Professional Documents
Culture Documents
7 - Polar Reactions PDF
Uploaded by
vkrOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
7 - Polar Reactions PDF
Uploaded by
vkrCopyright:
Available Formats
Engineering Chemistry III
Prof. Santosh Gharpure
Polar Reactions
Important points to be discussed in this module:
1. Chemistry of Carbocations with respect to:
a. 1,2-shifts of carbocations
b. Pinacol-pinacolone rearrangement
c. Semipinacolone rearrangement
2. Chemistry of Carbanions:
a. Alkylation and acylation of enolates
b. Aldol condensation
c. Michael reaction
d. Claisen and Dieckman condensation reactions
Chemistry of Carbocations
During the Friedel-Crafts alkylation, we saw that n-propyl cation (CH3CH2CH2+)
undergoes rearrangement to give rise to a more stable secondary carbocation
(CH3+CH2CH3). The driving force for this reaction is generation of a more stable
carbocation it is estimated that approximately 16 kcal/mol (67 kJ/mol) is gained by
conversion of a primary to a secondary or a secondary to tertiary carbocation. These
types of rearrangements are broadly classified as Wagner-Meerwein rearrangements and
involve 1,2-shift of an adjacent group. These types of reactions are common to all the
reactions which involve carbocation intermediate and the fate of the carbocation
intermediate is dependent of the reaction conditions. The reaction of neopentylic alcohol
with a mineral acid such as HCl can nicely illustrate this point. In this reaction,
depending on the exact reaction conditions, varying amounts 2-methyl-2-butene and 2chloro-2-methylbutane are formed and neopentyl chloride is conspicuous by its absence!
CH3 OH
H3 C
CH3 OH2
HCl
CH3
-H 2O
H 3C
H3 C
CH3
CH3
neopentyl
alcohol
1,2-shif t
H3C
CH 3
H 3C
-H+
CH3
CH3
CH3
2-methyl-2-butene
+Cl-
H 3C
Cl
CH3
CH 3
2-chloro-2-methylbutane
1
Indian Institute of Technology Madras
CH 2
CH3
Engineering Chemistry III
Prof. Santosh Gharpure
What happens in this transformation then? The answer to this question could be obtained
by writing the mechanism for this transformation. In the first step, the HCl protonates the
hydroxyl group of the neopentyl alcohol which leads to dehydration and subsequent
formation of neopentylic carboaction. This intermediate is extremely short lived and
rearranges via the migration of a methyl group along with its pair of bonding electrons to
generate a more stable tertiary carbocation. This process of intramolecular migration is
considerably faster than the intermolecular trapping of the carbocation with solvent or
chloride nucleophile. The migration of a group from one carbon to an adjacent
carbocation is commonly encountered when the reactive intermediate involved is
carbocations and is commonly referred to as 1,2-shift.
Pinacol Rearrangement:
1,2-diols, known as pinacols, also undergo a similar type of dehydration reaction when
treated with acid to furnish a ketone or aldehyde which have rearranged structure
compared to the initial diol. This transformation is commonly referred to as Pinacol
Rearrangement. Lets take an example when pinacol reacts with an acid, it leads to the
formation of pincolone. It is believed that this reaction also involves the formation of a
tertiary carbocation in the first step which is followed by 1,2-shift of the methyl group.
Now, the question will be if we already have a stable carbocation, why should it undergo
rearrangement? The answer is - the oxygen lone pair - which can stabilize the newly
formed carbocation by donating its lone pair to carbocation, thereby completing octet for
all the atoms involved. In fact, one could view this reaction as a push-pull reaction! The
carbocation pulls the alkyl group along with its bonding electrons towards itself while
the oxygen lone pair pushes the alkyl group away.
HO OH
H3C
CH3
H3C CH3
H+
HO OH2
H3C
CH3
H3C CH3
-H2O
OH
H3C
CH3
H3C CH3
pinacol
O
H3C
CH3
CH3
CH3
-H+
HO
H3C
CH3
CH3
CH3
pinacolone
2
Indian Institute of Technology Madras
HO CH3
H3C
CH3
CH3
Engineering Chemistry III
Prof. Santosh Gharpure
It is clear now that there are two important steps involved in pinacol rearrangement: (1)
loss of water from the protonated diol to form the carbocation; and (2) rearrangement of
the carbocation by a 1,2-shift to give rise to the protonated ketone.
OMe
HO OH
MeO
OMe
Ph Ph
H+
O
OMe
Ph
Ph
The next question that arises is what happens when the pinacol is unsymmetrical? In such
situations, the product obtained is determined (a) by which OH group is lost in step (1)
and then (b) by which group migrates in step (2) to the electron deficient carbon thus
formed. The OH group is usually lost from the carbon atom that will form the more
stable carbocation. The rate of migration of the groups in the second step is referred to as
the migratory aptitude of the group. Since the rearrangement involves movement of the
migrating group with its bonding electrons to an electron deficient centre, migratory
aptitudes are greatest for groups in which the migrating atom is most electron rich. It
should be kept in mind though that the direction of the rearrangement is largely
determined by the relative ease of removal of a OH group (first step) and rearrangement
may not necessarily involve the group of highest migratory aptitude.
