Professional Documents
Culture Documents
Crossm: Diagnostic Mycology: Xtreme Challenges
Uploaded by
rehanaOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Crossm: Diagnostic Mycology: Xtreme Challenges
Uploaded by
rehanaCopyright:
Available Formats
MINIREVIEW
crossm
Diagnostic Mycology: Xtreme Challenges
Brian L. Wickes,a Anna M. Romanellib
a
Department of Microbiology, Immunology, and Molecular Genetics, University of Texas Health Science Center at San Antonio, San Antonio, Texas, USA
b Department of Pathology and Laboratory Medicine, University of California, Davis Health System, Sacramento, California, USA
Downloaded from http://jcm.asm.org/ on March 22, 2021 by guest
ABSTRACT Developing any diagnostic assay that receives United States Food and
Drug Administration (FDA) approval can be a slow and difficult process. FDA-
approved assays for fungal diagnosis are generally few in number and are focused
mainly on diagnosing candidiasis, which is caused by several species of Candida, in
addition to a limited number of systemic mycotic agents. While all microbial diag-
nostic assays face challenges before they are FDA approved and reach the market,
there are a number of challenges to fungal diagnostic assay development that have
been difficult hurdles to overcome. These hurdles include template preparation, fun-
gal morphology, how many fungi should be identified in a single assay (scope), tax-
onomy and nomenclature, discriminating colonizers from invasive infection, combin-
ing identification with antifungal susceptibility, and navigating the administrative
hurdles required to integrate an assay into a clinical laboratory. Some of these chal-
lenges are easier to overcome than others, but all seem to be particularly difficult
for fungal diagnostic assays.
KEYWORDS antifungal, molds, mycoses, nomenclature, susceptibility testing, yeast
T he challenges facing diagnostic microbiology, and medical mycology in particular,
continue to grow in spite of newer technologies being employed to develop
diagnostic assays. Multiple factors have contributed to increase the number and
difficulty of challenges to the clinical microbiologist charged with identifying fungal
pathogens from patients. Most of the current challenges to the microbiology laboratory
are due to new populations of patients that are more immunocompromised for a
longer period of time during the course of their illness. These patients are increasingly
predisposed to becoming infected by a wider variety of fungi, which remain major
causes of morbidity and mortality. In spite of new diagnostic technologies entering the
clinical microbiology laboratory, they have been slow to be approved for diagnosis of
fungi. This deficiency has contributed to delayed diagnosis, which is a known risk factor
for increased morbidity and mortality in many systemic mycoses (1).
Part of the delayed diagnosis problem in clinical mycology is the diverse and
growing spectrum of fungi capable of causing disease. Many of these emerging fungal
pathogens have not been seen at a high enough frequency to be considered early on
in the course of infection, and in many cases, if a fungus is determined to be the Citation Wickes BL, Romanelli AM. 2020.
etiologic agent, it often cannot be identified to the species level. Consequently, rarely Diagnostic mycology: xtreme challenges. J Clin
seen or unusual fungi often need to be sent out to reference laboratories for proper Microbiol 58:e01345-19. https://doi.org/10
.1128/JCM.01345-19.
identification. In fact, the Centers for Disease Control and Prevention has played a
Editor Colleen Suzanne Kraft, Emory University
crucial role for multiple identifications of rare fungal pathogens within just the last few Copyright © 2020 American Society for
years (2–4). Other fungi, such as the recently emergent Candida auris, are close relatives Microbiology. All Rights Reserved.
of more commonly seen fungi and may not be discriminated from these more common Address correspondence to Brian L. Wickes,
relatives (5). In fact, emergent mycoses have proven to pose consistent diagnostic Wickes@uthscsa.edu.
Accepted manuscript posted online 29
challenges for many decades, due in part to changing patient susceptibilities.
January 2020
In this review, we summarize some of the more problematic challenges to devel- Published 25 March 2020
oping new molecular diagnostic assays. We have focused on molecular assays that
April 2020 Volume 58 Issue 4 e01345-19 Journal of Clinical Microbiology jcm.asm.org 1
Minireview Journal of Clinical Microbiology
target nucleic acids and proteins but recognize that assays directed toward metabo-
lites, antigens, or antibodies have their own challenges. Still, other assays that are more
traditional such as biochemical, serological, or morphological also have challenges and
have been reviewed elsewhere (6–9). Our perspective is from both a basic research
view, which in and of itself is often not encumbered by how difficult or expensive a new
and sensitive technique could be to implement in a clinical microbiology laboratory,
and a clinical perspective, which has to balance cost, ease of use, and consistency with
how sensitive and accurate an assay could be in the real world of a clinical microbiology
laboratory. There are numerous critical and even required characteristics of an assay
that do not need to be elaborated on here since they are goals of virtually any assay
and remain important challenges to any assay development. These goals include high
precision and accuracy, wide reportable range, high analytical sensitivity, rapid turn-
around, instrument and assay cost, instrument footprint, and high positive and nega-
Downloaded from http://jcm.asm.org/ on March 22, 2021 by guest
tive predictive values. For fungi, there are additional challenges, which we feel are
extreme and need to be considered for any new fungal diagnostic.
TEMPLATE PREPARATION
In spite of a diverse approach to fungal diagnostic assay development, one of the
most frequently overlooked components of any assay is template or target preparation.
Fungi are unusual compared to other microbes because of their rigid cell walls, which
necessitate preliminary preparation steps that require removal during the sample
preparation process. These steps can be chemical, physical, or enzymatic, and each of
these strategies has numerous methods. Pure cultures are much easier to process
because 100% of the assay target will be fungal derived, and there generally is no
limitation on the starting material amount. However, the best assays work directly on
human specimens with no need for fungal outgrowth in pure culture. This requirement
brings multiple problems. First, in general, fungal elements will typically be few in
number in tissue or body fluids and can be vastly overwhelmed by host material, which
can include whole tissue, blood, sputum, and other body fluids, each of which may
bring specific problems to the preparation. For example, hyphae from members of the
Mucorales are more delicate than other filamentous fungi and can be destroyed by
tissue preparations that utilize grinding or other physical methods, which can result in
erroneously low or even absent CFU values. Molds in general, in contrast to yeasts, can
be challenging to precisely enumerate because a single hyphal fragment, which may
contain many nuclei, can grow as a single CFU after plating but yield multiple CFU by
PCR. Second, fungal cell wall material often needs to be removed at some point during
the extraction process since it can inhibit downstream reactions. Melanin, a polyphe-
nolic compound found in many fungi, for example, is a known inhibitor of PCR (10).
Polysaccharides, which many fungi produce in abundance, can also inhibit PCR (11).
Because polysaccharides can be purified along with DNA if not removed in a separate
step, a common chemical used in fungal nucleic acid extraction is CTAB (cetyltrimeth-
ylammonium bromide), which is a cationic detergent that is used to remove polysac-
charides during DNA purification. However, incorporating this reagent into a protocol
adds another step and additional tube manipulations. Third, if the assay employs a PCR
step, the specific extraction protocol or device must be free of contamination, including
associated reagents, which requires great care to accomplish reliably. Fungal nucleic
acids have been found to contaminate primers, probes, and master mix solutions (12,
13). Furthermore, because of the sensitivity of PCR and the possibility of amplifying
contaminating DNA from any source, the various aspects of these assays, including
template preparation, PCR, and other downstream steps, may need to be performed in
separate areas and within containment hoods that protect samples from becoming
contaminated with aerosolized amplicons. While preventing contamination for any
microbe-specific PCR assay is crucial, fungi can be especially challenging because many
contaminating ubiquitous molds common in the environment can also be human
pathogens that could potentially be present in a clinical specimen.
Because clinical laboratories rely on standardized methods and are operated under
April 2020 Volume 58 Issue 4 e01345-19 jcm.asm.org 2
Minireview Journal of Clinical Microbiology
certification from a variety of entities, some automated extraction instruments have
been developed that work quite well. Most of these are magnetic bead-based instru-
ments (14) and may need a preprocessing step that removes the fungal cell wall prior
to the sample being loaded into the instrument. However, in the case of DNA, the
template can be purified to a very high level and with high efficiency. Unfortunately,
because extraction is such a challenging problem for fungal diagnosis, many proce-
dures or systems are independent of the actual assay and need to be purchased or
developed separately from the specific assay. One assay that incorporates sample
preparation into the assay is the T2Dx instrument (T2 Biosystems, Lexington, MA),
which is FDA approved for diagnosing Candida sp. infections from blood specimens.
