Professional Documents
Culture Documents
Titration Curve of H Po: Volume of Naoh (ML)
Titration Curve of H Po: Volume of Naoh (ML)
Uploaded by
Raven Poala Ramos0 ratings0% found this document useful (0 votes)
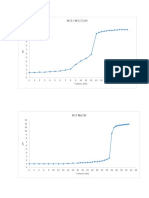
9 views1 pageThis titration curve shows the pH changes as sodium hydroxide (NaOH) is added to hydrogen phosphate (H2PO4-). The pH remains relatively constant between 0-5 mL of NaOH as H2PO4- is neutralized. From 5-15 mL of NaOH the pH increases sharply as neutralization of H2PO4- is complete and the solution becomes more basic. The pH levels off after 15 mL as excess NaOH is present.
Original Description:
T
Original Title
Titration
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis titration curve shows the pH changes as sodium hydroxide (NaOH) is added to hydrogen phosphate (H2PO4-). The pH remains relatively constant between 0-5 mL of NaOH as H2PO4- is neutralized. From 5-15 mL of NaOH the pH increases sharply as neutralization of H2PO4- is complete and the solution becomes more basic. The pH levels off after 15 mL as excess NaOH is present.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
9 views1 pageTitration Curve of H Po: Volume of Naoh (ML)
Titration Curve of H Po: Volume of Naoh (ML)
Uploaded by
Raven Poala RamosThis titration curve shows the pH changes as sodium hydroxide (NaOH) is added to hydrogen phosphate (H2PO4-). The pH remains relatively constant between 0-5 mL of NaOH as H2PO4- is neutralized. From 5-15 mL of NaOH the pH increases sharply as neutralization of H2PO4- is complete and the solution becomes more basic. The pH levels off after 15 mL as excess NaOH is present.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 1
12
Titration curve of H2PO4-‐
11
10
pH
6
0 5 10 Volume
of
NaOH
(mL)
15 20 25
You might also like
- Lehninger Principles of Biochemistry 6th Edition Nelson Solutions ManualDocument24 pagesLehninger Principles of Biochemistry 6th Edition Nelson Solutions ManualHollyBurnsPhDwadn100% (35)
- Kurva Titrasi PH Vs Volume Penambahan Naoh Kurva Titrasi PH Vs Volume Penambahan NaohDocument1 pageKurva Titrasi PH Vs Volume Penambahan Naoh Kurva Titrasi PH Vs Volume Penambahan NaohMaulinda Imansari ArmaytabinoyaNo ratings yet
- 1 0 Konsentrasi: HCL HCL 0Document6 pages1 0 Konsentrasi: HCL HCL 0Fina HandayaniNo ratings yet
- Y-Values: A. Making Titration CurveDocument3 pagesY-Values: A. Making Titration CurveFatin AtikahNo ratings yet
- Naoh Vol. Buret Max. Value Ph1 ### Ph2 Ph3 Ph4 - 1.65 HCL Va 5.9 ML Ma 0.06 M Naoh VB 10 ML MB 0 M KWDocument4 pagesNaoh Vol. Buret Max. Value Ph1 ### Ph2 Ph3 Ph4 - 1.65 HCL Va 5.9 ML Ma 0.06 M Naoh VB 10 ML MB 0 M KWafra afifahNo ratings yet
- 3Document2 pages3Juan R SyahidNo ratings yet
- PH MetriDocument1 pagePH MetriAnis DwiNo ratings yet
- Grafik Hubungan Antara PH CH Cooh Dengan Volume Total NaohDocument4 pagesGrafik Hubungan Antara PH CH Cooh Dengan Volume Total NaohAji SaputraNo ratings yet
- Dependența PH F (V (Naoh) ) P/U Acid OxalicDocument10 pagesDependența PH F (V (Naoh) ) P/U Acid OxalicEllenKojocaruNo ratings yet
- Quantitative Analysis of Phosphoric Acid Found in Commercial Soft Drink "Pool Cola" Via Potentiometric TitrationDocument4 pagesQuantitative Analysis of Phosphoric Acid Found in Commercial Soft Drink "Pool Cola" Via Potentiometric TitrationSofía EscobarNo ratings yet
