
9.5
40.5
36.0
10.0
9.5
47.0
28 .O
14.5
6.5
Case 1: Normalraw mix for Type I cement.
Case 11: Deviationinnormalraw mixcompositionintermsof
increase in SiO, content by one percent and decrease
in CaO content by one percent (note 11.5% difference
in C,S and C,S quantities).
Case 111: Deviation in normal raw. mixcompositioninterms
of
increase in A1,0, content by one percent and decrease
in Fe,O, content by one percent(note that all the
four phases are affected).
can be used for closed-loopcontrol of a cement kiln.
The QXDA method, by itself, provides a tool to obtain
such a regression model.
Without going into any elaborate detail, the illustration
in Fig. 3 gives a strategy for detecting the cause of shift in
clinker composition which can be used for kiln control. A
case study in Table 5 shows how slight variations in oxide
content of raw mix can significantly affect the composition
of clinker.
Acknowledgments
The authors are grateful to R. Van Volkenburg, Mrs. I.
Barlow and W.Traberforassistance
with the QXDA
analysis and to N. Erez for preparation of samples.
References
1. P. K. Mehta, Analytical Techniques for Improved Quality
Control in Portland Cement Plants, Pit and Quarry 57, 1,
141-145 (1964).
2. D. L. Kantro, L. E. Copeland, C. H. Weise, and S. Brunauer,
Quantitative Determination of the Major Phases in Portland Cements by X-Ray Diffraction Methods, Journ.
Portland Cement Assn. R & D Lab. (1964).
3. H. G. Smolczyk, The X-Ray Diffraction Analysis of the
Crystalline Phases of Portland Cement Clinker, ZementKalk-Gypsum, No. 12 (1961).
4. H. G. Midgley, K. E. Fletcher and A. G. Cooper, The
Identification and Determination of Alite in Portland Cement Clinker, Society of Chemical Industry, Monograph
No. 18 (1963).
5. R. L. Berger, G. J. C. Frohnsdorff, P. H. Harris and P. D.
Johnson, The Application of X-Ray Diffraction to the
Routine Mineralogical Analysis of Portland Cement,
presented at the 44th Conference of the Highway Research
Board, Washington, D. C. (1965).
6. H. P. Klug and L. E. Alexander, X-Ray Diffraction Procedures, John Wiley and Sons, Inc., 1962, Chapter 7, p. 410.
7. R. E. Stillman, Preliminary Survey of Cement Kiln Simulation and Optimization, IBM Research Note NJ-12, April 12,
1962.
8. R. E. Stillman, Cement Kiln Simulation Using Oxide
Chemistry, IBM Tech. Report 02.303, San Jose, June 30,
1964.
9. R. E. Stillman, Simulation of Cement Kiln Dynamics
Using Method of Characteristics, presented at the AZChE
56th National Meeting, San Francisco (1965).
Received June 16, 1966
113
RAPID MEASUREMENT OF CLINKER COMPOSITION
You might also like
- Gas Chromatography: Principles, Techniques, and ApplicationsFrom EverandGas Chromatography: Principles, Techniques, and ApplicationsNo ratings yet
- Finite Element Programming in Non-linear Geomechanics and Transient FlowFrom EverandFinite Element Programming in Non-linear Geomechanics and Transient FlowNo ratings yet
- Assessing Flow Response of Self-Compacting Mortar by Taguchi Method and ANOVA InteractionDocument8 pagesAssessing Flow Response of Self-Compacting Mortar by Taguchi Method and ANOVA InteractionTarek ChikerNo ratings yet
- A New On-Stream XRD Technology For Measurement and Control of Gypsum De-Hydration/Setting TimesDocument8 pagesA New On-Stream XRD Technology For Measurement and Control of Gypsum De-Hydration/Setting TimessaeedhoseiniNo ratings yet
- Microtrac Applications Notes Particle Size Measurement of Cement by Laser Diffraction Using S3500Document5 pagesMicrotrac Applications Notes Particle Size Measurement of Cement by Laser Diffraction Using S3500pas85No ratings yet
- Moses Alabi 2016 MSCE PredictiveModelforCementClinkerQualityParametersDocument18 pagesMoses Alabi 2016 MSCE PredictiveModelforCementClinkerQualityParametersaniket chakiNo ratings yet
- 1687814019896509Document13 pages1687814019896509Diego MorenoNo ratings yet