This system is an example of a “load and walk away” diagnostic system, where the
specimen is loaded into the instrument with little preprocessing and data are collected
at the end of the run leading to an identification. These are ideal assays for a clinical
Downloaded from http://jcm.asm.org/ on March 22, 2021 by guest
microbiology laboratory because they require only minimal technical skills and little
sample manipulation or interpretation. However, instruments that incorporate their
own extraction modules may be easier to develop for yeast pathogens since this
morphology is much easier to extract assay targets from than molds. While not
universally applicable, target extraction from yeast cells can sometimes be done
without physical breakage in contrast to hyphae, which almost always need a physical
breakage step. These differences can be difficult to address with a one-size-fits-all
instrument.
ACCOUNTING FOR FUNGAL MORPHOLOGIC VARIATION
While not unique in microbiology, fungi probably display more morphological
variation and to a larger degree than most microbes. In tissue, they can grow as yeast,
which can be similar to a typical bacterial cell in consistency, or mold, which is
filamentous and hyphal in nature. A smaller subset, the dimorphic fungi, which includes
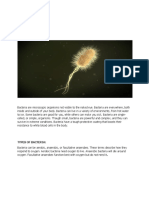
many of the major systemic mycoses, can grow in both morphologies, although few
exhibit both in vivo (Fig. 1). In vitro morphologies can be even more varied, especially
for the molds as sporulation is more common than that in vivo. For assays that rely on
cell lysates to release a target, morphology is less of a problem, although it often can
require different strategies for cell lysis and different degrees of cleanup after lysis to
remove cellular material that is not targeted by the assay. For all morphologies, lysing
cells becomes harder as the cultures age and typically yields better results on fresh or
logarithmic phase cultures, which can add additional delay if a subculture must be
made from the primary specimen.
Whole-cell assays can be very problematic. Yeast cells, because of their unicellular
nature, are easier to manage because they are smaller, easier to quantify by direct
counting or optical density (OD), more uniform in size, and usually not as rigid as
hyphae. Yeast cells also typically have a single nucleus per cell. A filamentous mor-
phology, conversely, can result in hyphal or pseudohyphal filaments that vary in length;
spores that also can vary in size, shape, and hydrophobicity; and other structures that
may grow off hyphae. Hyphae can also be multinucleate, making quantification more
difficult compared to that for a yeast cell. A single nucleus per cell allows a more precise
determination of CFU when trying to quantitate, such as by real-time PCR, which is very
difficult to do when working with a hyphal culture. The filamentous morphology
inhibits a precise cell count in liquid cultures, which may be needed prior to running
an assay because broth cultures of filamentous cells can result in growth as fungal balls,
a single clot of hyphae, or numerous clusters, which makes normalizing for cell number
extremely difficult or impossible. Agar cultures of hyphae need to be scraped, which
still presents problems, as hyphae will be removed as fragments in addition to potential
contaminating pieces of agar. If the culture is a sterile mold, which produces few if any
differentiating structures, it cannot be identified using classical morphologic methods.
Finally, while most yeasts can be grown satisfactorily in or on a nutrient medium such
as Sabouraud’s medium, molds can be more selective in their medium requirements.
For example, a definitive diagnosis of Histoplasma capsulatum may require induction of
April 2020 Volume 58 Issue 4 e01345-19 jcm.asm.org 3
Minireview Journal of Clinical Microbiology
Downloaded from http://jcm.asm.org/ on March 22, 2021 by guest
FIG 1 Dimorphism in Candida albicans. C. albicans is an example of a dimorphic fungus. These fungi can
grow as budding yeast or true septate hyphae. C. albicans also can produce pseudohyphae and,
depending on culture conditions and duration, chlamydospores. Depending on the type of assay,
morphology can affect the outcome or it can determine whether the culture can even be run on a
specific assay or instrument. (A) C. albicans yeast cells grown in broth culture for 20 h. Bar ⫽ 10 m. (B)
C. albicans grown in broth culture showing predominantly hyphal growth. Bar ⫽ 10 m. (C) C. albicans
growing on agar medium as a typical yeast colony with a creamy texture and smooth surface.
Bar ⫽ 1.0 cm. (D) C. albicans growing on agar medium showing a rough colony morphology that has
hyphae growing under the colony. Bar ⫽ 1.0 cm. (Images courtesy of David Kadosh, reproduced with
permission.)
the yeast or hyphal phase, which requires two different media and growth conditions,
and can takes weeks to grow.
Yeast cells are typically identified biochemically in classical microbiology laborato-
ries, often through commercial systems with a standardized substrate panel that is used
for all yeast identifications. These systems may be automated (e.g., Vitek, bioMérieux,
Inc., Durham, NC) or manual (e.g., API 20 C AUX, bioMérieux, Inc.). Cultures that grow
as molds are typically identified based on morphological characteristics, and compre-
hensive mold identification skills usually require specific training in mycology. Pres-
ently, there are few programs that provide this type of education (15), which has
resulted in only rudimentary identification skills typically learned on the job or through
limited laboratory or workshop training. In fact, the lack of trained mycologists in
clinical laboratories is an ongoing problem that continues to get worse (15) and,
ironically, has further increased the urgency for the development of new fungal
diagnostic assays that rely less on formal mycological training. An alternative solution
to the lack of technicians with a background in mycology is the development of new
assays that are panfungal, such that any potential pathogenic fungus’ profile is within
the diagnostic capability (e.g., database) of the assay. Examples would be matrix-
assisted laser desorption ionization–time of flight (MALDI-TOF) or ribosomal sequenc-
ing, such as the internal transcribed spacer (ITS) region and/or a region of the large
ribosomal subunit (D1/D2).
ASSAY SCOPE
One of the most challenging decisions in new assay development is which fungi
should the assay be able to identify. Assays that only identified Candida spp. would
capture the vast majority of human systemic fungal infections and reflect strategic
preferences based on financial projections. Inclusion of common systemic fungal
April 2020 Volume 58 Issue 4 e01345-19 jcm.asm.org 4
Minireview Journal of Clinical Microbiology
pathogens in an assay (Candida, Histoplasma, Cryptococcus, Pneumocystis, Fusarium,
Coccidioides, Aspergillus) would seem to be of great value to a clinical laboratory.
Depending on patient type or geographic region, some of these fungi might never, or
rarely, be encountered, so inclusion of the other fungi could be wasteful or time-
consuming, depending on the type of assay. Moreover, fungi that are rarely encoun-
tered can require the quickest identification due to their invasiveness or potentially
high morbidity and mortality rates. For example, rhinocerebral mucormycosis, an acute
mycotic infection of the sinuses, brain, and mouth, is caused by a number of mucor-
alean species and can be found in patients with poorly managed diabetes and other
immunosuppressed patients. For these infections, surgery is often required, and a delay
in diagnosis of even a few hours can be life-threatening. Because these infections are
generally rare, developing specific assays for their identification can be financially risky
for companies. For rhinocerebral mucormycosis, diagnosis would generally rely on
Downloaded from http://jcm.asm.org/ on March 22, 2021 by guest
patient history, known risk factors, symptoms, and pathology. Even then, a species level
identification would be nearly impossible without a live culture. However, for other
types of immunosuppressed patients, a differential diagnosis may be even more
challenging, leading clinicians to rely on histopathology or live culture, which can be
inconclusive, greatly delayed, or not possible.
