Professional Documents
Culture Documents
Oncogenic Osteomalacia - A Complex Dance of Factors: Thomas O. Carpenter, M.D
Uploaded by
Asticha Erlianing SariOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Oncogenic Osteomalacia - A Complex Dance of Factors: Thomas O. Carpenter, M.D
Uploaded by
Asticha Erlianing SariCopyright:
Available Formats
editorials
of regeneration in myocardium, treatment through
the induction of regeneration may prove feasible,
especially in patients with postinfectious and toxic
cardiomyopathy.
From the Department of Pediatrics, Vanderbilt University School
of Medicine, Nashville (A.S.); and the Department of Cardiology,
Childrens Hospital, Harvard Medical School, Boston (J.E.L.).
1. Report of the New England Regional Infant Cardiac Program.
Pediatrics 1980;65:375-461.
2. Boneva RS, Botto LD, Moore CA, Yang Q, Correa A, Erickson JD.
Mortality associated with congenital heart defects in the United
States: trends and racial disparities, 1979-1997. Circulation 2001;
103:2376-81.
3. Olson EN, Srivastava D. Molecular pathways controlling heart
development. Science 1996;272:671-6.
4. Kumar K, Thatai D, Saxena A, et al. Pediatric dilated cardiomyopathy: prognosis in a developing nation is comparable to developed nations. Int J Cardiol (in press).
5. Lipshultz SE. Ventricular dysfunction clinical research in infants,
children and adolescents. Prog Pediatr Cardiol 2000;12:1-28.
6. Bilgic A, Ozbarlas N, Ozkutlu S, Ozer S, Ozme S. Cardiomyopathies in children: clinical, epidemiological and prognostic evaluation. Jpn Heart J 1990;31:789-97.
7. Nugent AW, Daubeney PEF, Chondros P, et al. The epidemiology
of childhood cardiomyopathy in Australia. N Engl J Med 2003;348:
1639-46.
8. Lipshultz SE, Sleeper LA, Towbin JA, et al. The incidence of
pediatric cardiomyopathy in two regions of the United States. N Engl
J Med 2003;348:1647-55.
9. Arola A, Jokinen E, Ruuskanen O, et al. Epidemiology of idiopathic cardiomyopathies in children and adolescents: a nationwide
study in Finland. Am J Epidemiol 1997;146:385-93.
10. Michels VV, Moll PP, Miller FA, et al. The frequency of familial
dilated cardiomyopathy in a series of patients with idiopathic dilated
cardiomyopathy. N Engl J Med 1992;326:77-82.
11. Kelly DP, Strauss AW. Inherited cardiomyopathies. N Engl J Med
1994;330:913-9.
12. Schmitt JP, Kamisago M, Asahi M, et al. Dilated cardiomyopathy
and heart failure caused by a mutation in phospholamban. Science
2003;299:1410-3.
13. Arad M, Benson DW, Perez-Atayde AR, et al. Constitutively
active AMP kinase mutations cause glycogen storage disease mimicking hypertrophic cardiomyopathy. J Clin Invest 2002;109:357-62.
14. Bennett MJ, Rinaldo P, Strauss AW. Inborn errors of mitochondrial fatty acid oxidation. Crit Rev Clin Lab Sci 2000;37:1-44.
15. Strauss AW. Defects of mitochondrial proteins and pediatric
heart disease. Prog Pediatr Cardiol 1996;6:83-90.
16. Poss KD, Wilson LG, Keating MT. Heart regeneration in zebrafish. Science 2002;298:2188-90.
Copyright 2003 Massachusetts Medical Society.
Oncogenic Osteomalacia A Complex Dance of Factors
Thomas O. Carpenter, M.D.
