Professional Documents
Culture Documents
Cannizzaro Reaction Products
Uploaded by
SherlHolmesOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Cannizzaro Reaction Products
Uploaded by
SherlHolmesCopyright:
Available Formats
Chem 35.
1 – TEG
Espiritu, Walter Aljhon March 18, 2014
Silong, Rafaelle
Tumimbang, Glenn Vincent
I. Abstract
Chemical reactions vary and are different in nature. One of which is the oxidation-reduction
reaction, where a substance is being oxidized while another is being reduced. Organic compounds
behave the same way. In organic chemistry, redox reactions are focused on the carbon atom.
Oxidation of carbon means that there is an increase in number of carbon-oxygen bonds or a
decrease in number of carbon-hydrogen bonds; and reduction is the total opposite. In this
experiment, a reaction involving both oxidation and reduction on benzaldehyde will be observed
called Cannizzaro reaction.
II. Keywords: oxidation-reduction reaction, Cannizzaro reaction, base-catalyzed reaction
III. Introduction assemble the apparatus for heating under
One of the notable organic reactions reflux.
in chemistry is the Cannizzaro reaction,
Major Step 2: Reaction Process
named after its discoverer, Stanislao
Cannizzaro. It is limited to aldehydes lacking First, place the base of the 25 mL
alpha hydrogen centers. It produces an flask in a heating bath, preheated to ~75
alcohol and carboxylic acid at minimal degree celsius, and heat the reaction mixture
amounts, accounting only 50% to the yield with stirring for 1.5 hour. Then, remove the
even under ideal conditions. It is a base- flask from the bath and cool the mixture to
catalyzed reaction, which begins with the room temperature. Then, add 2.5 mL of water
nucleophilic attack of OH- on the carbonyl and transfer the mixture in a small separatory
center. The resulting anion attacks another funnel. Extract the aqueous mixture with 3
molecule of aldehyde, transferring a hydride separate 1.0 mL portions of dichloromethane
ion. In the final step, the acid and alkoxide and collect the organic layer. Combine all the
ions formed exchange a proton. All in all, the organic extracts in a centrifuge tube. Save
discovery of Cannizzaro has proven to be one both prgamoc and aqueous layers.
of the most important reactions in synthetic Major Step 3: Isolation and Purification of
organic chemistry. Products
IV. Methodology Wash the combined dichloromethane
extracts with two separate 0.5 mL portions of
Major Step 1: Cannizzaro Reaction Setup saturated aqueous sodium chloride. Then,
First, dissolve 1.0g of KOH in 1 mL transfer the dichloromethane layer to a dry
distilled H2O in a test tube. Then, weigh 200 centrifuge tube and add several tips full of
mg of 4-chlorobenzaldehyde and transfer it to anhydrous sodium sulfate. Swirl the tube
a 25 mL distilling flask, add 0.5 mL of occasionally for ~10-15 minutes. Using a
methanol and stir to dissolve the 4- small filter pipette, transfer the dried solution
chlorobenzaldehyde. Then, using a Pasteur in a clean 5 mL test tube and rinse the sodium
pipette, transfer 0.3 mL of the 50% aqueous sulfate with ~0.2 mL dichloromethane and
KOH sol’n to the flask (note: do not get any of transfer the rinse to the test tube. Remove the
the solution on the ground glass joint). Finally, dichloromethane by steam bath. The crude for
4-chlorobenzylalcohol may be recrystallized
Expt. 7: Base-Catalyzed Oxidation – Reduction of Aldehydes by the Cannizzaro Reaction Page 1
Chem 35.1 – TEG
from 4% acetone in hexane in a Craug tube benzaldehyde, generating a tetrahedral
and air dry the product. Then cool the intermediate. This tetrahedral intermediate
aqueous phase saved from the initial then functions as a hydride reducing agent by
dichloromethane extraction in an ice-water giving off a hydride ion to another molecule of
bath, acidify the solution by slowly adding benzaldehyde. In this manner, one molecule
0.5mL of concentrated HCl. Finally, collect the of benzaldehyde is oxidized (by the hydroxide
precipitate via vacuum filtration and wash the ion) and forms a benzoic acid molecule; and
filter cake with ~1-2 mL water and collect the another is reduced (by the hydride ion) and
air dry product, the crude 4-chlorobenzoic acid forms benzyl alcohol molecule.
may be recrystallized from methanol in a
Craig tube. The products are then separated by
liquid-liquid extraction through their distinctive
properties. Benzoic acid is polar, which make
V. Results and Discussion
it soluble to polar solvents such as water by
generating ions.However, benzyl alcohol is
The general equation of the reaction is:
nonpolar, making it soluble to nonpolar
solvents such as dichloromethane.