Beyond a handful of major human fungal pathogens, few if any FDA-approved
specific diagnostic tests exist beyond classical microbiological testing, and it is unlikely
that a specific diagnostic assay would be cost-effective for fungi that are important but
rarely seen. This lack of tests for a specific fungal infection argues for future develop-
ment to investigate panfungal assays. These assays would almost certainly be sequence
based in order to take advantage of the massive amount of sequence data already
available. However, the ability to obtain pure culture allows non-nucleic acid-based
assays, such as the macromolecule-based method, matrix-assisted laser desorption
ionization–time of flight mass spectrometry (MALDI-TOF MS), to be used. Because this
assay interrogates a reference library with assay-generated spectral data, the library can
be continuously expanded with new reference cultures as has been recently done for
Candida auris (16). The advantage of a panfungal assay is that a prior index of suspicion
about the causative agent arising from culture characteristics or patient history is not
needed to select a specific assay due to the one-size-fits-all nature of these assays.
However, an underlying major problem to getting a panfungal assay through the
approval process would be validating the assay against all potential fungi that the assay
might be tasked with identifying. Generally, culture collections are the only entities that
might hold a substantial number of human pathogenic reference cultures. However,
the cost of a single culture, depending on collection, is in the hundreds of dollars,
making the validation step extremely expensive for just a single culture, let alone
multiple isolates. In some cases, a fungus could be so rare that obtaining multiple
cultures may not be possible.
FUNGAL TAXONOMY AND NOMENCLATURE
The recent change in the International Code for Botanical Nomenclature, now
known as the International Code of Nomenclature for algae, fungi, and plants, leading
to “one fungus one name,” removed the issue of having two names (anamorph and
teleomorph) for some fungi (17). The previous naming system was extremely problem-
atic for medically important fungi and the clinicians that have to keep up with
identifying and treating infections caused by them. The explosion of molecular data
and subsequent taxonomic analysis and reanalysis of phylogenetically informative
sequences has led to even more debate regarding how to name and describe fungal
pathogens. On the one hand, exquisite taxonomic detail derived from DNA sequencing
has resulted in classifications below the species level (i.e., subspecies, varieties, etc.) or
the designation of new species. This detail can be useful in research and sometimes, as
in the case of differences in antifungal susceptibility or virulence, is imperative. For
example, while treating an invasive aspergillosis infection, Balajee et al. discovered a
morphologically indistinguishable sibling species of Aspergillus fumigatus, which was
April 2020 Volume 58 Issue 4 e01345-19 jcm.asm.org 5
Minireview Journal of Clinical Microbiology
named Aspergillus lentulus, based on reduced susceptibilities to multiple antifungal
drugs (18). On the other hand, recognizing pathogenic fungi that display small se-
quence variation and little or no morphologic or biochemical variation, which together
would not alter the treatment strategy, creates a cumbersome problem for clinicians
and clinical microbiologists. Assay developers can be caught in the middle because, in
an effort to show how sensitive a new assay is, reliably discriminating fungi at the
species level can be an asset. However, keeping current with ever-changing fungal
taxonomy and applying these changes accurately in accordance with accepted rules
can be a liability if done incorrectly or not in a timely manner.
An example of how complicated this issue can be is the identification of Fusarium.
These fungi are extremely important pathogens of plants and are one of the most
common mycotoxin producers. In fact, out of the top 100 economically most important
plants, 81 can be infected by Fusarium spp. (19). As a human pathogen, fusaria are
Downloaded from http://jcm.asm.org/ on March 22, 2021 by guest
broadly resistant to available drugs, and they can disseminate in the immunosup-
pressed to cause invasive and life-threatening infections (20). However, more than 60%
cannot be identified using morphological methods (20). Sequence-based identification
methods that do not use targets in the ribosomal locus are typically more complicated
than routine ITS-based sequence identification because a second or even third se-
quence target other than the ITS region may need to be included for proper identifi-
cation. For the fusaria, ITS sequence data alone cannot be used to identify most species.
The additional targets are less conserved than ribosomal targets and may require
genus-specific primers. Unfortunately, selection of which additional targets and primers
to use may be dependent on the ITS sequence. Sequencing multiple loci in a clinical
laboratory is often not feasible, particularly if the choice of the next sequence is
dependent on the identity of the first, which can add substantial delay to an identifi-
cation. Multilocus phylogenetic analyses provided a robust framework for identifying
Fusarium by grouping species that are virtually indistinguishable morphologically into
species complexes (21). Of the twenty or so Fusarium species complexes, the vast
majority containing human pathogens are nested within four complexes, which include
the Fusarium solani species complex (FSSC), Fusarium oxysporum species complex
(FOSC), Fusarium fujikuroi species complex (FFSC), and the Fusarium incarnatum-equiseti
species complex (FIESC) (21, 22). Similar classification strategies have been proposed for
the pathogenic Cryptococcus spp. (23) as well as the aspergilli (24), which groups
species into sections. Expecting new assays to keep up with, and incorporate, complex
taxonomic changes is a formidable challenge now and in the foreseeable future.
COLONIZER VERSUS INVADER DISCRIMINATION
A problem for diagnosing many human mycoses is that the causative agents are
often part of the normal human flora. For these fungi, e.g., Candida spp., background
levels characteristic of normal host stasis need to be distinguished from invasive
infection. An assay’s ability to make this distinction likely requires, for clinical specimens
that are tested directly, a quantitative component or a readout that is deemed positive
in an amount that is known to significantly exceed normal levels or which is associated
with an infection. For example, the Fungitell assay (Associates of Cape Cod, Inc., East
Falmouth, MA) measures fungal beta-D-glucan (BDG) levels in serum. Glucans are part
of the cell walls of many fungi but are not found in humans. A positive result from this
assay is ⱖ80 pg/ml, whereas 60 to 79 pg/ml is indeterminate, and ⱕ60 pg/ml is
negative. However, results of this assay may need to be interpreted with caution
depending on patient type, underlying disease conditions, and fungus that is being
detected. Mokaddas et al. used this assay in a study of pediatric cancer patients and
found BDG levels higher than the positive threshold in some colonized patients
(detected by swab) that did not develop candidemia and who were negative with other
assays (25). PCR-based assays may have more challenges in distinguishing colonization
from invasion because of the amplification nature of the assay and may be most
valuable when employed for negative predictive value or in combination with other
assays.
April 2020 Volume 58 Issue 4 e01345-19 jcm.asm.org 6
Minireview Journal of Clinical Microbiology
Fungi that are common in the environment also pose quantitative problems, with
Aspergillus spp. being one of the most difficult to determine whether it is colonizing or
invading, the so called “bystanders or pathogens” problem (26). For pulmonary asper-
gillosis, this distinction can be challenging as Aspergillus spp. spores are inhaled
regularly, and the fungus can be cultured routinely from sputum, which itself can be
contaminated with fungi in healthy patients. Because Aspergillus sp. do not circulate
frequently in the blood, invasive diagnostic methods, such as biopsy, or less sensitive
methods such as imaging, often must be used; however, some underlying diseases may
preclude biopsy due to potential complications. In addition to the aspergilli, numerous
other environmental fungi can be encountered normally in some clinical specimens.
These may include members of Fusarium, Cryptococcus, Aureobasidium, Cladosporium,
and Penicillium to name a few (27). However, in cases of trauma, where environmental
fungi can be inoculated into tissue (28) but may not be invading, it is difficult to
Downloaded from http://jcm.asm.org/ on March 22, 2021 by guest
determine which fungus treatment should be directed against, as multiple species
might be detected but only one may be invading tissue. Additionally, one of the most
important advantages to having an assay with a quantitative component is the ability
to determine whether antifungal treatment is working. This aspect is especially impor-
tant when an infection is treated empirically since it allows an ineffective antifungal to
be switched to an alternative.
COMBINING IDENTIFICATION AND ANTIFUNGAL SUSCEPTIBILITY TESTING IN A
SINGLE ASSAY
Given the importance of time to diagnosis in infection, diagnostic microbiology is
focusing increasingly on concurrent microbial identification and susceptibility testing.