Oncogenic osteomalacia has fascinated physiology-minded physicians for decades. The traditional
name for this peculiar disorder connotes its classification as a paraneoplastic phenomenon. Such a
characterization is a bit off the mark, however, in
that the involved neoplasm is often (but not always) of limited clinical significance apart from its
causal role in the musculoskeletal disease. Tumors
responsible for oncogenic osteomalacia are usually
benign rather than invasive, whereas generalized,
debilitating osteomalacia and rickets are the important clinical problems for the patient. The assay
for the measurement of circulating levels of fibroblast growth factor 23 (FGF-23), the development
of which is described by Jonsson et al.1 in this issue
of the Journal, may prove to be useful in the investigation and management of oncogenic osteomalacia.
clinical presentation
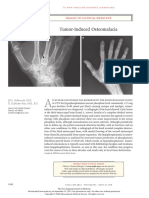
The clinical presentation of oncogenic osteomalacia is reminiscent of that of the more common
disorder X-linked hypophosphatemia,2 which has
been studied intensively and serves as the prototypic hypophosphatemic disorder. Oncogenic osteo-
n engl j med 348;17
malacia, like X-linked hypophosphatemia, is manifested by decreased mineralization of newly formed
bone and the clinical findings of osteomalacia. In
growing children, rachitic deformities of the growth
plates occur. A useful clinical distinction between
the two disorders is the patients age at the onset of
the disease: oncogenic osteomalacia is an acquired
phenotype, whereas X-linked hypophosphatemia
tends to become evident during the second year of
life. There are exceptions to this generalization: a
later onset of X-linked and autosomal dominant hypophosphatemic rickets does occur. Patients with
oncogenic osteomalacia frequently present with
fractures and more severe bone pain than that which
occurs in X-linked hypophosphatemia and often
report muscle weakness an unusual symptom
in patients with X-linked hypophosphatemia.
The characteristic hypophosphatemia results
from an excessive renal loss of phosphate. The serum calcium level is usually normal, but mild hypocalcemia has been described. Elevations of serum
alkaline phosphatase activity are typical, and the severity of this abnormality can exceed that seen in
X-linked hypophosphatemia. Serum levels of parathyroid hormone have been variably reported as low
www.nejm.org
april 24, 2003
The New England Journal of Medicine
Downloaded from nejm.org on September 25, 2013. For personal use only. No other uses without permission.
Copyright 2003 Massachusetts Medical Society. All rights reserved.
1705
The
new england journal
and as elevated but are most frequently normal.
Low or normal circulating levels of 1,25-dihydroxyvitamin D are observed, despite ambient hypophosphatemia, a major physiological stimulus for
1,25-dihydroxyvitamin D production; 1,25-dihydroxyvitamin D levels in oncogenic osteomalacia are
often lower than those in patients with X-linked hypophosphatemia. Thus, the typical clinical and biochemical phenotype is similar to that of X-linked
hypophosphatemia, but the severity of the abnormality is often greater in oncogenic osteomalacia.
evaluation
When these clinical features occur in an older child
or an adult, particularly in the absence of any relevant family history of disease, oncogenic osteomalacia should be suspected. A careful search for a tumor is an important step in the evaluation of such
patients. A substantial number of deaths can be
avoided by removal of the causative tumor, and in
some cases, an occult cancer may be identified. Tumors are often quite small and not detectable on
physical examination or routine radiography. There
appears to be a propensity for these tumors to arise
in the head and neck, and detailed computed tomography or magnetic resonance imaging of the
sinuses and jaw areas is suggested. The occurrence
of tumors at skeletal sites is not uncommon in patients with oncogenic osteomalacia. Successful localization of causative tumors with the use of indium-111 pentetreotide or octreotide scintigraphy
has been demonstrated.3,4 Technetium-99 methylene diphosphonate scintigraphy has also been used
but is likely to reveal uptake of isotope in areas of
active osteomalacia, rather than to localize a tumor.
With increasing awareness of this syndrome, and
increasingly refined imaging techniques, we may
discover that such tumors are not as rare as they
were once thought to be.
The report by Jonsson et al. of a serum assay
for the tumor-derived factor FGF-23 will most likely
add to the armamentarium for the diagnosis of this
condition and may help to establish whether tumors have been completely removed by surgery. Circulating levels of FGF-23, however, are increased
in patients with X-linked hypophosphatemia; thus,
this measure does not appear to distinguish between the two conditions. Furthermore, it is not
clear whether the assay preferentially recognizes
the intact FGF-23 molecule or C-terminal fragments
of it. Specific identification of various molecular
1706
n engl j med 348;17
of
medicine
species may provide critical information regarding
the role of FGF-23 processing in the pathogenesis
of hypophosphatemia.
treatment and course
The clinical course of oncogenic osteomalacia
is dramatically affected by removal of the tumor,
which, if it is possible, is the treatment of choice.