VI. Guide Questions
1. Write the mechanism for the reaction.
Figure 1. Cannizzaro Reaction of benzaldehyde
In this chemical reaction, two moles of
benzaldehyde are treated hydroxide, a
reaction in which one mole of benzaldehyde is
oxidized to form benzoic acid while the other
one is reduced to form benzyl alcohol. 2. After separating the organic and aqueous
layers, the dichloromethane layer is first
The mechanism is believed to be as follows: washed with two portions of saturated
aqueous sodium chloride prior to the drying
with anhydrous sodium sulfate. What is the
purpose of these washes?
The saturated aqueous sodium
chloride will wash away traces of water and
KOH in the dichloromethane, it will also
decrease the benzyl alcohol’s solubility in
water so the washings will not wash away the
product. The sodium sulfate also removes
traces of water from the solvent so that it will
evaporate and the product won’t be sticky wet
in the end.
Figure 2. Cannizzaro Reaction Mechanism 3. The Cannizzaro reaction much more slowly
in dilute than in concentrated KOH solution.
A hydroxide ion serves as a Explain.
nucleophile and attacks the carbonyl group of
Expt. 7: Base-Catalyzed Oxidation – Reduction of Aldehydes by the Cannizzaro Reaction Page 2
Chem 35.1 – TEG
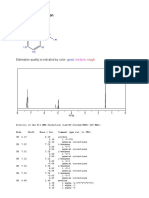
The overall Cannizzaro reaction is indicating the presence of aldehyde moiety. In
third order kinetics, that is, rate = H-NMR, a strong stretch is observed at 10.5
k[RCHO]2[OH-]. It is second order on the ppm, indicating an aldehydic moiety.
aldehyde and first order on the hydroxide. However, in IR spectra of benzoic
Dilute solutions provide less concentrations of acid, a broad peak at 3400cm- is observed,
OH- ions to the reaction, and therefore, the which confirms a hydroxyl group; and a
reaction proceeds much slower than in carbonyl peak at 1750 cm-. On its H-NMR
concentrated solutions. peak, it also has a chemical stretch at 10 ppm,
indicating a carboxylic acid moiety.
4. A carbon-based hydride reducing agent in On the other hand, the IR spectra of
biological systems is nicotine amine adenine benzyl alcohol, there is also a broad band at
dinucleotide, NADH, which reduces carbonyl 3300 cm- to 3600 cm-, confirming the
compounds by a mechanism similar to the presence of an alcohol. Also, there is a peak
Cannizzaro reaction. Using curved arrows to at 1450 cm-, which is due to an aliphatic
symbolize the flow of electrons, write the carbon bond. On H-NMR, there is a peak at 4
mechanism for the hydride transfer from ppm, indicating a C-O bond and at 2.5 ppm,
NADH to a carbonyl compound. Your which depicts the presence of an allylic
mechanism should show how NADH becomes hydrogen.
NAD+. What is the driving force for this
reaction? VII. Conclusion
In the experiment conducted to
observe Cannizzaro reaction, it was observed
that the reaction is done under basic
conditions. The base acts as the nucleophile
that will attack the carbonyl group, resulting
into benzoic acid (carboxylic acid) and benzyl
alcohol (alcohol). The order of kinetics also
affect the speed of the reaction, that is, at
higher base concentrations, it proceeds faster
than in dilute concentrations of base.
VIII. References
5. Discuss the differences observed in the IR
and NMR spectra of benzaldehyde, benzyl Carey, F. (2006). Organic Chemistry,
alcohol, and benzoic acid that are consistent 6th Edition
with the formation of the two products from
benzaldehyde by the Cannizzaro reaction. Klein, D. (2012).Organic Chemistry.
They all have overtones at 1650 to
2000 cm -, indicating the presence of an Cannizzaro reaction. Retrieved from:
aromatic ring, as well as peaks at 1600-1700 http://www.britannica.com/EBchecked/
cm-. On the H-NMR spectra, they all have topic/92753/Cannizzaro-reaction
peaks at 7 to 7.5 ppm, indicating the presence
of a hydrogen in aromatic ring.