- Dab Tanpa OptimasiDocument1 pageDab Tanpa OptimasiGenta Huda FauzanNo ratings yet
- Kurva AgriDocument2 pagesKurva AgriAgri PahleviNo ratings yet
- Darimbang - PPT - BSFT - Quantitative Chemistry 10 - Nadzma DarimbangDocument13 pagesDarimbang - PPT - BSFT - Quantitative Chemistry 10 - Nadzma DarimbangLoraine Ruth LumenNo ratings yet
- Experiment 3 Conductometric Titration: University of Bahrain College of Science Department of ChemistryDocument7 pagesExperiment 3 Conductometric Titration: University of Bahrain College of Science Department of ChemistryFaisal MumtazNo ratings yet
- Jurnal 1Document9 pagesJurnal 1SARAH AZIZAHNo ratings yet
- Dependența PH F (V (Naoh) ) P/U Acid OxalicDocument19 pagesDependența PH F (V (Naoh) ) P/U Acid OxalicEllenKojocaruNo ratings yet
- BIOQUIMICADocument2 pagesBIOQUIMICAZully Denisse Castillo CamaNo ratings yet
- Chart Title: PH 0 1 1 1.2 2 1.4 3 1.6 4 2 5 7 6 12 7 12.2 8 12.4 9 12.5 10 12.5 Naoh 0,1N (ML) PH 1Document2 pagesChart Title: PH 0 1 1 1.2 2 1.4 3 1.6 4 2 5 7 6 12 7 12.2 8 12.4 9 12.5 10 12.5 Naoh 0,1N (ML) PH 1Zully Denisse Castillo CamaNo ratings yet
- Simbol Bahaya AkaDocument9 pagesSimbol Bahaya AkaLuckyZainurRoziqinNo ratings yet
- Reaction of Aldehyde CompleteDocument1 pageReaction of Aldehyde CompleteJoko SusiloNo ratings yet
- Ionic Equilibrium-03-Objective and Subjective Assignments and Answer SheetDocument16 pagesIonic Equilibrium-03-Objective and Subjective Assignments and Answer SheetRaju SinghNo ratings yet
- Aep 8Document6 pagesAep 8devkaushik0613No ratings yet
- Sample Titration LabDocument11 pagesSample Titration LabJENGABOYNo ratings yet
- Potensio GrafikDocument2 pagesPotensio GrafikpiranindhaNo ratings yet
- Basic Systems of ChemDocument55 pagesBasic Systems of Chemvel muruganNo ratings yet
- Acid-Base Titration CurvesDocument14 pagesAcid-Base Titration CurvesAchmad AkbarNo ratings yet
- 2024 Carboxylic Acid and Derivatives Tutorial (Teacher)Document17 pages2024 Carboxylic Acid and Derivatives Tutorial (Teacher)Anarkin FitriNo ratings yet
- Ionic Equilibrium Sub ObjDocument13 pagesIonic Equilibrium Sub ObjAman9692No ratings yet
- HO / "' OH OH: Li SoDocument4 pagesHO / "' OH OH: Li So葉建豪No ratings yet
- Vnaohvsph: Analisis Data 1. Tabel Volume Naoh Vs PHDocument3 pagesVnaohvsph: Analisis Data 1. Tabel Volume Naoh Vs PHulul dwi yuliandaNo ratings yet
- Acids, Bases, and BuffersDocument8 pagesAcids, Bases, and BuffersPeshala NishadiNo ratings yet
- Ch312 Lecture05 Ch9 10 11 w09Document38 pagesCh312 Lecture05 Ch9 10 11 w09Diyu MNo ratings yet
- Supplementary Material - 31.05.19Document18 pagesSupplementary Material - 31.05.19pipitpiopio1234No ratings yet
- Conversion of Polyacrylamide To Sodium Polyacrylatesalt and Subsequent Conversions To Poly Acrylic Acid PDFDocument6 pagesConversion of Polyacrylamide To Sodium Polyacrylatesalt and Subsequent Conversions To Poly Acrylic Acid PDFaryanNo ratings yet
- Lecture 2a: Determination of The Concentration and The Acid Dissociation Constants of An Unknown Amino Acid (Part II)Document7 pagesLecture 2a: Determination of The Concentration and The Acid Dissociation Constants of An Unknown Amino Acid (Part II)Steve LiNo ratings yet