The serum phosphate level, indexes of renal phosphate wasting, and circulating levels of 1,25-dihydroxyvitamin D return to normal within hours to
days after the removal of the tumor.5 Serum biochemical markers of bone turnover, such as the
osteocalcin level and alkaline phosphatase activity,
tend to take longer to normalize; long-term skeletal
changes may require months to be corrected.6 The
rapid biochemical response is remarkable, however, and the complete resolution of long-standing
symptoms is a major relief to patients. Rarely in the
treatment of metabolic diseases does a physician
encounter such a complete reversal of debilitating
symptoms a truly gratifying experience.
If a tumor is not found, or if an identified tumor
is not resectable, it is recommended that vitamin
D metabolites (preferably calcitriol) and oral phosphate salts be used in a manner similar to that used
in the treatment of X-linked hypophosphatemia.2
This treatment usually provides some benefit, although it does not result in a complete clinical response. Long-term treatment with intravenous
phosphate, together with oral calcium and vitamin D, has been successfully used when oral phosphate salts were not tolerated.7 This therapy can result in bloodstream infection related to a central
venous catheter and thus should be used only when
the promise of a benefit is greater than the risk of
catheter-related complications. As a short-term
treatment, octreotide therapy rapidly corrected the
serum phosphate level and alkaline phosphatase
activity in a patient with a tumor that had been detected by octreotide scintigraphy.4
Most tumors associated with oncogenic osteomalacia are benign, consisting of mesenchymal
cells or mixed connective tissue. Soft-tissue tumors
are often vascular, with abundant spindle or giant
cells, and are frequently reported as hemangiopericytomas. Tumors that arise in bone are usually classified as chondroblasticosteoblastic, ossifying, or
nonossifying fibromas, as categorized by Weidner
and Santa Cruz.8 Occasional malignant tumors have
been reported.9 According to one report, osteosar-
www.nejm.org
april 24, 2003
The New England Journal of Medicine
Downloaded from nejm.org on September 25, 2013. For personal use only. No other uses without permission.
Copyright 2003 Massachusetts Medical Society. All rights reserved.
editorials
coma was discovered in a patient who had had oncogenic osteomalacia for many years before its source
was identified.10 Other patients have had recurrent
disease that has progressed and become fatal. Thus,
it is highly recommended that the surgeon establish
a margin of resection that is free of tumor, particularly in the case of tumors with substantial atypia or
mitotic elements.
1. Oncogenic osteomalaciaassociated tumor
FGF-23
pathophysiology and
candidate mediators
Despite improvements in our understanding of oncogenic osteomalacia and X-linked hypophosphatemia, the puzzle has not been completely solved.
Loss of function of the PHEX (phosphate-regulating gene with homologies to endopeptidases on the
X chromosome) protease determines the X-linked
hypophosphatemia phenotype, but how this occurs
remains a mystery.2 Specific mutations of FGF-23
result in autosomal dominant hypophosphatemic
rickets11; this may be due to a mutation-induced
inhibition of processing.12 The injection of FGF23transfected cells into mice results in the characteristic features of oncogenic osteomalacia.13 On
the other hand, other factors identified from tumors
responsible for oncogenic osteomalacia have been
implicated in phosphate homeostasis; these factors
include frizzled-related protein 4, matrix extracellular phosphoglycoprotein, and unidentified substances with small molecular weights.14 Figure 1
shows the central role of FGF-23 in the proposed
mechanisms underlying hypophosphatemic disorders.
A number of important questions remain. Does
PHEX degrade FGF-23, thereby accounting for increased levels in patients with X-linked hypophosphatemia? Is FGF-23 the direct mediator responsible for impaired transport of phosphate by the
renal tubules and the impaired regulation of vitamin D metabolism, or does another factor in a complex cascade of signals perform this function? Is
the bone disease simply a result of prolonged hypophosphatemia, or does FGF-23 or another tumorderived substance have a direct effect on the skeleton? What are the roles of frizzled-related protein
4 and matrix extracellular phosphoglycoprotein?