HO OH
Et
H
H3C H
H+
HO OH
Ph
CH3
H H
H+
Et
CHO
H3C
H
Ph
H3C
A general order of migratory aptitudes for the pinacol rearrangements is:
p-anisyl > p-tolyl > phenyl > tert.-alkyl > primary alkyl > H
It should be noted however, that even though the migratory aptitudes generally follow the
order given above, the absolute comparison depends on the actual reaction and condition.
Semipinacol rearrangement:
The pinacol type of rearrangement is not limited only to vicinal diols. Deamination of aamino alcohols also leads to a reaction which closely resembles the pinacol
3
Indian Institute of Technology Madras
Engineering Chemistry III
Prof. Santosh Gharpure
rearrangement and is often called the semipinacol rearrangement. In this reaction, nitrous
acid is used to form an alkanediazonium ion which readily looses nitrogen to give the
intermediate carbocation.
Ph
HO NH2
CH3
Ph H
O
Ph
HO N2
HNO2
Ph
OH
-N2
Ph
CH3
Ph H
Ph H
CH3
H
Ph
-H+
CH3
H
Ph
HO
Ph
CH3
Ph
HO CH3
H
Ph
This reaction can be used for expansion as well as the contraction of medium sized rings.
NH2
HNO2
CHO
N2
N2
OH
NH2
HNO2
OH
4
Indian Institute of Technology Madras
Engineering Chemistry III
Prof. Santosh Gharpure
Assignment
A.
Suggest a suitable mechanism for the following reactions. Write each step and
show the rearrangement clearly by means of arrow.
1.
Cl
OH
HCl
Me
Me
2.
OH
Me
H+
Me
Me
Me
3.
OH
H+
OH
4.
Me
Me
H+
OH
OH
CHO
Me
Me
5.
OH Me
Me
OH
H+
Me
Me
O
6.
OH
HBr
Br
5
Indian Institute of Technology Madras
Me
+
Me
O
Engineering Chemistry III
B.
Prof. Santosh Gharpure
Identify the products of the following reactions and suggest a mechanism for the
reaction.
1.
OMe
H+
Ph
MeO
Ph
HO OH
2.
OH
H+
HO
3.
Ph Ph
Me
H+
Me
HO OH
4.
Ph
Me
HNO 2
CH 2NH2
HO
5.
Ph Me
Ph
HNO 2
Me
HO NH 2
6.
OMe
HNO 2
Ph
MeO
Ph
HO NH 2
6
Indian Institute of Technology Madras
You might also like
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (122)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (589)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (401)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (842)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (897)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5806)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (345)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1091)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- Estimation & CostingDocument31 pagesEstimation & CostingKrztofer PrnzNo ratings yet
- Cape Chemistry Unit 1Document22 pagesCape Chemistry Unit 1Audi Sweetangel100% (1)
- RRKM TheoryDocument4 pagesRRKM TheoryjainankitmumNo ratings yet
- NSC-130 Stoichiometry Worksheet Answers PDFDocument2 pagesNSC-130 Stoichiometry Worksheet Answers PDFTristan Tabago ConsolacionNo ratings yet
- Construction Project Management Handbook PDFDocument149 pagesConstruction Project Management Handbook PDFLuis ItoNo ratings yet
- Reactors in ParallelDocument3 pagesReactors in ParallelAdrián D. Rodríguez100% (2)
- Date C Org UploadDocument2 pagesDate C Org UploadKrztofer PrnzNo ratings yet
- Casual Leave Application FormDocument2 pagesCasual Leave Application FormKrztofer PrnzNo ratings yet
- Dosing Pump LefletDocument2 pagesDosing Pump LefletKrztofer PrnzNo ratings yet
- Ce2201 QBDocument20 pagesCe2201 QBKrztofer PrnzNo ratings yet
- Strength of MaterialDocument12 pagesStrength of MaterialKrztofer PrnzNo ratings yet
- Learn Vasthu Sasthram Free-Vasthu Sastra in Tamil PDF - Vasthu Tips - About Vastu - What Is Vastu Shastra - House Vastu - Hindu Vastu-Vastu HomesDocument8 pagesLearn Vasthu Sasthram Free-Vasthu Sastra in Tamil PDF - Vasthu Tips - About Vastu - What Is Vastu Shastra - House Vastu - Hindu Vastu-Vastu HomesKrztofer Prnz0% (1)
