Professional Documents
Culture Documents
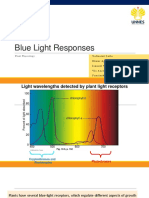
Photoreceptors mediate plant movements
Uploaded by
Hamdan Fatah0 ratings0% found this document useful (0 votes)
12 views3 pagesqwdsacdfrw
Original Title
hot1
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this Documentqwdsacdfrw
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
12 views3 pagesPhotoreceptors mediate plant movements
Uploaded by
Hamdan Fatahqwdsacdfrw
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 3
hot1, while second positive curvature is mediated by
both phot1 and phot2. Phot1 and photo2 contribute
equally to stomatal opening, while the avoidance movement of chloroplasts under high light intensities is
mediated only by phot2.
Recent evidence has also indicated a role for
phototropins in the promotion of cotyledon and leaf
expansion and the rhythmic sleep movements of kidney
bean leaves (see Chapter 24).
23.1.7 A HYBRID RED/BLUE LIGHT
PHOTORECEPTOR HAS BEEN
ISOLATED FROM A FERN
A particularly interesting photoreceptor has recently
been isolated from the fern Adiantum capillus-veneris.
Designated neochrome (formerly known as phy3),
this photoreceptor has properties of both phytochrome
and phototropin (Figure 23.7). The amino acid
sequence of the amino-terminal domain shows a
significant homology to the chromophore-binding
domain of phytochrome. Furthermore, when the gene
was expressed in yeast and the purified protein was
reconstituted with a phycocyanobilin chromophore, it
showed typical phytochrome photoreversible behavior.
But neochrome also has the two LOV domains, which
bind FMN, and a serine/threonine kinase domain
at the C-terminus that are virtually identical to
phototropin. Neochrome is required for phototropism
in Adiantum, which is regulated by red as well as blue
light. Neochrome is clearly a hybrid photoreceptor
that mediates red, far-red, and blue-light responses.
What is curious, however, is that Adiantum also has
two fully functional phototropins like their higher
plant counterparts and, again like their higher plant
counterparts, phot2 is solely responsible for mediating
high-light chloroplast avoidance movements.
Several other ferns, mosses, and algae have both
phototropins and neochromes, which raises interesting
questions regarding the evolution of photoreceptors as
well as their physiological action.
23.1.8 PHOTOTROPIN ACTIVITY
AND SIGNAL CHAIN
Participants in the phototropin signal chain are only
now just beginning to ‘‘come to light.’’ Some of the
more important factors can be summarized as follows:
1. Autophosphorylation of phototropin plays a significant
role in the phototropic response, probably by initiating
a phosphorylation cascade. Studies employing mutants
of the PHOT1 gene have shown that the protein is
apparently folded in such a way that the phosphorylation site is blocked by the LOV2 domain in the
dark. Absorption of blue light by the chromophore
induces a change in the conformation of the protein so that the phosphorylation site is available and
active. The role of the LOV1 domain is not clear.
Mutants lacking the LOV1 domain have shown that
LOV1 is not necessary for phosphorylation but its
presence does increase kinase activity.
2. Phototropins may be involved in gene regulation. No
substrates directly phosphorylated by phototropin
in planta have yet been identified, but there are
several proteins that interact with the photoreceptor and are necessary for a proper response.
For example, the proteins NONPHOTOTROPIC
HYPOCOTYL 3 (NPH3) from Arabidopsis and a
homologous protein (called an ortholog) from rice,
COLEOPTILE PHOTOTROPISM 1 (CPT1),
include domains that are characteristic of transcriptional regulators or proteins that are involved in
protein degradation. The mutants nph3 and cpt1,
in which these proteins are missing, show no phototropic response.