- Uniformity of SCC in Beams & ColumnsDocument20 pagesUniformity of SCC in Beams & ColumnsSyed Razi QuadriNo ratings yet
- Mix Design Using Rheology MeterDocument5 pagesMix Design Using Rheology MeterBhupesh MulikNo ratings yet
- Paper No. TRB #01-3486 Development of Testing Procedures To Determine The Water-Cement Ratio of Hardened Portland Cement Concrete Using Semi-Automated Image Analysis TechniquesDocument14 pagesPaper No. TRB #01-3486 Development of Testing Procedures To Determine The Water-Cement Ratio of Hardened Portland Cement Concrete Using Semi-Automated Image Analysis TechniquesGiora RozmarinNo ratings yet
- Research Results Digest: National Cooperative Highway Research ProgramDocument24 pagesResearch Results Digest: National Cooperative Highway Research ProgramsaeedhoseiniNo ratings yet
- Artigo InteressanteDocument20 pagesArtigo InteressanteAndré SousaNo ratings yet
- Applied Mixture Optimization Techniques For Paste Design of Bonded Roller-Compacted Fibre Reinforced Polymer Modified Concrete (BRCFRPMC) OverlaysDocument20 pagesApplied Mixture Optimization Techniques For Paste Design of Bonded Roller-Compacted Fibre Reinforced Polymer Modified Concrete (BRCFRPMC) OverlaysTarek ChikerNo ratings yet
- Kiln Control Using Continuous On-Stream XRD: A Proven TechnologyDocument8 pagesKiln Control Using Continuous On-Stream XRD: A Proven TechnologySiti Hanna100% (1)
- Report For Concrete Labs - CE 3111Document24 pagesReport For Concrete Labs - CE 3111MuhammadZAmjadNo ratings yet
- Reproducible mini-slump test procedure for measuring the yield stress of cementitious pastesDocument12 pagesReproducible mini-slump test procedure for measuring the yield stress of cementitious pastesrobertacivilNo ratings yet
- Modeling The Equilibrium Compressed Void Volume of Carbon BlackDocument30 pagesModeling The Equilibrium Compressed Void Volume of Carbon Blackkml786No ratings yet
- Production of High Strength Concrete in SudanDocument8 pagesProduction of High Strength Concrete in SudanInternational Organization of Scientific Research (IOSR)No ratings yet
- Development and Properties of Self Compacting Concrete Mixed With Fly AshDocument4 pagesDevelopment and Properties of Self Compacting Concrete Mixed With Fly AshHans Hans SadzNo ratings yet
- Extended Abstract Ezio LeoneDocument10 pagesExtended Abstract Ezio LeonececiliabiancardiNo ratings yet
- IJREISS_4111_59674Document6 pagesIJREISS_4111_59674anuragchandNo ratings yet
- Quantitative X-ray Diffraction Analysis of Cement Clinker SamplesDocument6 pagesQuantitative X-ray Diffraction Analysis of Cement Clinker SamplesyinglvNo ratings yet
- Void ContentDocument6 pagesVoid ContentAnilkumar MNo ratings yet
- 226 PDFDocument8 pages226 PDFBatepola BacNo ratings yet
- Study On Use of Bottom Ash and Silica Fume in Concrete: Submitted by T.SujathaDocument10 pagesStudy On Use of Bottom Ash and Silica Fume in Concrete: Submitted by T.SujathaGeethavaniTalupulaNo ratings yet
- Development of A Phase Diagram To Control Composite: Manufacturing Using Raman SpectrosDocument11 pagesDevelopment of A Phase Diagram To Control Composite: Manufacturing Using Raman SpectrosAkhil ModiNo ratings yet
- Proportioning Concrete Mix DesignDocument25 pagesProportioning Concrete Mix DesignGrachelle Andrea CapiralNo ratings yet
- Layer Dependent RLPD CoefficientsDocument33 pagesLayer Dependent RLPD CoefficientsMohamed ElgendyNo ratings yet
- Beta LactamDocument10 pagesBeta LactamKamal KishoreNo ratings yet
- JournalReview - Group No. 3 - Ikan, Jervis IvanDocument12 pagesJournalReview - Group No. 3 - Ikan, Jervis IvanCarlo King SantosNo ratings yet
- CFD of Styrene ReactorDocument9 pagesCFD of Styrene Reactorgharibi.ali98486No ratings yet
- ABS Capillary RheometerDocument17 pagesABS Capillary RheometersharontanasNo ratings yet
- W-Co DensificationDocument7 pagesW-Co DensificationPranith KumarNo ratings yet