Separating these two assays adds an inordinate amount of time to infection manage-
ment decision-making since identification would have to occur first, followed by
determining the best antibiotic strategy. Resistance has been shown to occur in almost
every major fungal pathogen, and it is a serious and still growing problem with Candida
spp. In fact, both Candida albicans and Candida glabrata are on the World Health
Organization antibiotic threat list (29). Some species, such as C. glabrata and C. auris can
show innate resistance to some antifungals or can become resistant at fairly high
frequencies. In the case of C. auris, resistance is problematic due to its high mortality
rate, which is attributed in part to its tendency to appear in immunosuppressed
patients who are already debilitated in their response to infection (30). Antifungal
resistance in invasive aspergillosis is also becoming a major cause of concern, and it has
been hypothesized to be due, in part, to the indiscriminate use of azoles in agriculture
(31). In some regions, greater than 10% of aspergillosis is drug resistant (32). Some
species of Aspergillus, such as Aspergillus terreus, have innate reduced susceptibility to
some antifungals, making rapid identification paramount for good outcomes. Innate
resistance is not uncommon in other fungi, such as Scedosporium or Fusarium; however,
assays that combine both identification and susceptibility in a single test are limited
and still early in development in fungi.
Presently, two general methods exist for susceptibility testing. These include phe-
notypic testing, which utilizes live culture to gauge growth in the presence of antibi-
otics, and genotypic testing, which looks for molecular markers associated with resis-
tance. Diagnostic assays that combine the two are obviously complicated, although
progress is being made for bacterial diagnosis. For examples, Accelerate Diagnostics,
Inc. (Tucson, AZ) has a commercially available assay that utilizes a fluorescent in situ
hybridization (FISH) component for identification and time lapse microscopy to mea-
sure susceptibility, all of which can be completed in ⬃5 h (33). The assay works on a
variety of bacteria (identification and susceptibility) as well as two species of Candida
(identification only).
In contrast to phenotypic assays, genotypic assays do not require a growth com-
ponent and likely could perform identification and susceptibility simultaneously.
Whole-genome sequencing is a likely candidate for this role; however, it is probably
years away, as template preparation, in the form of libraries to be sequenced, and
April 2020 Volume 58 Issue 4 e01345-19 jcm.asm.org 7
Minireview Journal of Clinical Microbiology
bioinformatic analysis that could output identification and markers of antifungal resis-
tance with little or no data manipulation would be challenging. Additionally, the
presence of a specific biomarker, such as a single nucleotide polymorphism (SNP)
associated with resistance, does not necessarily mean resistance would be expressed
given that numerous biomarkers can be associated with drug resistance, individually or
collectively. MALDI-TOF MS, conversely, may be more feasible, as it currently has been
used to explore resistance and identification in C. albicans and C. glabrata (34).
However, studies have focused mainly on comparing spectra produced when yeast
cultures are grown in different levels of antifungal (35), which could be costly or
time-consuming.
ADMINISTRATIVE CHALLENGES
Diagnosis of fungal infections relies heavily on the appropriate use of laboratory
Downloaded from http://jcm.asm.org/ on March 22, 2021 by guest
testing for effective treatment to be initiated in a timely manner but can be hindered
by a number of factors, including lack of personnel with specific mycology training, lack
of suitable diagnostic alternatives to live culture, and difficulties in standardizing these
alternative diagnostic options. The need for timely diagnosis also aligns with the goals
of an institution’s antimicrobial stewardship program (ASP), which is designed to
optimize patient outcomes while minimizing antimicrobial harm, such as toxicity,
unfavorable drug-drug interactions, and selection for resistant organisms (36). Antimi-
crobial stewardship programs and prescribing physicians depend on information and
guidance from the clinical microbiology laboratory in order to accomplish these goals.
While stewardship is critical for all infectious agents, fungi offer some unique problems.
Treatment options are much more limited for fungi than for bacteria due to the limited
antifungal repertoire, and the few remaining choices in cases of resistance, such as
amphotericin B, can have severe side effects.
There are many challenges that clinical laboratories face when asked to bring in new
laboratory testing for fungal identification. The clinical laboratory has witnessed a shift
away from classic morphologic identification of microorganisms to more molecular and
proteomic-based methods. However, PCR assays have not reached the same level of
acceptance for the detection of human fungal pathogens as for other microorganisms,
mainly because the low amount of fungal DNA in clinical specimens often challenges
the detection limits of PCR. One of the most common questions facing laboratories
when considering new fungal diagnostic tests is deciding which specific assay best suits
the needs of the lab, its patients, and its providers. The increasing diversity of fungal
pathogens poses a significant challenge in choosing the right test (37), which can be
exacerbated by trying to keep current with the seemingly constant reorganization of
fungal taxonomy. Because PCR assays are directed at a single or a few species, the
clinician needs to have an index of suspicion with regard to a given species prior to
selecting a specific PCR assay, which can make the assay confirmatory in nature.
Implementation of diagnostic platforms in the clinical microbiology laboratory
requires careful consideration of personnel requirements, ease of use and cost of an
instrument if one is required, facility requirements, workflow design, and institutional
buy-in. The number and types of fungi that come into the laboratory for identification
can affect this decision-making process. Importantly, fungi that come into the labora-
tory can be greatly affected by geography, as many are geographically restricted.
Coccidioides spp., for example, might be frequently seen in the desert southwest of the
United States but rarely if ever seen in the northeast, which would argue for keeping
a specific diagnostic assay for this fungus on hand in an Arizona clinical laboratory but
not in Maine. In fact, diagnosing endemic fungal infections in the United States, such
as coccidioidomycosis and histoplasmosis, is a serious enough issue that the National
Institutes of Health has recently released funding opportunities for diagnostic assays
that specifically target this class of fungi. Alternatively, patient type can affect the
spectrum of fungi seen in clinical specimens. Aspergillus and Fusarium might be
expected in clinical laboratories that support cancer or transplant patients. Trauma
centers, particularly if they support combat patients, might see elevated levels of
April 2020 Volume 58 Issue 4 e01345-19 jcm.asm.org 8
Minireview Journal of Clinical Microbiology
Mucorales. In fact, these centers might consider sequence-based identification ap-
proaches, as well as the associated equipment and technician costs, as identification
platforms due to the rarity of these fungi and the difficulty in identifying them to the
species level. However, the commercial assays currently available for fungal identifica-
tion are generally not comprehensive and typically only detect one or a few fungal
pathogens at a time (38). MALDI-TOF MS is an exception, however. It requires an
existing internal database that is interrogated by the mass spectra produced from
unknown specimens. The sensitivities of these assays are also low, meaning that the
fungal burden needs to be relatively high for the assay to be able to identify the fungus
if an assay can even be used directly on clinical specimens. The limitations of the
methodologies currently used for diagnosing invasive fungal infections often force
clinicians to take an empirical approach to antifungal therapy, which has led to both an
increase in antifungal resistance as well as time to diagnosis and illustrates the need for
Downloaded from http://jcm.asm.org/ on March 22, 2021 by guest
better assays to detect and identify fungal pathogens in a timely manner (39).
Finally, no assay can be implemented without considering cost savings. Cost savings
at the bedside and in the laboratory are a win-win for all parties involved. Intuitively,
if a diagnosis is provided earlier and is more accurate and reliable, patients who require
antifungal therapy will receive it sooner, potentially shortening the length of their
hospital stay, decreasing chances of acquiring nosocomial infections, and decreasing
mortality rates (40). This outcome will lead to a cost savings for the patient and will
have a positive financial impact on the institution. Most importantly, patient outcomes
will be substantially improved with a faster and more accurate laboratory test for fungal
identification.
CONCLUSIONS
Diagnosing fungal infections has always been challenging for numerous reasons,
some of which overlap with bacterial diagnosis. However, the unique nature of fungi
and the general rarity of infection compared to that with bacteria have resulted in
major impediments to rapid, comprehensive assays for fungal identification. Some of
the extreme challenges will continue to hold back the field of fungal diagnosis. Solving
these challenges will require new advances in technology and a more creative appli-
cation of current technologies to clinical problems. Conversely, better education and
training as well as novel ways to introduce and utilize new tests in the microbiology
laboratory may be one of the easier ways to improve fungal diagnosis.
ACKNOWLEDGMENTS
We thank David Kadosh for the Candida albicans images.
B.L.W. is supported by R21 AI128479-01A1 and R21 AI146700-01.