3. Phototropin disrupts polar auxin transport. One of the
challenges presented by phototropism is to establish
whether or not a link exists between the absorption
398 Chapter 23 / Tropisms and Nastic Movements: Orienting Plants in Space
of blue light by phototropin and the asymmetrical
auxin distribution proposed by the Cholodny-Went
hypothesis. As described previously, asymmetric
auxin distribution has been demonstrated experimentally. Recent experiments have focused on the
relationship between phototropin and auxin efflux
facilitator PIN1. PIN1 is normally localized at the
basal ends of xylem-associated cells where it
serves to facilitate the polar vertical flow of auxin
(Chapter 18). When the location of PIN1 in Arabidopsis hypocotyls was monitored by immunofluorescence
microscopy following phototropic stimulus,
the basal location of PIN1 in wildtype plants
was disrupted in the cortical cells on the shaded
side of the hypocotyl. A similar disruption was
not observed in phot1 mutants. These results
suggest that phototropic bending is initiated by
a phototropin-mediated decrease in the vertical
transport of auxin. This would lead to a retention
or sequestering of auxin, and consequent increased
growth, in those cells that are directly involved in
phototropic bending.
23.1.9 PHOTOTROPISM IN GREEN
PLANTS IS NOT WELL
UNDERSTOOD
A final area of concern is phototropism in light-grown
plants, where relatively little is known about the
phototropic process. As with phytochrome, discussed in
Chapter 22, most of what we know about phototropism
is derived from laboratory studies with etiolated seedlings. However, in light-grown cucumber (Cucumis
sativus) and sunflower (Helianthus annuus) seedlings
subjected to uniform lighting, curvature of the
stem can be induced by simply shading one of the
FIGURE 23.8 Curvature in cucumber (Cucumis sativus)
seedlings induced by shading cotyledons. The left-hand
cotyledon was covered with aluminum foil and the
seedling uniformly irradiated with white light for 8
hours. (After Shuttleworth, J. E., M. Black. 1977. Planta
135:51.)
cotyledons (Figure 23.8), and the phototropic response
of sunflower seedlings is markedly decreased if the
leaves are removed. The cucumber response, at least,
differs from the classical phototropic response in that
it is induced by red light rather than blue light. This
appears to be a phytochrome-mediated response and is
related to inhibition of hypocotyl elongation below the
irradiated cotyledon. Both the cucumber and sunflower
responses may be attributed to the fact that the leaves
are a prime source of auxin required for the growth
response. Both cucumber and white mustard (Sinapis
alba) also exhibit a classical phototropic response
induced by irradiating the hypocotyls directly with blue
light. Clearly the control of stem growth in green plants
is an area where there is still much to learn.
23.2 GRAVITROPISM
Gravitropism is probably one of the most unfailingly
obvious and familiar plant phenomena to most people
(Figure 23.9). Everyone is aware that shoots always
grow ‘‘up’’ and roots always grow ‘‘down.’’ Or do they?
A casual walk through the woods or garden should
reveal how overly simplified this view is. The lateral
branches of most trees and shrubs do not grow up; they
grow outward in a more or less horizontal position.
Stolons (or runners) of stra
You might also like
- Function of Quinones in Energy Conserving SystemsFrom EverandFunction of Quinones in Energy Conserving SystemsBernard TrumpowerNo ratings yet