In the IR spectra of benzaldehyde,
there is a strong peak at 1750 cm -, indicating
a carbonyl moiety and peaks at 2750 cm-,
Expt. 7: Base-Catalyzed Oxidation – Reduction of Aldehydes by the Cannizzaro Reaction Page 3
Chem 35.1 – TEG
I hereby certify that I substantially contribute
to this report.
_____________________
Walter Aljhon Espiritu
_____________________
Rafaelle Silong
_____________________
Glenn Vincent Tumimbang
Expt. 7: Base-Catalyzed Oxidation – Reduction of Aldehydes by the Cannizzaro Reaction Page 4
You might also like
- 6&7. Synthesis, Characterisation and Stability of An Interhalogen CompoundDocument4 pages6&7. Synthesis, Characterisation and Stability of An Interhalogen Compoundstudent_4_evaNo ratings yet
- Synthesis of Isobutyl AcetateDocument7 pagesSynthesis of Isobutyl AcetateRandy DavenportNo ratings yet
- P6-The Balance Between Enthalpy and EntropyDocument2 pagesP6-The Balance Between Enthalpy and Entropyjuanbuuren serna victoriaNo ratings yet
- Oxidation of Benzoin To BenzilDocument4 pagesOxidation of Benzoin To BenzilJonathan Shao100% (1)
- CHEM35.1 E7 Cannizzaro ReactionDocument4 pagesCHEM35.1 E7 Cannizzaro ReactionGlenn Vincent Tumimbang100% (7)
- Thermodynamics of The Ionization ofDocument2 pagesThermodynamics of The Ionization ofNicolás PeñaNo ratings yet
- M. Curini, F. Epifano, S. Genovese, M.C. Marcotullio, O. Rosati. Ytterbium Triflate-Promoted TandemDocument3 pagesM. Curini, F. Epifano, S. Genovese, M.C. Marcotullio, O. Rosati. Ytterbium Triflate-Promoted TandemMariel MedinaNo ratings yet
- Vogel's Textbook of Practical Organic Chemistry 5th Ed.Document1,540 pagesVogel's Textbook of Practical Organic Chemistry 5th Ed.MohanRNadigarNo ratings yet
- Functional Group TestsDocument2 pagesFunctional Group TestsSalman HusainNo ratings yet
- 302 338Document37 pages302 338Putri KartiniNo ratings yet
- Experimental Organic Chemistry Post-Lab 1 Photodimerization of BenzophenoneDocument5 pagesExperimental Organic Chemistry Post-Lab 1 Photodimerization of Benzophenoneapi-235187189No ratings yet
- Diapositiva de Recursos Humanos 1: Total Synthesis of CoprinolDocument22 pagesDiapositiva de Recursos Humanos 1: Total Synthesis of CoprinolAngelOrtegaZarcoNo ratings yet
- MPN TableDocument6 pagesMPN TableayachandraNo ratings yet
- Carboxylic Acids Carboxylic Acids Carboxylic Acids Carboxylic AcidsDocument25 pagesCarboxylic Acids Carboxylic Acids Carboxylic Acids Carboxylic AcidsOhmark VeloriaNo ratings yet
- 1-Phenylethanol H-NMR PDFDocument2 pages1-Phenylethanol H-NMR PDFkobir960% (1)
- Exp 3-Reduction of Cyclohexanone With Sodium BorohydrideDocument11 pagesExp 3-Reduction of Cyclohexanone With Sodium Borohydrideakuserai100% (3)
- Ch16 Benzene ChemistryDocument16 pagesCh16 Benzene ChemistrySherlHolmesNo ratings yet
- Air Stripping of AcetoneDocument19 pagesAir Stripping of AcetoneGabe100% (1)
- Thermodynamic and Kinetic Control of ReactionsDocument4 pagesThermodynamic and Kinetic Control of ReactionsjeeadvanceNo ratings yet
- A Stopped-Flow Kinetics Experiment For Advanced Undergraduate Laboratories: Formation of Iron (III) ThiocyanateDocument4 pagesA Stopped-Flow Kinetics Experiment For Advanced Undergraduate Laboratories: Formation of Iron (III) ThiocyanateJuraj AhelNo ratings yet
- Practica 1 ElectroDocument14 pagesPractica 1 ElectroAnna KoronaNo ratings yet
- Formal Diazo Dye ReportDocument9 pagesFormal Diazo Dye Reportbig504075% (4)
- Fisicoquimica CCCCDocument24 pagesFisicoquimica CCCCCarlos de la CruzNo ratings yet