- 4.94 Phase Diagram For Sodium Sulfate SolutionsDocument1 page4.94 Phase Diagram For Sodium Sulfate SolutionsTRY11E PRIYADHARSHINI.MNo ratings yet
- Organic RevisionDocument4 pagesOrganic RevisionalicejessicapreesNo ratings yet
- Kurva Titrasi Asam Basa: No Volume CH Cooh Volume Naoh PH 1 2 3 4 5 6 7 8 9 10 11 12Document3 pagesKurva Titrasi Asam Basa: No Volume CH Cooh Volume Naoh PH 1 2 3 4 5 6 7 8 9 10 11 12masrNo ratings yet
- U08 Notes Part4 BuffersDocument25 pagesU08 Notes Part4 Buffersapi-546066323No ratings yet
- Chapter 14 Principle of Neutralization TDocument29 pagesChapter 14 Principle of Neutralization TS. MartinezNo ratings yet
- Chem Lab ManualDocument63 pagesChem Lab ManualBala NandaNo ratings yet
- Chapter 15Document31 pagesChapter 15alyssaaaaaNo ratings yet
- Chem Lab Grade 10Document2 pagesChem Lab Grade 10Huck KimNo ratings yet
- Grafik Hubungan Antara PH Terhadap Volume (Sampel 2)Document2 pagesGrafik Hubungan Antara PH Terhadap Volume (Sampel 2)Laely PermanasariNo ratings yet
- Haldia Institute of Technology Engineering Chemistry Laboratory (CH 191 &CH 291)Document29 pagesHaldia Institute of Technology Engineering Chemistry Laboratory (CH 191 &CH 291)Shresth Sanskar100% (1)
- Chemistry LaboratoryDocument31 pagesChemistry LaboratoryBunty KhiljiNo ratings yet
- DEBER PROCESOS (Recuperado Automáticamente)Document14 pagesDEBER PROCESOS (Recuperado Automáticamente)Marx Santiago Ramon AlvaradoNo ratings yet
- Experiment 3 Conductometry HCLDocument5 pagesExperiment 3 Conductometry HCLMayank BajajNo ratings yet
- Reaction of AldehydesDocument1 pageReaction of AldehydesJoko SusiloNo ratings yet
- Acid - Base Free Response Questions Ver 1 Partial AnswersDocument5 pagesAcid - Base Free Response Questions Ver 1 Partial AnswersPrime JackNo ratings yet
- Nyb U4 Acids Bases Part 2Document81 pagesNyb U4 Acids Bases Part 2Aindrila KaziNo ratings yet
- Road Maps Organic Chemistry Set 3 Eklavya @JEEAdvanced - 2024Document6 pagesRoad Maps Organic Chemistry Set 3 Eklavya @JEEAdvanced - 2024puneethrgcNo ratings yet
- Shela PDFDocument11 pagesShela PDFAzizah AmaliyahNo ratings yet
- Reaksi EliminasiDocument23 pagesReaksi EliminasiAde FadilahNo ratings yet
- Lecture 14 PDFDocument4 pagesLecture 14 PDFVikas Verma V. KumarNo ratings yet
- Buffers and Solubility EquilibriaDocument24 pagesBuffers and Solubility EquilibriaeggyNo ratings yet
- ChemistryDocument4 pagesChemistrySanath SaragadamNo ratings yet
- 2 - 2 - 5 Related DensityDocument2 pages2 - 2 - 5 Related DensityHưng Lữ Nguyễn PhúcNo ratings yet
- Reactions in Aqueous Solutions II: CalculationsDocument39 pagesReactions in Aqueous Solutions II: CalculationsSantiago Gonzalez BogotaNo ratings yet
- Ch5 Embryo LecDocument3 pagesCh5 Embryo LecRaven Poala RamosNo ratings yet
- Embryo EssayDocument2 pagesEmbryo EssayRaven Poala RamosNo ratings yet
- The Extracellular Matrix and Cell InteractionsDocument7 pagesThe Extracellular Matrix and Cell InteractionsRaven Poala RamosNo ratings yet
- Graph PDFDocument1 pageGraph PDFRaven Poala RamosNo ratings yet
- W F D Cos Weight Mass G FG WG - FG H (Going Up) WG FG H (Going Down) Ps FG H (Going Up) T Ps - FG H (Going Down) T As TheDocument2 pagesW F D Cos Weight Mass G FG WG - FG H (Going Up) WG FG H (Going Down) Ps FG H (Going Up) T Ps - FG H (Going Down) T As TheRaven Poala RamosNo ratings yet