- Blue Light Receptors Regulate Plant Growth and DevelopmentDocument22 pagesBlue Light Receptors Regulate Plant Growth and Developmentisnaeni nur khasanahNo ratings yet
- Photophysiology: Current Topics in Photobiology and PhotochemistryFrom EverandPhotophysiology: Current Topics in Photobiology and PhotochemistryArthur C. GieseNo ratings yet
- Illuminating Progress in Phytochrome Mediated LighDocument10 pagesIlluminating Progress in Phytochrome Mediated LighSudhakar SivaNo ratings yet
- Plants Orient in Space Through Tropisms and Nastic MovementsDocument55 pagesPlants Orient in Space Through Tropisms and Nastic MovementsReena Clarisse AviñanteNo ratings yet
- Phyto ChromeDocument7 pagesPhyto ChromeNovan AdhiNo ratings yet
- Introduction To PhototropismDocument6 pagesIntroduction To PhototropismBenjamin SalopNo ratings yet
- Phytochromes promote seedling light responses by inhibiting four negatively-acting phytochrome-interacting factorsDocument6 pagesPhytochromes promote seedling light responses by inhibiting four negatively-acting phytochrome-interacting factorsManas BeheraNo ratings yet
- Light-Regulated Transcriptional Networks in Higher Plants: Nature Reviews Genetics March 2007Document15 pagesLight-Regulated Transcriptional Networks in Higher Plants: Nature Reviews Genetics March 2007meiNo ratings yet
- AP Bio HW #13A Chap 39 130114Document13 pagesAP Bio HW #13A Chap 39 130114BiseshNo ratings yet
- CryptochromesDocument17 pagesCryptochromesMubeena MoossaNo ratings yet
- Lecture 14 PPIDocument17 pagesLecture 14 PPIabeehazakeeshNo ratings yet
- Photoreceptors in Plant Photomorphogenesis To Date. Five Phytochromes, Two Cryptochromes, One Phototropin, and One SuperchromeDocument4 pagesPhotoreceptors in Plant Photomorphogenesis To Date. Five Phytochromes, Two Cryptochromes, One Phototropin, and One SuperchromeCarmi MaternalNo ratings yet
- Photosystem IIDocument4 pagesPhotosystem IIMauricioNo ratings yet
- Phytochrome and Light Control of Plant DevelopmentDocument30 pagesPhytochrome and Light Control of Plant Developmenttram tramNo ratings yet
- S.No Page No 1) 1Document13 pagesS.No Page No 1) 1gopi4krishna4reddyNo ratings yet
- Pnas 0607751103Document6 pagesPnas 0607751103Khyna Ayessa Mae SorianoNo ratings yet
- Annurev 2earplant 2E59 2E032607 2E092859Document31 pagesAnnurev 2earplant 2E59 2E032607 2E092859andiNo ratings yet
- CryptochromeDocument5 pagesCryptochromehannahlralstonNo ratings yet
- 2 26 1620453066 Ijbtrjun20213Document8 pages2 26 1620453066 Ijbtrjun20213TJPRC PublicationsNo ratings yet
- Li Et Al-2013-Journal of Agronomy and Crop ScienceDocument8 pagesLi Et Al-2013-Journal of Agronomy and Crop ScienceAzhari RizalNo ratings yet
- Lemeille-Rochaix2010 Article StateTransitionsAtTheCrossroad PDFDocument14 pagesLemeille-Rochaix2010 Article StateTransitionsAtTheCrossroad PDFGester GutiérrezNo ratings yet
- Cholorophyll Content in Five Different Species of PlantDocument14 pagesCholorophyll Content in Five Different Species of Plantlatest tamil moviesNo ratings yet
- Bio 2D03 Lab 5 - Responses To Light and Hormones F2021Document12 pagesBio 2D03 Lab 5 - Responses To Light and Hormones F2021Vikraman BhargavNo ratings yet
- Shedding Light On Plant Development: Light Signalling in The Model Plant Arabidopsis ThalianaDocument12 pagesShedding Light On Plant Development: Light Signalling in The Model Plant Arabidopsis ThalianaDivya DharshiniNo ratings yet
- PhotomorphogenesisDocument7 pagesPhotomorphogenesisTahreem NisarNo ratings yet