- Conceptos Basicos CronocoulomDocument3 pagesConceptos Basicos Cronocoulombettypaz89No ratings yet
- Tablas Alcoholimetricas BDocument3 pagesTablas Alcoholimetricas Bfgranobles100% (1)
- Sintesis de 2-FenilindolDocument4 pagesSintesis de 2-FenilindolRodrigo WolfordNo ratings yet
- Ejercicios de AlacanosDocument16 pagesEjercicios de AlacanosRicardo Alcántara ReyesNo ratings yet
- College Chemistry Tutorial on Reaction RatesDocument7 pagesCollege Chemistry Tutorial on Reaction RatesbebsybiswezNo ratings yet
- Thread-based analytical devicesDocument12 pagesThread-based analytical devicesMichelle TagalogNo ratings yet
- Separate Mixture of Benzoic Acid, Naphthalene and AnilineDocument8 pagesSeparate Mixture of Benzoic Acid, Naphthalene and Anilinejarissa bannerNo ratings yet
- The Acid Base Titration of A Very Weak Acid: Boric Acid: M. Celeste C. Azevedo and Ana M. V. CavaleiroDocument4 pagesThe Acid Base Titration of A Very Weak Acid: Boric Acid: M. Celeste C. Azevedo and Ana M. V. CavaleiroAitor PastorNo ratings yet
- NMR Practice Problems SolvedDocument9 pagesNMR Practice Problems SolvedVivek AgrahariNo ratings yet
- Taller - Módulo 2 - Tema 3 PDFDocument3 pagesTaller - Módulo 2 - Tema 3 PDFPUFFWHEELS SASNo ratings yet
- Exp 5Document8 pagesExp 5Azli AzmanNo ratings yet
- Practica 8 AdsorcionDocument7 pagesPractica 8 AdsorcionMónica MedinaNo ratings yet
- Solutions CH 04Document10 pagesSolutions CH 04Emily EstrellaNo ratings yet
- Amines Via Hofmann, Curtius, Schmidt, and Lossen - Comprehensive Organic Synthesis PDFDocument34 pagesAmines Via Hofmann, Curtius, Schmidt, and Lossen - Comprehensive Organic Synthesis PDFpcube100% (1)
- Resolução Lehninger - Capítulo 10Document8 pagesResolução Lehninger - Capítulo 10hugo0% (1)
- Sintesis de ParacetamolDocument10 pagesSintesis de ParacetamolMaria Camila Alvarado MartinezNo ratings yet
- JACS Article Examines Cannizzaro Reaction MechanismDocument8 pagesJACS Article Examines Cannizzaro Reaction MechanismDaniel LieNo ratings yet
- Diels Alder LabDocument8 pagesDiels Alder Labfatevilcow0% (1)
- Synthesis and Determination of A Cobalt Bromide Ammine Complex - YCZengDocument15 pagesSynthesis and Determination of A Cobalt Bromide Ammine Complex - YCZengJack Zeng100% (1)
- Woodwardrulestructural Analysis (Application of Woodward-Fieser Rules) of SpectrosDocument20 pagesWoodwardrulestructural Analysis (Application of Woodward-Fieser Rules) of Spectrosahmadhelmiase7621100% (3)
- Membrane Conference ProblemsDocument6 pagesMembrane Conference ProblemsGuhanNo ratings yet
- Studies On Schiff Base Transition Metal ComplexesDocument10 pagesStudies On Schiff Base Transition Metal ComplexesDebraj Dhar PurkayasthaNo ratings yet
- Chromatography: The Separation Technique of The 20th CenturyDocument11 pagesChromatography: The Separation Technique of The 20th CenturyAnonymous qOxE6C7kNo ratings yet
- Qoiii I Class1Document25 pagesQoiii I Class1valentina viveros riosNo ratings yet
- Isolation of Invertase Formal ReportDocument3 pagesIsolation of Invertase Formal ReportGabbySantosNo ratings yet
- Carbonic Anhydrase (CA) Pinki..Document13 pagesCarbonic Anhydrase (CA) Pinki..Riniya NajeebNo ratings yet
- Entropy Tutorial + SolutionsDocument2 pagesEntropy Tutorial + SolutionsAnonymous 8SemaIglBvNo ratings yet
- Nitropentaamminecobalt (III) Chloride - Wikipedia, The Free EncyclopediaDocument2 pagesNitropentaamminecobalt (III) Chloride - Wikipedia, The Free EncyclopediaDanielAminFemminellaNo ratings yet