- Modeling and Control of A Riser Type Fluid Catalytic Cracking (FCC)Document12 pagesModeling and Control of A Riser Type Fluid Catalytic Cracking (FCC)krg09100% (1)
- EasyChair Preprint 6653Document9 pagesEasyChair Preprint 6653Diego MorenoNo ratings yet
- Matecconf cmss2018 02086Document6 pagesMatecconf cmss2018 02086IvanRomanovićNo ratings yet
- A Study On Properties of Recycled Coarse Aggregate and It's ConcreteDocument11 pagesA Study On Properties of Recycled Coarse Aggregate and It's ConcreteJenish PatelNo ratings yet
- CFD Simulation and Optimization of a Cement Plant Preheater Cyclone PerformanceDocument55 pagesCFD Simulation and Optimization of a Cement Plant Preheater Cyclone Performancealfapetra meiskyNo ratings yet
- Is.13311.2.1992 Rebound HammerDocument12 pagesIs.13311.2.1992 Rebound HammerChockalingamNo ratings yet
- CE152p-2-e02 Experiment No.3Document9 pagesCE152p-2-e02 Experiment No.3jeraldramores166No ratings yet
- Application of hard grade binders for high modulus asphalt mixturesDocument8 pagesApplication of hard grade binders for high modulus asphalt mixturesAnonymous x7VY8VF7No ratings yet
- Comparative Field and Lab Tests on Using SCMs to Control ASR in ConcreteDocument10 pagesComparative Field and Lab Tests on Using SCMs to Control ASR in ConcreteMichael Stanley Diaz GuevaraNo ratings yet
- Anempirical Approach Tothe Design Oftrommel Screens Forfine Screening Ofdomestic RefuseDocument13 pagesAnempirical Approach Tothe Design Oftrommel Screens Forfine Screening Ofdomestic RefuseNcls BMNo ratings yet
- Astm - CBRDocument9 pagesAstm - CBRGuillermo Garcia SerafinNo ratings yet
- Hasanina 2017Document10 pagesHasanina 2017Chiew Fei HaNo ratings yet
- A Mix Design Procedure For Self Compacti PDFDocument6 pagesA Mix Design Procedure For Self Compacti PDFمصطفى شعبانNo ratings yet
- Marshall Stability Test (Ini Machiem)Document7 pagesMarshall Stability Test (Ini Machiem)Azrol Azmir Long100% (1)
- Schroefl 2017Document19 pagesSchroefl 2017MOHAMED DawiNo ratings yet
- Laboratory Exercise No3Document4 pagesLaboratory Exercise No3JorgieNo ratings yet
- IPTC 14008 Inclusions of Well-Test Interpretation and Sector Model Simulation Into A Full Field Model: An Integration Modeling ApproachDocument11 pagesIPTC 14008 Inclusions of Well-Test Interpretation and Sector Model Simulation Into A Full Field Model: An Integration Modeling Approachamramazon88No ratings yet
- Is 13311 - 2Document9 pagesIs 13311 - 2Sravan KumarNo ratings yet
- Mortar Bar Expansion Test: InterimDocument7 pagesMortar Bar Expansion Test: InterimbilalNo ratings yet
- Application of Laboratory Asphalt Cracking Tests To Cold in Place Recycled MixturesDocument20 pagesApplication of Laboratory Asphalt Cracking Tests To Cold in Place Recycled MixturesharNo ratings yet
- Combining Experiments, Polyumod®, Kornucopia®, and Abaqus® To Create Accurate Fe Scratch SimulationsDocument34 pagesCombining Experiments, Polyumod®, Kornucopia®, and Abaqus® To Create Accurate Fe Scratch SimulationsNagaraj RamachandrappaNo ratings yet
- Fabricacion Del Cemento-YesoDocument11 pagesFabricacion Del Cemento-YesoJosé Alberto BarretoNo ratings yet
- Modern Practices in UCGDocument4 pagesModern Practices in UCGVăn Đại - BKHNNo ratings yet
- Estimating The Strength of Concrete Using Surface Rebound Value and Design Parameters of Concrete MaterialDocument9 pagesEstimating The Strength of Concrete Using Surface Rebound Value and Design Parameters of Concrete Materialshaikh85No ratings yet
- 1 s2.0 S0950061818307645 MainDocument15 pages1 s2.0 S0950061818307645 MainZONA LAKINo ratings yet
- Effect of Nano-Silica On Properties of Blended Cement: G.Reddy BabuDocument6 pagesEffect of Nano-Silica On Properties of Blended Cement: G.Reddy BabuInternational Journal of computational Engineering research (IJCER)No ratings yet