- Botany Lab Activity 7Document5 pagesBotany Lab Activity 7PaulNo ratings yet
- series of eventsDocument10 pagesseries of eventsMarvin JeaNo ratings yet
- Time-Course Transcriptome Analysis Reveals Regulation of Arabidopsis Seed Dormancy by The Transcription Factors WOX11/12Document17 pagesTime-Course Transcriptome Analysis Reveals Regulation of Arabidopsis Seed Dormancy by The Transcription Factors WOX11/12Aghila SamjiNo ratings yet
- Biophotons in Radiobiology Inhibitors, Communicators and ReactorsDocument6 pagesBiophotons in Radiobiology Inhibitors, Communicators and ReactorsRodrigo GalvãoNo ratings yet
- Pengaruh Fotosintesis Terhadap Pertumbuhan Tanaman Kentang (Solanum Tuberosum L.) Dalam Lingkungan Fotoautotrof Secara InvitroDocument7 pagesPengaruh Fotosintesis Terhadap Pertumbuhan Tanaman Kentang (Solanum Tuberosum L.) Dalam Lingkungan Fotoautotrof Secara InvitrodahlanNo ratings yet
- Luciferin in DepthDocument3 pagesLuciferin in DepthVictoire GueroultNo ratings yet
- NIH Public Access: Self-Feedback in Actin PolymerizationDocument12 pagesNIH Public Access: Self-Feedback in Actin Polymerizationafert_7No ratings yet
- chp_39_lectureDocument21 pageschp_39_lectureasadukimbugwe20No ratings yet
- COP1 Mediates Light-Dependent Regulation of Flavonol Biosynthesis Through HY5 in ArabidopsisDocument30 pagesCOP1 Mediates Light-Dependent Regulation of Flavonol Biosynthesis Through HY5 in ArabidopsismNo ratings yet
- Lab ReportDocument8 pagesLab Reportthey12No ratings yet
- Name - Lindo STR-WPS OfficeDocument3 pagesName - Lindo STR-WPS OfficeJay ar ContiverosNo ratings yet
- Plant Responses To LightDocument16 pagesPlant Responses To LightNiamat UllahNo ratings yet
- Mutations in The NPH1 Locus of Arabidopsis Disrupt The Perception of Phototropic StimuliDocument13 pagesMutations in The NPH1 Locus of Arabidopsis Disrupt The Perception of Phototropic StimuliRania ChitourNo ratings yet
- Biology Project 1Document23 pagesBiology Project 1Aashray KothaNo ratings yet
- Cytokinins-Discovery, Biosynthesis and Physiological Role: Unit 5.1Document6 pagesCytokinins-Discovery, Biosynthesis and Physiological Role: Unit 5.1RimeiaNo ratings yet
- Phytochrome-Mediated Photomorphogenesis in Plants: Journal of Plant Biology, June 2007, 50 (3) : 230-240Document11 pagesPhytochrome-Mediated Photomorphogenesis in Plants: Journal of Plant Biology, June 2007, 50 (3) : 230-240RachanaNo ratings yet
- Amount of Chlorophyll in Five LeafDocument24 pagesAmount of Chlorophyll in Five LeafRakshitha MgowdaNo ratings yet
- General Biology Quarter 2 - Week 7 Name: Rona Mae N. Betita Grade and Section: STEM-12 Vygotsky Intext Questions 11.1Document4 pagesGeneral Biology Quarter 2 - Week 7 Name: Rona Mae N. Betita Grade and Section: STEM-12 Vygotsky Intext Questions 11.1Rona Mae BetitaNo ratings yet
- Phe and Cry PlantDocument2 pagesPhe and Cry PlantKeerthi KalesanNo ratings yet
- An Introduction To Photosynthesis PDFDocument15 pagesAn Introduction To Photosynthesis PDFNyimas Hoirunisa100% (2)
- Chloroplasts convert light energy through photosynthesisDocument10 pagesChloroplasts convert light energy through photosynthesisKhey AgamNo ratings yet
- Fluorescence Spectroscopy of Normal Mouse Skin Exposed To 5-Aminolaevulinic Acid and Red LightDocument9 pagesFluorescence Spectroscopy of Normal Mouse Skin Exposed To 5-Aminolaevulinic Acid and Red LightAditya Budi FauziNo ratings yet
- Genetic Analyses of Bolting in Bulb Onion Allium CepaDocument10 pagesGenetic Analyses of Bolting in Bulb Onion Allium CepaManuel VegasNo ratings yet