REFERENCES
1. Kozel TR, Wickes B. 2014. Fungal diagnostics. Cold Spring Harb Perspect medicine and veterinary sciences. Adv Appl Microbiol 66:29 –52. https://
Med 4:a019299. https://doi.org/10.1101/cshperspect.a019299. doi.org/10.1016/S0065-2164(08)00802-2.
2. Duffy J, Harris J, Gade L, Sehulster L, Newhouse E, O’Connell H, Noble- 7. Arastehfar A, Wickes BL, Ilkit M, Pincus DH, Daneshnia F, Pan W, Fang W,
Wang J, Rao C, Balajee SA, Chiller T. 2014. Mucormycosis outbreak Boekhout T. 2019. Identification of mycoses in developing countries. J
associated with hospital linens. Pediatr Infect Dis J 33:472– 476. https:// Fungi (Basel) 5:E90. https://doi.org/10.3390/jof5040090.
doi.org/10.1097/INF.0000000000000261. 8. Pincus DH, Orenga S, Chatellier S. 2007. Yeast identification–past, pres-
3. Neblett Fanfair R, Benedict K, Bos J, Bennett SD, Lo YC, Adebanjo T, ent, and future methods. Med Mycol 45:97–121. https://doi.org/10.1080/
Etienne K, Deak E, Derado G, Shieh WJ, Drew C, Zaki S, Sugerman D, 13693780601059936.
Gade L, Thompson EH, Sutton DA, Engelthaler DM, Schupp JM, Brandt 9. Lau A, Chen S, Sleiman S, Sorrell T. 2009. Current status and future
ME, Harris JR, Lockhart SR, Turabelidze G, Park BJ. 2012. Necrotizing perspectives on molecular and serological methods in diagnostic my-
cutaneous mucormycosis after a tornado in Joplin, Missouri, in 2011. N cology. Future Microbiol 4:1185–1222. https://doi.org/10.2217/fmb.09
Engl J Med 367:2214 –2225. https://doi.org/10.1056/NEJMoa1204781. .70.
4. Larone DH, Walsh TJ. 2013. Exserohilum rostratum: anatomy of a national 10. Arastehfar A, Fang W, Daneshnia F, Al-Hatmi AM, Liao W, Pan W, Khan Z,
outbreak of fungal meningitis. Clin Microbiol News 35:185–193. https:// Ahmad S, Rosam K, Lackner M, Lass-Flörl C, Hagen F, Boekhout T. 2019.
doi.org/10.1016/j.clinmicnews.2013.11.001. Novel multiplex real-time quantitative PCR detecting system approach
5. Jeffery-Smith A, Taori SK, Schelenz S, Jeffery K, Johnson EM, Borman A, for direct detection of Candida auris and its relatives in spiked serum
Candida auris Incident Management Team, Manuel R, Brown CS. 2018. samples. Future Microbiol 14:33– 45. https://doi.org/10.2217/fmb-2018
Candida auris: a review of the literature. Clin Microbiol Rev 31:e00029-17. -0227.
https://doi.org/10.1128/CMR.00029-17. 11. Inglis PW, Pappas MCR, Resende LV, Grattapaglia D. 2018. Fast and
6. Sparagano O, Foggett S. 2009. Diagnosis of clinically relevant fungi in inexpensive protocols for consistent extraction of high quality DNA and
April 2020 Volume 58 Issue 4 e01345-19 jcm.asm.org 9
Minireview Journal of Clinical Microbiology
RNA from challenging plant and fungal samples for high-throughput lus, p 55– 62. In Samson RA, Pitt JI (ed), Advances in Penicillium and
SNP genotyping and sequencing applications. PLoS One 13:e0206085. Aspergillus systematics. Plenum Press, New York, NY.
https://doi.org/10.1371/journal.pone.0206085. 25. Mokaddas E, Burhamah MH, Khan ZU, Ahmad S. 2010. Levels of (1¡3)-
12. Czurda S, Smelik S, Preuner-Stix S, Nogueira F, Lion T. 2016. Occurrence beta-D-glucan, Candida mannan and Candida DNA in serum samples of
of fungal DNA contamination in PCR reagents: approaches to control pediatric cancer patients colonized with Candida species. BMC Infect Dis
and decontamination. J Clin Microbiol 54:148 –152. https://doi.org/10 10:292. https://doi.org/10.1186/1471-2334-10-292.
.1128/JCM.02112-15. 26. Chotirmall SH, McElvaney NG. 2014. Fungi in the cystic fibrosis lung:
13. Wagner K, Springer B, Pires VP, Keller PM. 2018. Molecular detection of bystanders or pathogens? Int J Biochem Cell Biol 52:161–173. https://
fungal pathogens in clinical specimens by 18S rDNA high-throughput doi.org/10.1016/j.biocel.2014.03.001.
screening in comparison to ITS PCR and culture. Sci Rep 8:6964. https:// 27. Ghannoum MA, Jurevic RJ, Mukherjee PK, Cui F, Sikaroodi M, Naqvi A,
doi.org/10.1038/s41598-018-25129-w. Gillevet PM. 2010. Characterization of the oral fungal microbiome (my-
14. Perry MD, White PL, Barnes RA. 2014. Comparison of four automated cobiome) in healthy individuals. PLoS Pathog 6:e1000713. https://doi
nucleic acid extraction platforms for the recovery of DNA from Aspergil- .org/10.1371/journal.ppat.1000713.
lus fumigatus. J Med Microbiol 63:1160 –1166. https://doi.org/10.1099/ 28. Ganesan A, Wells J, Shaikh F, Peterson P, Bradley W, Carson L, Petfield J,
jmm.0.076315-0. Klassen-Fischer M, Akers K, Downing K, Bialek R, Tribble D, Wickes BL.
15. Steinbach WJ, Mitchell TG, Schell WA, Espinel-Ingroff A, Coico RF, Walsh 2019. Molecular detection of filamentous fungi in formalin-fixed
TJ, Perfect JR. 2003. Status of medical mycology education. Med Mycol paraffin-embedded specimens in invasive fungal wound infections is
Downloaded from http://jcm.asm.org/ on March 22, 2021 by guest
41:457– 467. https://doi.org/10.1080/13693780310001631322. feasible with high specificity. J Clin Microbiol 58:e01259-19. https://doi
16. Bao JR, Master RN, Azad KN, Schwab DA, Clark RB, Jones RS, Moore EC, .org/10.1128/JCM.01259-19.
Shier KL. 2018. Rapid, accurate identification of Candida auris by using a 29. WHO. 2014. Antimicrobial resistance: global report on surveillance 2014.
novel matrix-assisted laser desorption ionization-time of flight mass World Health Organization, Geneva, Switzerland.
spectrometry (MALDI-TOF MS) database (library). J Clin Microbiol 56: 30. Chowdhary A, Sharma C, Meis JF. 2017. Candida auris: a rapidly emerg-
e01700-17. https://doi.org/10.1128/JCM.01700-17. ing cause of hospital-acquired multidrug-resistant fungal infections
17. Hawksworth DL. 2011. A new dawn for the naming of fungi: impacts of globally. PLoS Pathog 13:e1006290. https://doi.org/10.1371/journal.ppat
decisions made in Melbourne in July 2011 on the future publication and .1006290.
regulation of fungal names. IMA Fungus 2:155–162. https://doi.org/10 31. Berger S, El Chazli Y, Babu AF, Coste AT. 2017. Azole resistance in
.5598/imafungus.2011.02.02.06. Aspergillus fumigatus: a consequence of antifungal use in agriculture?
18. Balajee SA, Gribskov JL, Hanley E, Nickle D, Marr KA. 2005. Aspergillus Front Microbiol 8:1024. https://doi.org/10.3389/fmicb.2017.01024.
lentulus sp. nov., a new sibling species of A. fumigatus. Eukaryot Cell 32. Rivero-Menendez O, Alastruey-Izquierdo A, Mellado E, Cuenca-Estrella M.