The finding that increased circulating FGF-23 levels
occur in X-linked hypophosphatemia as well as in
oncogenic osteomalacia argues that these two disorders do share a common pathophysiological pathway. Further clinical investigation made possible by
n engl j med 348;17
2. Mutation of arginine residues (176 or 179)
Increased
circulating levels
of FGF-23
179 180
3. PHEX function
PHEX
Intermediate
?
mechanism?
Loss of
PHEX function
Phosphate wasting
Figure 1. Proposed Roles of Fibroblast Growth Factor 23 (FGF-23) in the Generation of Phosphate Wasting.
It is speculated that three mechanisms may occur. First, FGF-23 is a secretory
product of tumors associated with oncogenic osteomalacia, accounting for
increased circulating levels of FGF-23 in patients with oncogenic osteomalacia. The secreted form of the molecule is not known, but it is depicted here as
an intact protein. Second, specific mutations of arginine residues at position
176 or 179 of FGF-23 disrupt the sequence motif recognized by furin proteases, presumably resulting in altered processing or decreased degradation of
the protein the putative mechanism of disease in autosomal dominant hypophosphatemic rickets. Third, the loss of function of PHEX (phosphate-regulating gene with homologies to endopeptidases on the X chromosome),
which occurs in X-linked hypophosphatemia, results in increased circulating
levels of FGF-23. Whether the PHEX protein functions as a protease to cleave
FGF-23 at the depicted site or elsewhere is unknown. It is not clear whether
FGF-23 acts directly on the renal tubule or whether another FGF-23mediated
signal is necessary. The putative cleavage site of FGF-23 between the arginine
residue at position 179 and the serine residue at position 180 is noted.
www.nejm.org
april 24, 2003
The New England Journal of Medicine
Downloaded from nejm.org on September 25, 2013. For personal use only. No other uses without permission.
Copyright 2003 Massachusetts Medical Society. All rights reserved.
1707
editorials
this new assay may shed further light on the complex pathophysiology involved.
Dr. Carpenter reports having received consulting fees from Merck
and Genzyme and a grant from CuraGen.
From the Section of Pediatric Endocrinology, Yale University School
of Medicine, New Haven, Conn.
1. Jonsson KB, Zahradnik R, Larsson T, et al. Fibroblast growth
factor 23 in oncogenic osteomalacia and X-linked hypophosphatemia. N Engl J Med 2003;348:1656-63.
2. Carpenter TO. New perspectives on the biology and treatment
of X-linked hypophosphatemic rickets. Pediatr Clin North Am 1997;
44:443-66.
3. Nguyen BD, Wang EA. Indium-111 pentetreotide scintigraphy of
mesenchymal tumor with oncogenic osteomalacia. Clin Nucl Med
1999;24:130-1.
4. Seufert J, Ebert K, Mller J, et al. Octreotide therapy for tumorinduced osteomalacia. N Engl J Med 2001;345:1883-8.
5. Agus ZS. Oncogenic hypophosphatemic osteomalacia. Kidney
Int 1983;24:113-23.
6. Shane E, Parisien M, Henderson JE, et al. Tumor-induced osteomalacia: clinical and basic studies. J Bone Miner Res 1997;12:
1502-11.
7. Yeung SJ, McCutcheon IE, Schultz P, Gagel RF. Use of long-term
intravenous phosphate infusion in the palliative treatment of tumorinduced osteomalacia. J Clin Endocrinol Metab 2000;85:549-55.
8. Weidner N, Santa Cruz D. Phosphaturic mesenchymal tumors:
a polymorphous group causing osteomalacia or rickets. Cancer
1987;59:1442-54.
9. Drezner MK. Tumor-induced osteomalacia. In: Favus MJ, ed.
Primer on the metabolic bone diseases and disorders of mineral
metabolism. 4th ed. Philadelphia: Lippincott Williams & Wilkins,
1999:331-7.
10. Cheng CL, Ma J, Wu PC, Mason RS, Posen S. Osteomalacia secondary to osteosarcoma: a case report. J Bone Joint Surg Am 1989;
71:288-92.
11. The ADHR Consortium. Autosomal dominant hypophosphataemic rickets is associated with mutations in FGF23. Nat Genet
2000;26:345-8.
12. White KE, Carn G, Lorenz-Depiereux B, Benet-Pages A, Strom
TM, Econs MJ. Autosomal-dominant hypophosphatemic rickets
(ADHR) mutations stabilize FGF-23. Kidney Int 2001;60:2079-86.