- Chloroplasts and the role of chlorophyll in photosynthesisDocument10 pagesChloroplasts and the role of chlorophyll in photosynthesisKhey AgamNo ratings yet
- Light Intensity and Photosynthetic Efficiency in Shade Plants andDocument6 pagesLight Intensity and Photosynthetic Efficiency in Shade Plants andShabrina AzzahraNo ratings yet
- lucock-2011-folic-acid-beyond-metabolismDocument12 pageslucock-2011-folic-acid-beyond-metabolismadinda.larastitiNo ratings yet
- Li Et Al 2011Document26 pagesLi Et Al 2011Natalia Andrea Nuñez ToledoNo ratings yet
- WUNSCH A - Skin Eye and BrainDocument4 pagesWUNSCH A - Skin Eye and BrainAlexander WunschNo ratings yet
- Photosynthesis in Higher PlantsDocument22 pagesPhotosynthesis in Higher PlantsSourav WiseNo ratings yet
- Basic Principles of Photodynamic TherapyDocument25 pagesBasic Principles of Photodynamic TherapyĐảm PhạmNo ratings yet
- Chapter 13 - Plant ResponseDocument36 pagesChapter 13 - Plant ResponseNURUL AINUN MUHAMMAD NORNo ratings yet
- Chapter PP.5 (Signals)Document68 pagesChapter PP.5 (Signals)NaHuynJungNo ratings yet
- Module 3 Sec 4Document18 pagesModule 3 Sec 4James MagnoNo ratings yet
- Sr AIIMS Super-60 Key & Solved QuestionsDocument15 pagesSr AIIMS Super-60 Key & Solved QuestionsAdithya BharadwajNo ratings yet
- X 24Document3 pagesX 24Hamdan FatahNo ratings yet
- MAMMALIADocument24 pagesMAMMALIAHamdan FatahNo ratings yet
- PISCESDocument27 pagesPISCESHamdan FatahNo ratings yet
- DaslingkuDocument8 pagesDaslingkuHamdan FatahNo ratings yet
- REPTILIADocument20 pagesREPTILIAHamdan FatahNo ratings yet
- Re 23Document3 pagesRe 23Hamdan FatahNo ratings yet
- 76ftsdvnfiZZZZZZZZZZZzzvshidfrthuj Iorklgfvijlsdfahudnvhfyufyuvngyersgzdhjzderhuzfhfhjbfhjbvhund Thought Leaders That Have A Sincere Interest in Implementing Fundamental Changes That Not OnlyDocument1 page76ftsdvnfiZZZZZZZZZZZzzvshidfrthuj Iorklgfvijlsdfahudnvhfyufyuvngyersgzdhjzderhuzfhfhjbfhjbvhund Thought Leaders That Have A Sincere Interest in Implementing Fundamental Changes That Not OnlyHamdan FatahNo ratings yet
- SEFALOKORDATADocument9 pagesSEFALOKORDATAHamdan FatahNo ratings yet
- Equired For The Gravity Response of The Arabidopsis RootDocument2 pagesEquired For The Gravity Response of The Arabidopsis RootHamdan FatahNo ratings yet
- Ntensified by Screening Within The OrganDocument3 pagesNtensified by Screening Within The OrganHamdan FatahNo ratings yet
- Phaseolus Vulgaris, Fully 30 Percent of The Osmotic Potential Change Can Be Accounted For by KDocument3 pagesPhaseolus Vulgaris, Fully 30 Percent of The Osmotic Potential Change Can Be Accounted For by KHamdan FatahNo ratings yet
- Epinasty and Thermonasty Are Examples of NasticDocument2 pagesEpinasty and Thermonasty Are Examples of NasticHamdan FatahNo ratings yet
- Transport Inhibitor NPADocument2 pagesTransport Inhibitor NPAHamdan FatahNo ratings yet
- Gravitropism Is Generally Absent in Plant Species ThatDocument3 pagesGravitropism Is Generally Absent in Plant Species ThatHamdan FatahNo ratings yet
- Unlike Most Other Environmental StimuliDocument3 pagesUnlike Most Other Environmental StimuliHamdan FatahNo ratings yet
- Root Gravitropism Occurs in Four PhasesDocument3 pagesRoot Gravitropism Occurs in Four PhasesHamdan FatahNo ratings yet
- Avena Coleoptiles. First Positive, First Negative, and Second Positive Curvatures Are IndicatedDocument3 pagesAvena Coleoptiles. First Positive, First Negative, and Second Positive Curvatures Are IndicatedHamdan FatahNo ratings yet