- Application of ANOVADocument19 pagesApplication of ANOVAabhay_prakash_ranjan100% (2)
- Sintesis de Metronidazol A Partir de La Etilendiamina PDFDocument3 pagesSintesis de Metronidazol A Partir de La Etilendiamina PDFAstridChoqueNo ratings yet
- Sweet Indigestion: A Directed Case Study On Carbohydrates: Part I-Of Cows and CarbsDocument3 pagesSweet Indigestion: A Directed Case Study On Carbohydrates: Part I-Of Cows and CarbsLillian MuellerNo ratings yet
- Preparation of Acids and BasesDocument4 pagesPreparation of Acids and BasesAbhishek RajNo ratings yet
- Synthesis of LidocaineDocument2 pagesSynthesis of LidocaineRoldan SardonaNo ratings yet
- Espectrofotómetro Ftir C103-E076a Iraffinity-1Document28 pagesEspectrofotómetro Ftir C103-E076a Iraffinity-1Gustavo RuizNo ratings yet
- Diazotization, KF TitrationsDocument15 pagesDiazotization, KF Titrationsiasnit100% (2)
- Cannizaro ReactionDocument13 pagesCannizaro Reactionhussein alnasryNo ratings yet
- Extraction PDFDocument19 pagesExtraction PDFMohamed DahmaneNo ratings yet
- Experiment 5: The Preparation of 1-Bromobutane From 1-ButanolDocument6 pagesExperiment 5: The Preparation of 1-Bromobutane From 1-ButanolNoor Azizah ZakariaNo ratings yet
- Determining The Density of Some Recyclable PlasticsDocument9 pagesDetermining The Density of Some Recyclable PlasticsSherlHolmesNo ratings yet
- Bottle), Is A Flask With A Close-Fitting Ground Glass StopperDocument4 pagesBottle), Is A Flask With A Close-Fitting Ground Glass StopperSherlHolmesNo ratings yet
- Qualitative Analysis of Organic CompoundsDocument22 pagesQualitative Analysis of Organic CompoundsKarisseGarciaNo ratings yet
- 36.5 - Gravimetric AnalysisDocument1 page36.5 - Gravimetric AnalysisAnnaReyesNo ratings yet
- 11 GravimetricDeterm PDFDocument2 pages11 GravimetricDeterm PDFMilton Dela RosaNo ratings yet
- "Instruments of Neocolonialism" in The Philippine and The United States - A Study of NeocolonialismDocument22 pages"Instruments of Neocolonialism" in The Philippine and The United States - A Study of NeocolonialismSherlHolmesNo ratings yet
- Presentation Notes - The Filipino Struggle For IndependenceDocument3 pagesPresentation Notes - The Filipino Struggle For IndependenceSherlHolmesNo ratings yet
- Le Chatelier's Principle - Chromate-Dichromate - C12!4!07Document5 pagesLe Chatelier's Principle - Chromate-Dichromate - C12!4!07John Michael Maulas Vargas100% (1)
- Castles - New MigrationsDocument13 pagesCastles - New MigrationsSherlHolmesNo ratings yet
- Statistics and Probability: Bill Thaddeus PadasasDocument102 pagesStatistics and Probability: Bill Thaddeus PadasasSherlHolmesNo ratings yet
- Mohrig Qual ProcDocument54 pagesMohrig Qual ProcSherlHolmesNo ratings yet
- AMASDocument10 pagesAMASSherlHolmesNo ratings yet
- The Value Proposition Canvas PDFDocument1 pageThe Value Proposition Canvas PDFSherlHolmesNo ratings yet
- Beilock & Maloney (2015) Math AnxietyDocument9 pagesBeilock & Maloney (2015) Math AnxietySherlHolmesNo ratings yet
- Chemistry Theory Sample Paper (2022-23) on Aldehydes, Ketones and Carboxylic AcidsDocument8 pagesChemistry Theory Sample Paper (2022-23) on Aldehydes, Ketones and Carboxylic AcidsJems Chaudhary100% (1)
- Name Reactions-IDocument33 pagesName Reactions-ISatya KamNo ratings yet
- Named ReactionsDocument15 pagesNamed ReactionsSony mulgundNo ratings yet
- 306 Cannizzaro ReactionDocument9 pages306 Cannizzaro Reactionayesha sanaNo ratings yet