13. Shimada T, Mizutani S, Muto T, et al. Cloning and characterization of FGF23 as a causative factor of tumor-induced osteomalacia.
Proc Natl Acad Sci U S A 2001;98:6500-5.
14. Schiavi SC, Moe OW. Phosphatonins: a new class of phosphateregulating proteins. Curr Opin Nephrol Hypertens 2002;11:423-30.
Copyright 2003 Massachusetts Medical Society.
posting presentations at medical meetings on the internet
Posting an audio recording of an oral presentation at a medical meeting on the
Internet, with selected slides from the presentation, will not be considered prior
publication. This will allow students and physicians who are unable to attend the
meeting to hear the presentation and view the slides. If there are any questions
about this policy, authors should feel free to call the Journal s Editorial Offices.
1708
n engl j med 348;17
www.nejm.org
april 24, 2003
The New England Journal of Medicine
Downloaded from nejm.org on September 25, 2013. For personal use only. No other uses without permission.
Copyright 2003 Massachusetts Medical Society. All rights reserved.
You might also like
- 2013 ABFM ITE Critique - HighlightsDocument81 pages2013 ABFM ITE Critique - Highlightskelvin273219No ratings yet
- Demand Letter - Peter Pan II 81616Document5 pagesDemand Letter - Peter Pan II 81616api-326532936No ratings yet
- AAFP 2016 AnswerDocument88 pagesAAFP 2016 AnswerAbdirahman AliNo ratings yet
- Placental AbnormalitiesDocument3 pagesPlacental AbnormalitiesThakoon TtsNo ratings yet
- Abfm 2011-2016 Ite ExamsDocument499 pagesAbfm 2011-2016 Ite ExamsJames Moore44% (9)
- Coronary Artery DiseaseDocument4 pagesCoronary Artery DiseaseCharles MatabangNo ratings yet
- A. Biology of Aging PDFDocument9 pagesA. Biology of Aging PDFlolipopckpNo ratings yet
- b7 PPT, AntihistaminesDocument56 pagesb7 PPT, AntihistaminesKeziah GillNo ratings yet
- A Student Manual of Clinical Skills 13 7 8Document194 pagesA Student Manual of Clinical Skills 13 7 8mofath100% (5)
- Legionella Toolbox TalkDocument4 pagesLegionella Toolbox TalkJohn GeddesNo ratings yet
- Neuro-Ophthalmology: Introduction: James Goodwin, MD (Attending)Document4 pagesNeuro-Ophthalmology: Introduction: James Goodwin, MD (Attending)Mariano FioreNo ratings yet
- Preconceptional Counseling and CareDocument12 pagesPreconceptional Counseling and CareGaurav ThakurNo ratings yet
- Severe Sickle Cell Disease-Pathophysiology and Therapy 2010Document4 pagesSevere Sickle Cell Disease-Pathophysiology and Therapy 2010Shoua XiongNo ratings yet
- A Premature Neonate LueucicitosisDocument3 pagesA Premature Neonate Lueucicitosisgonococo29No ratings yet
- Manejo IncidentalomasDocument3 pagesManejo IncidentalomasAlexandra FreireNo ratings yet
- Literature Review On Sickle Cell DiseaseDocument8 pagesLiterature Review On Sickle Cell Diseaseafmzwflmdnxfeb100% (1)
- HandbookDocument4 pagesHandbookDr Venkatachalapathy T S Ped SurgeonNo ratings yet
- A Way To Reverse CADDocument11 pagesA Way To Reverse CADArthurNo ratings yet
- Leukemia Genetics and Prognostic FactorsDocument6 pagesLeukemia Genetics and Prognostic FactorsEdgard CubasNo ratings yet
- Prediction, Progression, and Outcomes of Chronic Kidney Disease in Older AdultsDocument11 pagesPrediction, Progression, and Outcomes of Chronic Kidney Disease in Older AdultsAndreea GheorghiuNo ratings yet