- The Hormone Concept in Plants: Chapter 18 / Hormones I: AuxinsDocument4 pagesThe Hormone Concept in Plants: Chapter 18 / Hormones I: AuxinsHamdan FatahNo ratings yet
- E Power of Movement Is Not Normally Associated WithDocument2 pagesE Power of Movement Is Not Normally Associated WithHamdan FatahNo ratings yet
- Array of Molecules With Auxin ActivityDocument3 pagesArray of Molecules With Auxin ActivityHamdan FatahNo ratings yet
- IAA Is Also Synthesized From Indole or FromDocument4 pagesIAA Is Also Synthesized From Indole or FromHamdan FatahNo ratings yet
- An Earlier ChapterDocument3 pagesAn Earlier ChapterHamdan FatahNo ratings yet
- Ve in An Entirely Inorganic EnvironmentDocument4 pagesVe in An Entirely Inorganic EnvironmentHamdan FatahNo ratings yet
- Casparian strip blocks apoplast pathwayDocument2 pagesCasparian strip blocks apoplast pathwayHamdan FatahNo ratings yet
- Cereale L. CV Musketeer) - Growth Irradiance Is inDocument5 pagesCereale L. CV Musketeer) - Growth Irradiance Is inHamdan FatahNo ratings yet
- And Can Dehydrate The PlantDocument1 pageAnd Can Dehydrate The PlantHamdan FatahNo ratings yet
- Complementary Chromatic Acclimation and Photoreversible Responses in CyanobacteriaDocument4 pagesComplementary Chromatic Acclimation and Photoreversible Responses in CyanobacteriaHamdan FatahNo ratings yet
- Plant respiration rates and temperature dependenceDocument4 pagesPlant respiration rates and temperature dependenceHamdan FatahNo ratings yet
- Chapter 14 / Acclimation To Environmental Stress: Quencher of Energy. Support For The Molecular SwitchDocument4 pagesChapter 14 / Acclimation To Environmental Stress: Quencher of Energy. Support For The Molecular SwitchHamdan FatahNo ratings yet
- Transport in Plants (Vascular Bundle)Document6 pagesTransport in Plants (Vascular Bundle)Ahmed Kaleem Khan NiaziNo ratings yet
- Symbiotic RelationshipsDocument2 pagesSymbiotic RelationshipsBianca BroccoNo ratings yet
- 26-03-2023 - Major Test - Part Test-4 - English - StudentDocument30 pages26-03-2023 - Major Test - Part Test-4 - English - StudentSuman LattaNo ratings yet
- Florida Wetland PlantsDocument613 pagesFlorida Wetland PlantsAllan EP100% (2)
- Sense OrgansDocument9 pagesSense OrgansKamarul ArifinNo ratings yet
- Revised Breeding of SpicesDocument69 pagesRevised Breeding of SpicesSavita BhoutekarNo ratings yet
- HorticultureDocument6 pagesHorticultureAlbyziaNo ratings yet
- Bhuira Jams-Inspiring StoryDocument4 pagesBhuira Jams-Inspiring StoryRjNo ratings yet
- Pedigree BlazeDocument1 pagePedigree Blazeapi-324296396No ratings yet
- Field identification of common weed speciesDocument4 pagesField identification of common weed speciesJAY MARK CUBAONo ratings yet
- Referat Engleza - OrangeDocument2 pagesReferat Engleza - OrangeRecalo PaulinaNo ratings yet
- (Kerry Hughes) The Incense BibleDocument241 pages(Kerry Hughes) The Incense BibleMar GomezNo ratings yet
- Explore Antoniadis Villa and Gardens AlexandriaDocument8 pagesExplore Antoniadis Villa and Gardens AlexandriaAbeer AdnanNo ratings yet
- Plant NutrientsDocument10 pagesPlant NutrientsAdrian GligaNo ratings yet
- Instant Download International Politics Power and Purpose in Global Affairs 3rd Edition Paul Danieri Test Bank PDF Full ChapterDocument33 pagesInstant Download International Politics Power and Purpose in Global Affairs 3rd Edition Paul Danieri Test Bank PDF Full Chaptereliasvykh6in8100% (8)
- JackfruitDocument9 pagesJackfruitHarry JonitzNo ratings yet