- Gis For AgricultureDocument32 pagesGis For Agricultureportatil31164No ratings yet
- WATER-" The Source of Life": All The Water That Will Ever Be Is, RightDocument21 pagesWATER-" The Source of Life": All The Water That Will Ever Be Is, RightKrztofer PrnzNo ratings yet
- Exp 20 Formal Lab ReportDocument7 pagesExp 20 Formal Lab Reportapi-592105594No ratings yet
- 9 Edition, Global Edition L. G. Wade, JR.: Organic ChemistryDocument60 pages9 Edition, Global Edition L. G. Wade, JR.: Organic ChemistryRoma Blaise G FloresNo ratings yet
- Enzyme Kinetics Report FormatDocument5 pagesEnzyme Kinetics Report FormatOnrcn BgntsNo ratings yet
- Biochemistry PRC 2 Data Analysis EnzymeDocument4 pagesBiochemistry PRC 2 Data Analysis EnzymeSARAH SAFIAH TAJUL ARIFFINNo ratings yet
- Bhayankar SawaalDocument6 pagesBhayankar SawaalNidhi SisodiaNo ratings yet
- HW 1Document1 pageHW 1rahulsaxenaNo ratings yet
- Tetrahedron Letters: Lydie Harmand, Pavel Drabina, Vladimír Pejchal, Lenka Husáková, Miloš SedlákDocument4 pagesTetrahedron Letters: Lydie Harmand, Pavel Drabina, Vladimír Pejchal, Lenka Husáková, Miloš Sedlák'Licenza AdagioNo ratings yet
- CHGV 101 Tutorial 3 Questions KineticsDocument4 pagesCHGV 101 Tutorial 3 Questions KineticsOvayo TyalaNo ratings yet
- Enzyme Station Lab 1Document21 pagesEnzyme Station Lab 1api-233187566No ratings yet
- Organic Reaction MechanismsDocument14 pagesOrganic Reaction MechanismstylerNo ratings yet
- Chapter 13-ChemicalKineticsDocument4 pagesChapter 13-ChemicalKineticsKhurram KhanNo ratings yet
- 7 Baylis-Hillman PDFDocument25 pages7 Baylis-Hillman PDFWilly YantoNo ratings yet
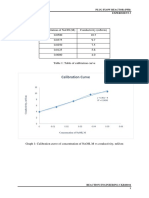
- Calibration Curve: Concentration of Naoh (M) Conductivity (MS/CM) 0.0500 10.7 0.0375 9.7 0.0250 7.5 0.0125 5.6 0.0000 4.0Document7 pagesCalibration Curve: Concentration of Naoh (M) Conductivity (MS/CM) 0.0500 10.7 0.0375 9.7 0.0250 7.5 0.0125 5.6 0.0000 4.0Kholidi ChooNo ratings yet
- Chemical Kinetics CBSE Class XII NotesDocument5 pagesChemical Kinetics CBSE Class XII NotesRahul SharmaNo ratings yet
- L3 Nucleophilic AdditionDocument13 pagesL3 Nucleophilic AdditionCheng FuNo ratings yet
- Chemical Kinetics: Class 9.2Document4 pagesChemical Kinetics: Class 9.2Voora GowthamNo ratings yet
- Chapter 5a - Alkyl Halides (Substitution Reaction) PDFDocument13 pagesChapter 5a - Alkyl Halides (Substitution Reaction) PDFsachinNo ratings yet
- On The Mechanism of The Baeyer-Drewsen Synthesis of Indigo: February 2016Document6 pagesOn The Mechanism of The Baeyer-Drewsen Synthesis of Indigo: February 2016SamNo ratings yet
- OrchemDocument75 pagesOrchemGregorius EnrikoNo ratings yet
- Perception Test 4 Organic Chem Sem 2Document5 pagesPerception Test 4 Organic Chem Sem 2gypsydavesbrotherNo ratings yet
- KV Chan Set 4 QPDocument6 pagesKV Chan Set 4 QPdeepaNo ratings yet
- Flashcards - Topic 11 Kinetics - Edexcel IAL Chemistry A-LevelDocument69 pagesFlashcards - Topic 11 Kinetics - Edexcel IAL Chemistry A-LevelzuveriaNo ratings yet
- 1L SelectivityDocument13 pages1L SelectivityCarlos Javier Orellana OrtizNo ratings yet
- Chemical KineticsDocument19 pagesChemical KineticsUttam SinghNo ratings yet
- Lecture 11 Hydrogenation and Its ThermodynamicsDocument28 pagesLecture 11 Hydrogenation and Its ThermodynamicsHuraira AbidNo ratings yet
- (This Question Paper Consists of Seven (7) Printed Pages Including This Page)Document7 pages(This Question Paper Consists of Seven (7) Printed Pages Including This Page)aizaNo ratings yet