- Denosumab For Treatment of Hypercalcemia of MalignancyDocument9 pagesDenosumab For Treatment of Hypercalcemia of MalignancySandra PargaNo ratings yet
- A Double-Blind, Placebo-Controlled Trial of Ruxolitinib For MyelofibrosisDocument9 pagesA Double-Blind, Placebo-Controlled Trial of Ruxolitinib For MyelofibrosisharlessitompulNo ratings yet
- Novel p53 Splicing Site Mutation in Li-Fraumeni-like Syndrome With OsteosarcomaDocument5 pagesNovel p53 Splicing Site Mutation in Li-Fraumeni-like Syndrome With OsteosarcomaMissy MishaNo ratings yet
- Retinoblastoma: ProblemsDocument1 pageRetinoblastoma: ProblemsArdianNo ratings yet
- Karen A. Gelmon: Desmoplastic Small Round-Cell Tumor of The PancreasDocument3 pagesKaren A. Gelmon: Desmoplastic Small Round-Cell Tumor of The PancreasAdam HartonoNo ratings yet
- Guia para El MedicoDocument6 pagesGuia para El MedicoJuan Sebastian OrdoñezNo ratings yet
- Scurvy Masquerading as Rheumatic ConditionsDocument6 pagesScurvy Masquerading as Rheumatic ConditionsZach Segmuel MiñanoNo ratings yet
- A Question Well Put: Clinical Problem-SolvingDocument6 pagesA Question Well Put: Clinical Problem-SolvingNadia Mrz Mattew MuseNo ratings yet
- Achondroplasia Natural History Study (CLARITY)Document7 pagesAchondroplasia Natural History Study (CLARITY)jakelinelagoadvNo ratings yet
- CaseDocument24 pagesCasecintaNo ratings yet
- DMD Verma2010mdDocument8 pagesDMD Verma2010mdFajri MuhammadNo ratings yet
- Research Paper On Acute Lymphoblastic LeukemiaDocument7 pagesResearch Paper On Acute Lymphoblastic Leukemiafzkk43h9100% (1)
- Rheumatology 2011 Muenzer v4 v12Document9 pagesRheumatology 2011 Muenzer v4 v12BoNo ratings yet
- Literature Review of Sickle Cell AnaemiaDocument5 pagesLiterature Review of Sickle Cell Anaemiaafdtveepo100% (1)
- Dissertation On Sickle Cell DiseaseDocument4 pagesDissertation On Sickle Cell DiseaseBuyACollegePaperMobile100% (1)
- Effects on Neuroblastoma Tumor Initiating Cells and Established Neuroblastoma Cell Lines Using Putative Inhibitors of HIF-2αDocument18 pagesEffects on Neuroblastoma Tumor Initiating Cells and Established Neuroblastoma Cell Lines Using Putative Inhibitors of HIF-2αDavid SchmidtNo ratings yet
- JBMR-37-2602Document13 pagesJBMR-37-2602Juanjo Moreno LópezNo ratings yet
- Síndrome de Cushing. Maybe Not So Uncommon of An Endocrine DiseaseDocument10 pagesSíndrome de Cushing. Maybe Not So Uncommon of An Endocrine DiseaseStephany Gutiérrez VargasNo ratings yet
- Adult Minimal-Change Disease: Clinical Characteristics, Treatment, and OutcomesDocument9 pagesAdult Minimal-Change Disease: Clinical Characteristics, Treatment, and OutcomesMutiara RizkyNo ratings yet
- Long-Term Kidney Function of Childhood Cancer Survivors-Who Needs To Be Monitored?Document4 pagesLong-Term Kidney Function of Childhood Cancer Survivors-Who Needs To Be Monitored?Irawati Eka PutriNo ratings yet
- Primary Midgut VolvulusDocument2 pagesPrimary Midgut VolvulusNihal S KiranNo ratings yet
- Simon 2016Document12 pagesSimon 2016yoNo ratings yet
- Research Paper On Duchenne Muscular DystrophyDocument4 pagesResearch Paper On Duchenne Muscular Dystrophycantfp00100% (1)
- WC 500020933Document5 pagesWC 500020933Anonymous DqeRReNo ratings yet
- Chemotherapy For Tuberculous Meningitis: Peter R. Donald, M.DDocument3 pagesChemotherapy For Tuberculous Meningitis: Peter R. Donald, M.DDirga Rasyidin LNo ratings yet