- Ethnomedicinal plants used by Deori tribes of AssamDocument5 pagesEthnomedicinal plants used by Deori tribes of Assammkshri_inNo ratings yet
- Walton-Vine Mealybug P. FicusDocument9 pagesWalton-Vine Mealybug P. Ficusbrkica2011No ratings yet
- The Drift Newsletter For Tatworth & Forton Edition 073Document8 pagesThe Drift Newsletter For Tatworth & Forton Edition 073russ_wardell6953No ratings yet
- Defects in TimberDocument29 pagesDefects in TimberPrashant Sunagar100% (1)
- Sexual Reproduction in Anthoceros:: Antheridium: Development and Structure of AntheridiumDocument4 pagesSexual Reproduction in Anthoceros:: Antheridium: Development and Structure of AntheridiumadekNo ratings yet
- Wrightia AntidysentericaDocument12 pagesWrightia AntidysentericaAllona Guinita Reasol100% (1)
- Alfalfa Variety Ratings 2020 GuideDocument8 pagesAlfalfa Variety Ratings 2020 Guidebruce leeNo ratings yet
- 12th Bio Botany Chapter Wise Questions English Medium PDFDocument12 pages12th Bio Botany Chapter Wise Questions English Medium PDFSalome ChristinaNo ratings yet
- Mould Contamination in Production of Semi-Hard CheeseDocument9 pagesMould Contamination in Production of Semi-Hard Cheesefdobonat613No ratings yet
- Unit 2 Test - CellsDocument4 pagesUnit 2 Test - CellsAndrae GenusNo ratings yet
- Bianca's Folder Types of FlowersDocument9 pagesBianca's Folder Types of FlowersCrow MinheeBgNo ratings yet
- Tugas Bahasa InggrisDocument3 pagesTugas Bahasa InggrisNXADARKSTAR NOXNo ratings yet
- CBSE Class 7 Science Sample Paper Set ADocument3 pagesCBSE Class 7 Science Sample Paper Set Asiba padhyNo ratings yet
- Effect of Plant Growth Regulators GA3 NAA and Water Stress On Leaf Area and Yield Attributes of WheatDocument6 pagesEffect of Plant Growth Regulators GA3 NAA and Water Stress On Leaf Area and Yield Attributes of WheatInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- When the Body Says No by Gabor Maté: Key Takeaways, Summary & AnalysisFrom EverandWhen the Body Says No by Gabor Maté: Key Takeaways, Summary & AnalysisRating: 3.5 out of 5 stars3.5/5 (2)
- Why We Die: The New Science of Aging and the Quest for ImmortalityFrom EverandWhy We Die: The New Science of Aging and the Quest for ImmortalityRating: 3.5 out of 5 stars3.5/5 (2)
- The Ancestor's Tale: A Pilgrimage to the Dawn of EvolutionFrom EverandThe Ancestor's Tale: A Pilgrimage to the Dawn of EvolutionRating: 4 out of 5 stars4/5 (811)
- 10% Human: How Your Body's Microbes Hold the Key to Health and HappinessFrom Everand10% Human: How Your Body's Microbes Hold the Key to Health and HappinessRating: 4 out of 5 stars4/5 (33)
- All That Remains: A Renowned Forensic Scientist on Death, Mortality, and Solving CrimesFrom EverandAll That Remains: A Renowned Forensic Scientist on Death, Mortality, and Solving CrimesRating: 4.5 out of 5 stars4.5/5 (397)
- Human: The Science Behind What Makes Your Brain UniqueFrom EverandHuman: The Science Behind What Makes Your Brain UniqueRating: 3.5 out of 5 stars3.5/5 (38)
- Tales from Both Sides of the Brain: A Life in NeuroscienceFrom EverandTales from Both Sides of the Brain: A Life in NeuroscienceRating: 3 out of 5 stars3/5 (18)
- Crypt: Life, Death and Disease in the Middle Ages and BeyondFrom EverandCrypt: Life, Death and Disease in the Middle Ages and BeyondRating: 4 out of 5 stars4/5 (3)
- Who's in Charge?: Free Will and the Science of the BrainFrom EverandWho's in Charge?: Free Will and the Science of the BrainRating: 4 out of 5 stars4/5 (65)