4:625– 632. https://doi.org/10.1128/EC.4.3.625-632.2005. 2016. Triazole resistance in Aspergillus spp.: a worldwide problem? J
19. Moretti AN. 2009. Taxonomy of Fusarium genus, a continuous fight Fungi 2:21. https://doi.org/10.3390/jof2030021.
between lumpers and splitters. Zb Mat Srp Prir Nauk 117:7–13. https:// 33. Burnham JP, Wallace MA, Fuller BM, Shupe A, Burnham CD, Kollef MH.
doi.org/10.2298/ZMSPN0917007M. 2019. Clinical effect of expedited pathogen identification and suscepti-
20. O’Donnell K, Sutton DA, Rinaldi MG, Sarver BAJ, Balajee SA, Schroers H-J, bility testing for Gram-negative bacteremia and candidemia by use of
Summerbell RC, Robert VARG, Crous PW, Zhang N, Aoki T, Jung K, Park the Accelerate Pheno system. J Appl Lab Med 3:569 –579. https://doi
J, Lee Y-H, Kang S, Park B, Geiser DM. 2010. Internet-accessible DNA .org/10.1373/jalm.2018.027201.
sequence database for identifying fusaria from human and animal in- 34. Florio W, Tavanti A, Ghelardi E, Lupetti A. 2018. MALDI-TOF MS applica-
fections. J Clin Microbiol 48:3708 –3718. https://doi.org/10.1128/JCM tions to the detection of antifungal resistance: state of the art and future
.00989-10. perspectives. Front Microbiol 9:2577. https://doi.org/10.3389/fmicb.2018
21. van Diepeningen A, Al-Hatmi AMS, Brankovics B, Sybren de Hoog G. .02577.
2014. Taxonomy and clinical spectra of Fusarium species: where do we 35. OPATHY Consortium, Gabaldon T. 2019. Recent trends in molecular
stand in 2014? Curr Clin Micro Rep 1:10 –18. https://doi.org/10.1007/ diagnostics of yeast infections: from PCR to NGS. FEMS Microbiol Rev
s40588-014-0003-x. 43:517–547. https://doi.org/10.1093/femsre/fuz015.
22. O’Donnell K, Rooney AP, Proctor RH, Brown DW, McCormick SP, Ward TJ, 36. Alanio A, Bretagne S. 2014. Difficulties with molecular diagnostic tests
Frandsen RJN, Lysøe E, Rehner SA, Aoki T, Robert VARG, Crous PW, for mould and yeast infections: where do we stand? Clin Microbiol Infect
Groenewald JZ, Kang S, Geiser DM. 2013. Phylogenetic analyses of RPB1 20(Suppl):36 – 41. https://doi.org/10.1111/1469-0691.12617.
and RPB2 support a middle Cretaceous origin for a clade comprising all 37. Klutts JS, Robinson-Dunn B. 2011. A critical appraisal of the role of the
agriculturally and medically important fusaria. Fungal Genet Biol 52: clinical microbiology laboratory in diagnosis of invasive fungal infec-
20 –31. https://doi.org/10.1016/j.fgb.2012.12.004. tions. J Clin Microbiol 49:S39 –S42. https://doi.org/10.1128/JCM.00468
23. Kwon-Chung KJ, Bennett JE, Wickes BL, Meyer W, Cuomo CA, Wollen- -11.
burg KR, Bicanic TA, Castaneda E, Chang YC, Chen J, Cogliati M, Dromer 38. Wolk DW, Dunne WM. 2011. New technologies in clinical microbiology.
F, Ellis D, Filler SG, Fisher MC, Harrison TS, Holland SM, Kohno S, Kronstad J Clin Microbiol 49:S62–S67. https://doi.org/10.1128/JCM.00834-11.
JW, Lazera M, Levitz SM, Lionakis MS, May RC, Ngamskulrongroj P, 39. Enoch DA, Ludlam HA, Brown NM. 2006. Invasive fungal infections: a
Pappas PG, Perfect JR, Rickerts V, Sorrell TC, Walsh TJ, Williamson PR, Xu review of epidemiology and management options. J Med Microbiol
J, Zelazny AM, Casadevall A. 2017. The case for adopting the “species 55:809 – 818. https://doi.org/10.1099/jmm.0.46548-0.
complex” nomenclature for the etiologic agents of cryptococcosis. 40. Fournier PE, Drancourt M, Colson P, Rolain JM, La Scola B, Raoult D. 2013.
mSphere 2:e00357-16. https://doi.org/10.1128/mSphere.00357-16. Modern clinical microbiology: new challenges and solutions. Nat Rev
24. Gams W, Christensen M, Onions AH. 1985. Infrageneric taxa of Aspergil- Microbiol 11:574 –585. https://doi.org/10.1038/nrmicro3068.
April 2020 Volume 58 Issue 4 e01345-19 jcm.asm.org 10
You might also like
- 45 Supplement - 2 S99Document13 pages45 Supplement - 2 S99Mustaqeem DawarNo ratings yet
- J. Clin. Microbiol.-2011-Diekema-S57-60 PDFDocument4 pagesJ. Clin. Microbiol.-2011-Diekema-S57-60 PDFmela renNo ratings yet
- JM 002169Document4 pagesJM 002169edisonballaNo ratings yet
- CaseDocument2 pagesCase21100533No ratings yet
- Antimicrobial Susceptibility Testing - A Primer For Clinicians 2009 - ReadDocument18 pagesAntimicrobial Susceptibility Testing - A Primer For Clinicians 2009 - ReadJayelle2No ratings yet
- Clinical Microbiology and Infection PreventionDocument5 pagesClinical Microbiology and Infection PreventionRabboni gospelNo ratings yet
- Blood Cultures 2 IdsaDocument94 pagesBlood Cultures 2 IdsaM fNo ratings yet
- A Guide To Utilization of The Microbiology Laboratory - IDSADocument94 pagesA Guide To Utilization of The Microbiology Laboratory - IDSAKarla Novoa PérezNo ratings yet
- The Revolution of Lateral Flow Assay in the Field of AMRDetectionDocument26 pagesThe Revolution of Lateral Flow Assay in the Field of AMRDetectionalbertNo ratings yet
- Antimicrobial Susceptibility Testing - Primer For CliniciansDocument20 pagesAntimicrobial Susceptibility Testing - Primer For Cliniciansdipzhit100% (1)
- Environmental Mycology in Public Health: Fungi and Mycotoxins Risk Assessment and ManagementFrom EverandEnvironmental Mycology in Public Health: Fungi and Mycotoxins Risk Assessment and ManagementCarla ViegasRating: 5 out of 5 stars5/5 (6)
- Antimicrobial Susceptibility Testing Paradigms Current Status and Future DirectionsDocument22 pagesAntimicrobial Susceptibility Testing Paradigms Current Status and Future DirectionsrehanaNo ratings yet
- DOC-20240208-WA0011.Document11 pagesDOC-20240208-WA0011.umayamajidNo ratings yet
- A Guide To Utilization of The Microbiology Laboratory For Diagnosis of Infectious Diseases: 2018 Update by The Infectious Diseases Society of America and The American Society For MicrobiologyaDocument94 pagesA Guide To Utilization of The Microbiology Laboratory For Diagnosis of Infectious Diseases: 2018 Update by The Infectious Diseases Society of America and The American Society For MicrobiologyaGestaltNo ratings yet
- Young MicrobiologistsDocument82 pagesYoung Microbiologiststummalapalli venkateswara raoNo ratings yet
- 2018 Update - IDSA and ASM Diagnosis of Infectious Diseases Laboratory Guidelines 2018Document94 pages2018 Update - IDSA and ASM Diagnosis of Infectious Diseases Laboratory Guidelines 2018Bolohan BiatriceNo ratings yet
- Idsa 2018Document94 pagesIdsa 2018Gaby Galarza TaboadaNo ratings yet
- Current Technologies in Antimicrobial Susceptibility Testing and Microbial Identification:-Advantages, Limitations/challenges & Future PerspectiveDocument21 pagesCurrent Technologies in Antimicrobial Susceptibility Testing and Microbial Identification:-Advantages, Limitations/challenges & Future Perspectivebonsa dadhiNo ratings yet