- Editorial: Editorial Current Drug Targets, 2007, Vol. 8, No. 4 491Document76 pagesEditorial: Editorial Current Drug Targets, 2007, Vol. 8, No. 4 491laraalvarenga5832No ratings yet
- Sickle Cell Disease: Advances in TreatmentDocument13 pagesSickle Cell Disease: Advances in TreatmentarturomarticarvajalNo ratings yet
- Prophylactic Thyroidectomy in Children at Risk of Medullary Thyroid CancerDocument4 pagesProphylactic Thyroidectomy in Children at Risk of Medullary Thyroid CancerDr Venkatachalapathy T S Ped SurgeonNo ratings yet
- Acute Lymphoblastic Leukemia ThesisDocument5 pagesAcute Lymphoblastic Leukemia Thesisljctxlgld100% (2)
- Statin Intolerance, Bempedoic Acid, and Cardiovascular OutcomesDocument4 pagesStatin Intolerance, Bempedoic Acid, and Cardiovascular OutcomesAnderson Joel GomezNo ratings yet
- Literature Review of Chronic Liver DiseaseDocument6 pagesLiterature Review of Chronic Liver Diseaseafmzodjhpxembt100% (1)
- CM HearttransplantDocument8 pagesCM HearttransplantarthoclaseNo ratings yet
- Lycopene and Myocardial Infarction Risk in The EURAMIC StudyDocument9 pagesLycopene and Myocardial Infarction Risk in The EURAMIC StudySUBHADIP MANIKNo ratings yet
- Congestion Cardiaca y PeriodontitisDocument7 pagesCongestion Cardiaca y Periodontitiscruzjulio480No ratings yet
- Refeeding Syndrome A Literature Review KhanDocument7 pagesRefeeding Syndrome A Literature Review Khangw060qpy100% (1)
- CaseDocument7 pagesCaseEka NurjannahNo ratings yet
- Haematology Thesis TopicsDocument6 pagesHaematology Thesis Topicsheidiowensspringfield100% (2)
- Pathophysiology, Diagnosis, and Management of Dyslipidemia: Gerald T. Gau, MD, and R. Scott Wright, MD, FACC, FAHA, FESCDocument42 pagesPathophysiology, Diagnosis, and Management of Dyslipidemia: Gerald T. Gau, MD, and R. Scott Wright, MD, FACC, FAHA, FESCSherleen HeiNo ratings yet
- Pathophysiology, Diagnosis, and Management of Dyslipidemia: Gerald T. Gau, MD, and R. Scott Wright, MD, FACC, FAHA, FESCDocument42 pagesPathophysiology, Diagnosis, and Management of Dyslipidemia: Gerald T. Gau, MD, and R. Scott Wright, MD, FACC, FAHA, FESCCalvin LitraNo ratings yet
- Heart Failure With Preserved Ejection Fraction - JAMA (2023)Document12 pagesHeart Failure With Preserved Ejection Fraction - JAMA (2023)cheve glzNo ratings yet
- Heart Failure in Patients With Metabolic Syndrome XDocument12 pagesHeart Failure in Patients With Metabolic Syndrome XErwin SiahaanNo ratings yet
- Sickle Cell Anemia A Succinct Look Into Recently Tested Disease Modifying InterventionsDocument10 pagesSickle Cell Anemia A Succinct Look Into Recently Tested Disease Modifying InterventionsAthenaeum Scientific PublishersNo ratings yet
- Venous Thrombosis,: Atopic Dermatitis (AD)Document5 pagesVenous Thrombosis,: Atopic Dermatitis (AD)Kenneth DomirezNo ratings yet
- A New Step in The Treatment of Sickle Cell DiseaseDocument2 pagesA New Step in The Treatment of Sickle Cell Disease422073224No ratings yet
- Peters Et Al. 2003. Hematopoietic Cell Transplantation For Inherited Metabolic Diseases - An Overview of Outcomes and Practice GuidelinesDocument11 pagesPeters Et Al. 2003. Hematopoietic Cell Transplantation For Inherited Metabolic Diseases - An Overview of Outcomes and Practice GuidelinesBoNo ratings yet
- Sarcoidosis Review. JAMADocument9 pagesSarcoidosis Review. JAMARafael ReañoNo ratings yet