- LTRC Capsule 10-6B Implementation of GPC Characterization of Asphalt Binders at Louisiana Materials LaboratoryDocument2 pagesLTRC Capsule 10-6B Implementation of GPC Characterization of Asphalt Binders at Louisiana Materials LaboratorywalaywanNo ratings yet
- Unstabilized and Stabilized Compressed Earth Blocks With Partial Incorporation of Recycled AggregatesDocument17 pagesUnstabilized and Stabilized Compressed Earth Blocks With Partial Incorporation of Recycled AggregatesqamhNo ratings yet
- Fire Resistance of Walls Made of Soil-Cement and Kraftterra Compressed Earth BlocksDocument16 pagesFire Resistance of Walls Made of Soil-Cement and Kraftterra Compressed Earth BlocksqamhNo ratings yet
- Water Absorption Characteristic of Interlocking Compressed Earth Brick UnitsDocument7 pagesWater Absorption Characteristic of Interlocking Compressed Earth Brick UnitsqamhNo ratings yet
- Most common verbs in Spanish, English and PortugueseDocument3 pagesMost common verbs in Spanish, English and PortugueseSilvia AlexandrinoNo ratings yet
- (Mesoporous Biomaterials) Rice Husk and Rice Husk Ash Reutilization Intonanoporous Materials For Adsorptive Biomedicalapplications A ReviewDocument12 pages(Mesoporous Biomaterials) Rice Husk and Rice Husk Ash Reutilization Intonanoporous Materials For Adsorptive Biomedicalapplications A ReviewqamhNo ratings yet
- Use Vaseline To Cover The Surface of Alkali Activated Cement PasteDocument2 pagesUse Vaseline To Cover The Surface of Alkali Activated Cement PasteqamhNo ratings yet
- BlocksDocument3 pagesBlocksqamhNo ratings yet
- Design of Hybrid Powered Automated Compressed Stabilized Earth Block (CSEB) MachineDocument6 pagesDesign of Hybrid Powered Automated Compressed Stabilized Earth Block (CSEB) MachineqamhNo ratings yet
- Floculantes de AlmidonDocument81 pagesFloculantes de AlmidonqamhNo ratings yet
- AN EMRES102 Porous Ceramics ENDocument4 pagesAN EMRES102 Porous Ceramics ENqamhNo ratings yet
- Environmental Implications of GeopolymersDocument21 pagesEnvironmental Implications of GeopolymersqamhNo ratings yet
- Ejercicios Verbos Presente y PasadoDocument36 pagesEjercicios Verbos Presente y PasadoqamhNo ratings yet
- Art - Estab Con Ceniza de Caña de Azucar PDFDocument9 pagesArt - Estab Con Ceniza de Caña de Azucar PDFqamhNo ratings yet
- Triple Cone Stove for Burning Rice Hulls and WoodsmokeDocument2 pagesTriple Cone Stove for Burning Rice Hulls and WoodsmokeqamhNo ratings yet
- Colorado Cultural Resource Survey FormDocument45 pagesColorado Cultural Resource Survey FormqamhNo ratings yet
- Construction and Building MaterialsDocument1 pageConstruction and Building MaterialsqamhNo ratings yet
- Characteristics of Volcanic Ash and Natural Lime Based Stabilized Clayey SoilsDocument1 pageCharacteristics of Volcanic Ash and Natural Lime Based Stabilized Clayey SoilsqamhNo ratings yet
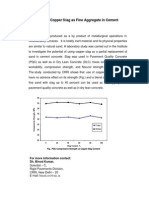
- Utilization of Copper Slag in Concrete - Strength UnaffectedDocument1 pageUtilization of Copper Slag in Concrete - Strength UnaffectedqamhNo ratings yet
- Use of Slag in Road MakingDocument3 pagesUse of Slag in Road MakingRahul PandeyNo ratings yet
- CONDORMAN SuperheroDocument1 pageCONDORMAN SuperheroqamhNo ratings yet
- Modeling Using FEA: FEA Uses A Complex System of Points Called Which Make A Grid Called ADocument8 pagesModeling Using FEA: FEA Uses A Complex System of Points Called Which Make A Grid Called AqamhNo ratings yet
- National Educators' Workshop: Update 93: Sfandard Experiments in Engineering Materials Science and TechnologyDocument560 pagesNational Educators' Workshop: Update 93: Sfandard Experiments in Engineering Materials Science and Technologyqamh0% (1)
- Efecto de Los Residuos de Flotación de Cobre en El CementoDocument7 pagesEfecto de Los Residuos de Flotación de Cobre en El CementoqamhNo ratings yet
- Stabilized Soils Using PNG ResourcesDocument2 pagesStabilized Soils Using PNG ResourcesqamhNo ratings yet