- Diagnosis of Infectious Diseases: MolecularDocument4 pagesDiagnosis of Infectious Diseases: MolecularKaily TrầnNo ratings yet
- Infecciones y Examenes IDSA 2018Document94 pagesInfecciones y Examenes IDSA 2018Gaby Galarza TaboadaNo ratings yet
- Antimicrobial Susceptibility Testing: A Review of General Principles and Contemporary PracticesDocument7 pagesAntimicrobial Susceptibility Testing: A Review of General Principles and Contemporary Practicesabhishek yadavNo ratings yet
- Film Array PDFDocument10 pagesFilm Array PDFW Antonio Rivera MartínezNo ratings yet
- IARC Sci Pub 163 - Chapter 10Document14 pagesIARC Sci Pub 163 - Chapter 10ashin411No ratings yet
- Culturing of Microbiology Specimens in LaboratoriesDocument100 pagesCulturing of Microbiology Specimens in Laboratoriestummalapalli venkateswara raoNo ratings yet
- Wa0006.Document12 pagesWa0006.Jose VillasmilNo ratings yet
- 2009-Antimicrobial SusceptilityDocument7 pages2009-Antimicrobial SusceptilityStephanie MurillejoNo ratings yet
- Current Promising Opportunities of Treating Ploymicro DiseaesDocument29 pagesCurrent Promising Opportunities of Treating Ploymicro Diseaesczikkely23No ratings yet
- B Cepacia What Is It and Why Is It A ConcernDocument5 pagesB Cepacia What Is It and Why Is It A ConcernBlueSagaNo ratings yet
- Utilization of The Microbiology Laboratory For Diagnosis of Infectious Diseases 2018 Update by The Infectious Diseases Society of America and The American Society For MicrobiologyDocument94 pagesUtilization of The Microbiology Laboratory For Diagnosis of Infectious Diseases 2018 Update by The Infectious Diseases Society of America and The American Society For MicrobiologyDragonNo ratings yet
- Diagnosis MikrobiologiDocument14 pagesDiagnosis Mikrobiologiarmelya aisyahNo ratings yet
- Balouri, DKK., 2016Document31 pagesBalouri, DKK., 2016Muhajir_samNo ratings yet
- Anti Micro Biii AlDocument7 pagesAnti Micro Biii AltimiiniyiiNo ratings yet
- Fdocuments - in Biology Investigatory Project 561e79b91f5a0Document18 pagesFdocuments - in Biology Investigatory Project 561e79b91f5a0ASHOKNo ratings yet
- Paso Corto 7 - Diagnostic Microbiology in Veterinary Dermatology-1Document10 pagesPaso Corto 7 - Diagnostic Microbiology in Veterinary Dermatology-1ABIGAIL AKEMI RODRIGUEZ CHAVEZNo ratings yet
- In Vitro vs In Vivo Antimicrobial ActivityDocument2 pagesIn Vitro vs In Vivo Antimicrobial ActivityDebbyNovriozaNo ratings yet
- Viral Pathogenesis: From Basics to Systems BiologyFrom EverandViral Pathogenesis: From Basics to Systems BiologyRating: 5 out of 5 stars5/5 (2)
- Bio Film: Sowmya Subramanian, Ryan C. Huiszoon, Sangwook Chu, William E. Bentley, Reza GhodssiDocument16 pagesBio Film: Sowmya Subramanian, Ryan C. Huiszoon, Sangwook Chu, William E. Bentley, Reza GhodssiOussama El BouadiNo ratings yet
- Archives of Clinical MicrobiologyDocument86 pagesArchives of Clinical MicrobiologyInternational Medical PublisherNo ratings yet
- PROA 4 Texto Explicativo SpanishDocument7 pagesPROA 4 Texto Explicativo SpanishLili VargasNo ratings yet
- Civ 609Document17 pagesCiv 609Descargar scribdNo ratings yet
- Clinical Microbiology and InfectionDocument20 pagesClinical Microbiology and InfectionArthur SchulzNo ratings yet
- M.kansasii 2Document22 pagesM.kansasii 2Ikeh ChisomNo ratings yet
- Neisseria Meningitidis - Advanced Methods and Protocols (PDFDrive)Document442 pagesNeisseria Meningitidis - Advanced Methods and Protocols (PDFDrive)RaffaharianggaraNo ratings yet
- Klebsiella Pneumoniae: An Increasing Threat: To Public HealthDocument9 pagesKlebsiella Pneumoniae: An Increasing Threat: To Public HealthPhuong NguyenNo ratings yet
- Syndromic PanelDocument4 pagesSyndromic PanelMarcellia AngelinaNo ratings yet
- DiagnosisofbacterialinfectionDocument4 pagesDiagnosisofbacterialinfectionagung kurniaNo ratings yet
- Silva 2021Document13 pagesSilva 2021sara95nnNo ratings yet
- 02 MicrobiologyDocument30 pages02 Microbiologyzenagit123456No ratings yet
- The Ethics of Prophylactic Antibiotics For Neurosurgical ProceduresDocument6 pagesThe Ethics of Prophylactic Antibiotics For Neurosurgical ProceduresBahtiar ArasNo ratings yet
- PARASITIC INFECTIONS (Why We Need Better Diagnostic Approaches)Document2 pagesPARASITIC INFECTIONS (Why We Need Better Diagnostic Approaches)tummalapalli venkateswara raoNo ratings yet
- Wajib - Tuberculosis - Pathophysiology, Clinical Features, and DiagnosisDocument12 pagesWajib - Tuberculosis - Pathophysiology, Clinical Features, and DiagnosisnurulahdiahNo ratings yet
- New and Developing Diagnostic Technologies For Urinary Tract Infections - PMCDocument25 pagesNew and Developing Diagnostic Technologies For Urinary Tract Infections - PMCPuran pataNo ratings yet
- SuperbugsDocument3 pagesSuperbugsMatei BuneaNo ratings yet
- Book 2Document56 pagesBook 2PintilieNicoletaNo ratings yet
- TB PDFDocument12 pagesTB PDFHana AshrafNo ratings yet
- 1819 Nurs1701 Co 2019.02.27Document14 pages1819 Nurs1701 Co 2019.02.27nolifeNo ratings yet
- Research Center Diagnosis of Pulmonary Tuberculosis - How Rapid Can It Get?Document10 pagesResearch Center Diagnosis of Pulmonary Tuberculosis - How Rapid Can It Get?lenysfaridaNo ratings yet
- Covid-19 Vaccine Development: Executive SummaryDocument13 pagesCovid-19 Vaccine Development: Executive SummaryNasir AbdulraheemNo ratings yet
- 2022Shah_Infections (1)Document20 pages2022Shah_Infections (1)rehanaNo ratings yet
- Sadia Perween 2 EtalDocument12 pagesSadia Perween 2 EtalrehanaNo ratings yet
- Ajidm 1 4 2Document6 pagesAjidm 1 4 2rehanaNo ratings yet
- 11Document12 pages11Anonymous S0MyRHNo ratings yet
- ch-068-ophthalmia-neonatorumDocument7 pagesch-068-ophthalmia-neonatorumrehanaNo ratings yet
- Int J of Urology - 2009 - Christensen - How To Structure Research PapersDocument2 pagesInt J of Urology - 2009 - Christensen - How To Structure Research PapersrehanaNo ratings yet
- How To Access Ppts and Other Resource Materials On Iecho Platform??Document4 pagesHow To Access Ppts and Other Resource Materials On Iecho Platform??rehanaNo ratings yet
- 2019 Carbapenemases inDocument12 pages2019 Carbapenemases inrehanaNo ratings yet
- Risk Factors For Disease ProgressionDocument6 pagesRisk Factors For Disease ProgressionrehanaNo ratings yet
- Diagnosis MeningitisDocument25 pagesDiagnosis MeningitisnoorgianilestariNo ratings yet
- Implementation of A Sample Pooling Strategy For TheDocument9 pagesImplementation of A Sample Pooling Strategy For TherehanaNo ratings yet
- The Antibiogram Key Considerations For Its DevelopmentDocument6 pagesThe Antibiogram Key Considerations For Its DevelopmentrehanaNo ratings yet
- Spirochete Flagella and MotilityDocument16 pagesSpirochete Flagella and MotilityrehanaNo ratings yet