- Neuroendocrine Tumors: Surgical Evaluation and ManagementFrom EverandNeuroendocrine Tumors: Surgical Evaluation and ManagementJordan M. CloydNo ratings yet
- Table 1Document2 pagesTable 1Asticha Erlianing SariNo ratings yet
- In Order To Print Your Document Correctly, You Should Click "Done" in Pdffiller and Choose "Print" On The Next ScreenDocument1 pageIn Order To Print Your Document Correctly, You Should Click "Done" in Pdffiller and Choose "Print" On The Next ScreenAsticha Erlianing SariNo ratings yet
- Saoidhsaoldkahskldhakldhdhw Qidoh W DHGW J.DBHKJ D Hasgdulgdjkwhqdu HDocument1 pageSaoidhsaoldkahskldhakldhdhw Qidoh W DHGW J.DBHKJ D Hasgdulgdjkwhqdu HAsticha Erlianing SariNo ratings yet
- Nej Mic M 066066Document1 pageNej Mic M 066066Asticha Erlianing SariNo ratings yet
- Oncogenic Osteomalacia - A Complex Dance of Factors: Thomas O. Carpenter, M.DDocument4 pagesOncogenic Osteomalacia - A Complex Dance of Factors: Thomas O. Carpenter, M.DAsticha Erlianing SariNo ratings yet
- Understand The Facts - Anxiety and Depression Association of America, ADAADocument5 pagesUnderstand The Facts - Anxiety and Depression Association of America, ADAAAhmad Badius ZamanNo ratings yet
- Tablet binders and excipientsDocument30 pagesTablet binders and excipientsmary navalNo ratings yet
- Banned Drugs in IndiaDocument3 pagesBanned Drugs in IndiaGottumukkala Venkateswara RaoNo ratings yet
- Ayurvedic Treatment of PCOSDocument5 pagesAyurvedic Treatment of PCOSArun AchalamNo ratings yet
- Holoxan Pi PDFDocument18 pagesHoloxan Pi PDFKarol IonasNo ratings yet
- Aids 2013Document404 pagesAids 2013kovaron80No ratings yet
- A Prospective Cohort Study of 122 Adult Patients Presenting To An Otolaryngologist's Office With Globus Pharyngeus (RASMUSSEN 2018)Document20 pagesA Prospective Cohort Study of 122 Adult Patients Presenting To An Otolaryngologist's Office With Globus Pharyngeus (RASMUSSEN 2018)DANDYNo ratings yet
- The Delirium Rating Scale (DRS)Document12 pagesThe Delirium Rating Scale (DRS)Cristian SolísNo ratings yet
- Emerging and Reemerging InfectionDocument1 pageEmerging and Reemerging InfectionKurt Bryan CortezNo ratings yet
- Parasitology Lab Study ChartDocument2 pagesParasitology Lab Study ChartnoneyabuNo ratings yet
- IMI RANO Critieria Booklet Nov 2011Document18 pagesIMI RANO Critieria Booklet Nov 2011johnthomas75No ratings yet
- Diagnostic Test DesignDocument30 pagesDiagnostic Test DesignDika Gita PratamaNo ratings yet
- Newborn AssessmentDocument6 pagesNewborn AssessmentSirine AjourNo ratings yet
- Critical Nursing IntroductionDocument4 pagesCritical Nursing IntroductionJo Traven AzueloNo ratings yet
- Home Care For Your Kids TeethDocument2 pagesHome Care For Your Kids TeethDr. Kristin ElliotNo ratings yet
- PhotokeratitisDocument15 pagesPhotokeratitisSelase Di MariaNo ratings yet
- BB Pheresis GuidelinesDocument190 pagesBB Pheresis GuidelinesBill ThomNo ratings yet
- ResearchDocument6 pagesResearchhaerul ikhsanNo ratings yet
- 10 Mcqs Canada For SampleDocument5 pages10 Mcqs Canada For SampleNavjot BrarNo ratings yet
- PKMDocument3 pagesPKMSarah ArumNo ratings yet
- Cebu Institute of Technology - University: Nursing Care PlanDocument2 pagesCebu Institute of Technology - University: Nursing Care PlanSergiNo ratings yet
- Gastritis DDDocument78 pagesGastritis DDsotoshaNo ratings yet