- Good Without God: What a Billion Nonreligious People Do BelieveFrom EverandGood Without God: What a Billion Nonreligious People Do BelieveRating: 4 out of 5 stars4/5 (66)
- The Consciousness Instinct: Unraveling the Mystery of How the Brain Makes the MindFrom EverandThe Consciousness Instinct: Unraveling the Mystery of How the Brain Makes the MindRating: 4.5 out of 5 stars4.5/5 (93)
- This Is Your Brain On Parasites: How Tiny Creatures Manipulate Our Behavior and Shape SocietyFrom EverandThis Is Your Brain On Parasites: How Tiny Creatures Manipulate Our Behavior and Shape SocietyRating: 3.5 out of 5 stars3.5/5 (31)
- Undeniable: How Biology Confirms Our Intuition That Life Is DesignedFrom EverandUndeniable: How Biology Confirms Our Intuition That Life Is DesignedRating: 4 out of 5 stars4/5 (11)
- Buddha's Brain: The Practical Neuroscience of Happiness, Love & WisdomFrom EverandBuddha's Brain: The Practical Neuroscience of Happiness, Love & WisdomRating: 4 out of 5 stars4/5 (215)
- The Lives of Bees: The Untold Story of the Honey Bee in the WildFrom EverandThe Lives of Bees: The Untold Story of the Honey Bee in the WildRating: 4.5 out of 5 stars4.5/5 (44)
- The Molecule of More: How a Single Chemical in Your Brain Drives Love, Sex, and Creativity--and Will Determine the Fate of the Human RaceFrom EverandThe Molecule of More: How a Single Chemical in Your Brain Drives Love, Sex, and Creativity--and Will Determine the Fate of the Human RaceRating: 4.5 out of 5 stars4.5/5 (515)
- The Other Side of Normal: How Biology Is Providing the Clues to Unlock the Secrets of Normal and Abnormal BehaviorFrom EverandThe Other Side of Normal: How Biology Is Providing the Clues to Unlock the Secrets of Normal and Abnormal BehaviorNo ratings yet
- Eels: An Exploration, from New Zealand to the Sargasso, of the World's Most Mysterious FishFrom EverandEels: An Exploration, from New Zealand to the Sargasso, of the World's Most Mysterious FishRating: 4 out of 5 stars4/5 (30)
- A Brief History of Intelligence: Evolution, AI, and the Five Breakthroughs That Made Our BrainsFrom EverandA Brief History of Intelligence: Evolution, AI, and the Five Breakthroughs That Made Our BrainsRating: 4.5 out of 5 stars4.5/5 (4)
- Superlative: The Biology of ExtremesFrom EverandSuperlative: The Biology of ExtremesRating: 4.5 out of 5 stars4.5/5 (51)
- Wayfinding: The Science and Mystery of How Humans Navigate the WorldFrom EverandWayfinding: The Science and Mystery of How Humans Navigate the WorldRating: 4.5 out of 5 stars4.5/5 (18)
- The Second Brain: A Groundbreaking New Understanding of Nervous Disorders of the Stomach and IntestineFrom EverandThe Second Brain: A Groundbreaking New Understanding of Nervous Disorders of the Stomach and IntestineRating: 4 out of 5 stars4/5 (17)
- Younger for Life: Feel Great and Look Your Best with the New Science of AutojuvenationFrom EverandYounger for Life: Feel Great and Look Your Best with the New Science of AutojuvenationRating: 4 out of 5 stars4/5 (1)
- Darwin's Dangerous Idea: Evolution and the Meaning of LifeFrom EverandDarwin's Dangerous Idea: Evolution and the Meaning of LifeRating: 4 out of 5 stars4/5 (523)
- Fearfully and Wonderfully: The Marvel of Bearing God's ImageFrom EverandFearfully and Wonderfully: The Marvel of Bearing God's ImageRating: 5 out of 5 stars5/5 (40)
- The Dragons of Eden: Speculations on the Evolution of Human IntelligenceFrom EverandThe Dragons of Eden: Speculations on the Evolution of Human IntelligenceRating: 4 out of 5 stars4/5 (632)