- Current Methods For The Identification of CarbapenemasesDocument20 pagesCurrent Methods For The Identification of CarbapenemasesrehanaNo ratings yet
- Evaluation of Sample Pooling For Diagnosis of COVID 19 by Real Time PCR - A Resource Saving Combat StrategyDocument1 pageEvaluation of Sample Pooling For Diagnosis of COVID 19 by Real Time PCR - A Resource Saving Combat StrategyrehanaNo ratings yet
- Pooling Upper Respiratory Specimens For Rapid Mass Screening of COVID-19 by Real-Time RT-PCRDocument4 pagesPooling Upper Respiratory Specimens For Rapid Mass Screening of COVID-19 by Real-Time RT-PCRrehanaNo ratings yet
- Fna 90heroicminutes ReferenceDocument3 pagesFna 90heroicminutes ReferencerehanaNo ratings yet
- PoolingDocument5 pagesPoolingrehanaNo ratings yet
- Etiological and Epidemiological Features of AcuteDocument11 pagesEtiological and Epidemiological Features of AcuterehanaNo ratings yet
- Risk Factors For Disease ProgressionDocument6 pagesRisk Factors For Disease ProgressionrehanaNo ratings yet
- Pooling Upper Respiratory Specimens For Rapid Mass Screening of COVID-19 by Real-Time RT-PCRDocument4 pagesPooling Upper Respiratory Specimens For Rapid Mass Screening of COVID-19 by Real-Time RT-PCRrehanaNo ratings yet
- Category - Thesis Writing - Elite EditingDocument8 pagesCategory - Thesis Writing - Elite EditingrehanaNo ratings yet
- Thesis Publication - International Journal of Research (IJR)Document3 pagesThesis Publication - International Journal of Research (IJR)rehanaNo ratings yet
- Policy Guidance On Drug-Susceptibility Testing (DST) of Second-Line Antituberculosis DrugsDocument20 pagesPolicy Guidance On Drug-Susceptibility Testing (DST) of Second-Line Antituberculosis DrugsrehanaNo ratings yet
- WHO GUIDE TO Cost-Effectiveness Analysis PDFDocument329 pagesWHO GUIDE TO Cost-Effectiveness Analysis PDFdsshhkNo ratings yet
- WHO GUIDE TO Cost-Effectiveness Analysis PDFDocument329 pagesWHO GUIDE TO Cost-Effectiveness Analysis PDFdsshhkNo ratings yet
- A Look Inside Oral Sampling For Detection of Non-OralDocument14 pagesA Look Inside Oral Sampling For Detection of Non-OralrehanaNo ratings yet
- 7 Series Controversial Issues in Tuberculosis''Document10 pages7 Series Controversial Issues in Tuberculosis''rehanaNo ratings yet
- Nonconventional and New Methods in TheDocument12 pagesNonconventional and New Methods in Themaria_brocheroNo ratings yet
- DSTslides Updated FINALDocument65 pagesDSTslides Updated FINALrehanaNo ratings yet
- Lab Manual Pharmaceutical MicrobiologyDocument31 pagesLab Manual Pharmaceutical MicrobiologyAravindhNo ratings yet
- Disease by BacteriaDocument10 pagesDisease by BacteriaPratimaNo ratings yet
- K Park 24 TH Edition ChangesDocument8 pagesK Park 24 TH Edition ChangesAnkit ChoudharyNo ratings yet
- Antimycobacterial DrugsDocument36 pagesAntimycobacterial Drugsmadhu bonamNo ratings yet
- University of Delaware College of Health and Nursing Sciences - Hepatitis B Vaccine Declination Form Students)Document2 pagesUniversity of Delaware College of Health and Nursing Sciences - Hepatitis B Vaccine Declination Form Students)DonnaNo ratings yet
- Frequencies found by Richard Loyd and John Loyd as of 2/12/2021Document4 pagesFrequencies found by Richard Loyd and John Loyd as of 2/12/2021Ricardo GomesNo ratings yet
- Outline Biofilms ReportDocument4 pagesOutline Biofilms ReportDaphne Kyara AlgarmeNo ratings yet
- Soil Transmitted HelminthiasisDocument9 pagesSoil Transmitted HelminthiasisSuperboinkyNo ratings yet
- Employsure - COVID Safety Plan TemplateDocument30 pagesEmploysure - COVID Safety Plan TemplateEvonne TanNo ratings yet
- Epidemiology of Lymphatic FilariasisDocument26 pagesEpidemiology of Lymphatic FilariasisvaishnaviNo ratings yet
- Probiotic Strains in American Probiotic ProductsDocument2 pagesProbiotic Strains in American Probiotic ProductsexpandNo ratings yet
- Review of Related Literature and StudiesDocument4 pagesReview of Related Literature and StudiesKimNo ratings yet
- Antibiotics MindmapDocument5 pagesAntibiotics Mindmapsuman goyetNo ratings yet
- Casauay and Pacunana Group 15 1Document4 pagesCasauay and Pacunana Group 15 1Steve EstebanNo ratings yet
- Notes in Sanitation TleDocument2 pagesNotes in Sanitation TleKyrieEzraNo ratings yet
- 2.03.01 Vaccines New TechDocument10 pages2.03.01 Vaccines New TechPrabhu LancerNo ratings yet
- BacteriaDocument3 pagesBacteriaburgerNo ratings yet
- The New Normal of Education in the Philippines During COVID-19Document3 pagesThe New Normal of Education in the Philippines During COVID-19Lovely GrospeNo ratings yet
- Rotogravure Printing Machine, Flexo Printing Machine, Coating Machine, Dry Lamination Machine, Solvent-Less Laminator Suppliers, ShaanxibeirenDocument6 pagesRotogravure Printing Machine, Flexo Printing Machine, Coating Machine, Dry Lamination Machine, Solvent-Less Laminator Suppliers, ShaanxibeirenFoufou IshakNo ratings yet
- Biology Cbse Class 12th Investigatory ProjectDocument33 pagesBiology Cbse Class 12th Investigatory ProjectRajesh BiswalNo ratings yet
- Knowledge, Attitude, and Practice Regarding Sterilization Among Health-Care Staff in A Tertiary Hospital of Western IndiaDocument6 pagesKnowledge, Attitude, and Practice Regarding Sterilization Among Health-Care Staff in A Tertiary Hospital of Western IndiaVin BitzNo ratings yet
- 69-RIAM FinalDocument3 pages69-RIAM FinalSoraya Eugenia Morales LópezNo ratings yet
- 1 Historical Overview and Future Perspective: Bernhard Eikmanns, Marcella Eikmanns, and Christopher J. PaddonDocument22 pages1 Historical Overview and Future Perspective: Bernhard Eikmanns, Marcella Eikmanns, and Christopher J. PaddonDr. Laxman RautNo ratings yet
- Chapter 65 (Introduction To Viruses)Document16 pagesChapter 65 (Introduction To Viruses)leoNo ratings yet
- Akkermansia Wikipedia Page Safari - Sep 18 2022Document1 pageAkkermansia Wikipedia Page Safari - Sep 18 2022Rodrigo FerreiraNo ratings yet
- Jurnal Internasional Asuhan Keperawatan Gerontik Dengan DiareDocument13 pagesJurnal Internasional Asuhan Keperawatan Gerontik Dengan Diarefandhik setyawanNo ratings yet
- Clinical Bacteriology Lab Gram Staining ProcedureDocument5 pagesClinical Bacteriology Lab Gram Staining ProcedureDavid WolfyNo ratings yet
- Feline Panleukopenia Balram YadavDocument16 pagesFeline Panleukopenia Balram Yadavsanchi ranaNo ratings yet
- Who Guidelines For The Treatment of Treponema Pallidum (Syphilis) - Block 5 - Blue TeamDocument32 pagesWho Guidelines For The Treatment of Treponema Pallidum (Syphilis) - Block 5 - Blue TeamSheena PasionNo ratings yet
- Dengue Fever Facts: Causes, Symptoms & PreventionDocument35 pagesDengue Fever Facts: Causes, Symptoms & PreventionSanjay NavaleNo ratings yet