- Why Do We Use A Dilute Base in An Aldol Condensation Reaction, But A Concentrated Base in The Cannizzaro Reaction - QuoraDocument2 pagesWhy Do We Use A Dilute Base in An Aldol Condensation Reaction, But A Concentrated Base in The Cannizzaro Reaction - QuoraShreeyesh BiswalNo ratings yet
- Aldol CondensationDocument71 pagesAldol CondensationJeyanthiNo ratings yet
- Alde & Ket-6&7Document15 pagesAlde & Ket-6&7ayesha sheikhNo ratings yet
- Cannizzaro Reaction ProductsDocument4 pagesCannizzaro Reaction ProductsSherlHolmesNo ratings yet
- Aldehydes and Ketones Structure and ReactionsDocument104 pagesAldehydes and Ketones Structure and ReactionsCharin Kadian75% (4)
- Koskinen2015 PDFDocument200 pagesKoskinen2015 PDFNastase DamianNo ratings yet
- Organic Ncert SolutionsDocument50 pagesOrganic Ncert SolutionsManasvi MehraNo ratings yet
- 5358chemistry Class XII Question Bank (First Part) (2022-23)Document27 pages5358chemistry Class XII Question Bank (First Part) (2022-23)Jiya PandeyNo ratings yet
- Aldehydes & Ketones (Booklet-2Document15 pagesAldehydes & Ketones (Booklet-2kraken monsterNo ratings yet
- Cannizaro ReactionsDocument3 pagesCannizaro ReactionsvrchemieNo ratings yet
- 01 - Carbonyl Compound (Aldehyde & Ketone) (Theory) Module-5Document18 pages01 - Carbonyl Compound (Aldehyde & Ketone) (Theory) Module-5Raju SinghNo ratings yet
- Aiits 1416 PT III Jeem Jeea Advanced Paper 2 Questions PaperDocument26 pagesAiits 1416 PT III Jeem Jeea Advanced Paper 2 Questions PaperRavi Kumar Varma100% (1)
- Chapter 12 Mechanism of Reaction: Aldol CondensationDocument17 pagesChapter 12 Mechanism of Reaction: Aldol CondensationTiya KapoorNo ratings yet
- Free Live Online Master Classes for StudentsDocument39 pagesFree Live Online Master Classes for StudentsRohan Agrawal 961No ratings yet
- Jee-Aldehyde Ketone & Carboxylic Acid Chem XiiDocument72 pagesJee-Aldehyde Ketone & Carboxylic Acid Chem Xiiswastik aroraNo ratings yet
- Reduction of Aldehydes and Ketones: Hydrogen Transfer Redox ReactionsDocument20 pagesReduction of Aldehydes and Ketones: Hydrogen Transfer Redox ReactionsDhewi LayNo ratings yet
- Applied Chemistry (MS-133) : Cannizzaro Reaction With Complete MechanismDocument5 pagesApplied Chemistry (MS-133) : Cannizzaro Reaction With Complete MechanismHammad Raza 362-FET/BTECHME/F18No ratings yet
- McMurry 7e Ch19-23 Notes 5-29-07Document55 pagesMcMurry 7e Ch19-23 Notes 5-29-07Kay BradyNo ratings yet
- CH 11. Aldehydes - Ketones Tatva (NM)Document48 pagesCH 11. Aldehydes - Ketones Tatva (NM)chandan mallikNo ratings yet
- NCERT Solutions Class 12 Chemistry Chapter 12 Aldehydes, Ketones and Carboxylic AcidsDocument41 pagesNCERT Solutions Class 12 Chemistry Chapter 12 Aldehydes, Ketones and Carboxylic AcidsVidyakulNo ratings yet
- BaseCatalyzedOxidation ReductionofAldehydesbytheCannizzaroReactionDocument4 pagesBaseCatalyzedOxidation ReductionofAldehydesbytheCannizzaroReactionMike TranNo ratings yet
- XII Chemistry - Frequently Asked Question Bank PDFDocument175 pagesXII Chemistry - Frequently Asked Question Bank PDFYASH PATELNo ratings yet
- GPAT 2011 Answer KeyDocument19 pagesGPAT 2011 Answer KeyDeepak Kumar MauryaNo ratings yet
- 11 Aldehyde Ketone and C-Acids 19-02-2022 MCQ With SolutionsDocument26 pages11 Aldehyde Ketone and C-Acids 19-02-2022 MCQ With SolutionsANIKET BATTINWARNo ratings yet
- Experiment 60 - Aldehyde DisproportionationDocument4 pagesExperiment 60 - Aldehyde DisproportionationJoseph PhamNo ratings yet
- Aldehyde KetonesDocument64 pagesAldehyde KetonesDebayanbasu.juNo ratings yet