- Paper Clip ExperimentDocument5 pagesPaper Clip ExperimentqamhNo ratings yet
- Principles and Objectives of Oven Design OptimizationDocument13 pagesPrinciples and Objectives of Oven Design OptimizationqamhNo ratings yet
- Dimensional Analysis Is A ConceptualDocument3 pagesDimensional Analysis Is A ConceptualqamhNo ratings yet
- Reactivity of IronDocument3 pagesReactivity of IronqamhNo ratings yet
- Monte CarloDocument9 pagesMonte CarloqamhNo ratings yet
- Production Process Flow DiagramDocument4 pagesProduction Process Flow DiagramSuntech Engineering CorporationNo ratings yet
- Acs Langmuir 7b01682Document10 pagesAcs Langmuir 7b01682harry mahfuzanNo ratings yet
- ElangovanDocument23 pagesElangovanElangoNo ratings yet
- Polymer Blends and CompositesDocument25 pagesPolymer Blends and CompositesHamza ChelaliNo ratings yet
- Lecture 2a: Determination of The Concentration and The Acid Dissociation Constants of An Unknown Amino Acid (Part II)Document7 pagesLecture 2a: Determination of The Concentration and The Acid Dissociation Constants of An Unknown Amino Acid (Part II)Steve LiNo ratings yet
- Diy Home RepairDocument560 pagesDiy Home RepairRohan100% (1)
- Review Article: Greener Approach Towards Corrosion InhibitionDocument11 pagesReview Article: Greener Approach Towards Corrosion InhibitionErraFatihaNo ratings yet
- 334 Grossly Polluted Industries - West Bengal Pollution Control BoardDocument16 pages334 Grossly Polluted Industries - West Bengal Pollution Control BoardSagnik DasNo ratings yet
- Determination of Conductivity Exp3 - PDFDocument12 pagesDetermination of Conductivity Exp3 - PDFSusheel TalrejaNo ratings yet
- Din 1623-05Document11 pagesDin 1623-05dietersimaNo ratings yet
- Wood Connectors - Houtverbindingen - 2013 - LRDocument168 pagesWood Connectors - Houtverbindingen - 2013 - LRjanm2008No ratings yet
- Glass ContainerDocument1 pageGlass Containerjes pristoNo ratings yet
- FLOW 3D General BrochureDocument12 pagesFLOW 3D General BrochurehhadNo ratings yet
- WCP, SPDocument2 pagesWCP, SPAnil Kumar Sabu100% (1)
- Paints & CoatsDocument29 pagesPaints & CoatsMaitreyi YellapragadaNo ratings yet
- List of Is CodesDocument12 pagesList of Is CodesSanjeev DhariwalNo ratings yet
- Fractional Distillation of An Ethanol - Water MixtureDocument7 pagesFractional Distillation of An Ethanol - Water Mixturekrupa_patel_8784% (25)
- MSDS XW15Document8 pagesMSDS XW15mk7718No ratings yet
- Chapter 4 Food Sanitation SafetyDocument24 pagesChapter 4 Food Sanitation SafetySamille Joy SanchezNo ratings yet
- SAE 660 Bronze - Technical InformationDocument2 pagesSAE 660 Bronze - Technical Informationtanha56313955No ratings yet
- Microbiology A Laboratory Manual 10th Edition Cappuccino Solutions ManualDocument3 pagesMicrobiology A Laboratory Manual 10th Edition Cappuccino Solutions Manualzeldaguinevereiur100% (28)
- Marine Sandwich Composites for High PerformanceDocument12 pagesMarine Sandwich Composites for High Performancecarlos ivan carvajal ortizNo ratings yet
- Sill Plate/Nailer Connection Design Based On NDS 2018Document18 pagesSill Plate/Nailer Connection Design Based On NDS 2018NameNo ratings yet
- Iron Ore Deposits M S KrishnanDocument8 pagesIron Ore Deposits M S KrishnandeshmukhgeolNo ratings yet
- Materials for MEMs and MicrosystemsDocument12 pagesMaterials for MEMs and MicrosystemsiangarvinsNo ratings yet
- Cemfil 62 - 3Document2 pagesCemfil 62 - 3Kesavan DhuraiNo ratings yet
- Chemical Purification Methods ListDocument8 pagesChemical Purification Methods ListMd.Mehdi MasudNo ratings yet
- Design Example ASD GirderDocument12 pagesDesign Example ASD GirderEdward van MartinoNo ratings yet
- Equivalent Grades of Gray Iron, Ductile Iron, SG Iron, Nodular Graphite IronDocument2 pagesEquivalent Grades of Gray Iron, Ductile Iron, SG Iron, Nodular Graphite IronNagarjun ReddyNo ratings yet
- Aluminium ConductorsDocument2 pagesAluminium ConductorsNgô Tiến PhúNo ratings yet