Professional Documents
Culture Documents
1996 Cellular Immunology CD38
Uploaded by
Andrea la SalaCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
1996 Cellular Immunology CD38
Uploaded by
Andrea la SalaCopyright:
Available Formats
CELLULAR IMMUNOLOGY 173, 192–197 (1996)
ARTICLE NO. 0267
Secretion of IFN-g, IL-6, Granulocyte-Macrophage Colony-Stimulating
Factor and IL-10 Cytokines after Activation of Human Purified T
Lymphocytes upon CD38 Ligation
CLARA M. AUSIELLO,*,† ANDREA LA SALA,* CARLO RAMONI,‡ FRANCESCA URBANI,*
ADA FUNARO,§ AND FABIO MALAVASIØ
Departments of *Bacteriology and Medical Mycology and ‡Immunology, Istituto Superiore di Sanita’, 00161 Rome, Italy; †Institute CNR
of Tissue Typing and Dialysis, 67100 L’Aquila, Italy; §Laboratory of Cell Biology, Department of Genetics, Biology, and Medical
Chemistry, University of Torino, Torino Italy; and ØInstitute of Biology and Genetics, University of Ancona, Ancona, Italy
Received May 29, 1996; accepted July 16, 1996
Like other surface receptors, human CD38 can be
Human CD38, a surface glycoprotein expressed by shed: indeed, a soluble CD38 molecule was found both
different immunocompetent cells, is associated with in culture supernatants of activated T lymphocytes and
distinct transmembrane signaling molecules and plays tumor cell lines and in vivo in biological fluids from
a key role in the synthesis of cyclic ADP-ribose, a cal- healthy and diseased individuals (12, 13).
cium-mobilizing compound. This study reports that Last, CD38 is also a complex bifunctional ectoen-
CD38 ligation by specific monoclonal antibodies (mAb) zyme, catalyzing the formation of cyclic ADP-ribose
in purified peripheral blood T cells is followed by se- (cADPR), a second messenger involved in the regula-
cretion of discrete cytokines. IL-6, granulocyte-macro- tion of calcium-induced calcium release flux in several
phage colony-stimulating factor (GM-CSF), IFN-g, and cell systems (14–16). A recently defined ligand for hu-
IL-10 mRNA expression were constant findings. Low man CD38 molecule adds further complexity in placing
levels of IL-2 mRNA were also detected in CD38-acti- all these observations within the context of a surface
vated T lymphocyte cultures of all subjects studied. receptor (17).
Low levels of IL-4 and IL-5 mRNA were detected in the Some of the results attributed to CD38 ligation could
majority of CD38-activated T cultures. Moreover, CD38
be due to factors following immune cells activation via
mediated cytokine induction does not require T cell
CD38. In this context, cytokines released after CD38
proliferation or the addition of antigen presenting
activation could be responsible for some of the features
cells. In conclusion, human CD38 runs an activation
pathway in purified T cells which operates through
attributed to this pleiotropic molecule (1, 3, 6). A previ-
the induction of a cytokine profile shared by Th1 or ous study has shown that CD38 triggering induces the
Th2 cells. q 1996 Academic Press, Inc. expression of a discrete pattern of cytokine in cultured
peripheral blood mononuclear cells (PBMC) (18). Here
we have evaluated the effects mediated by human
CD38 in purified resting T cells analyzed in terms of
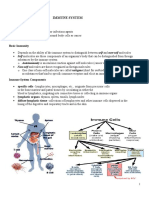
INTRODUCTION
messages and released cytokines. The interplay be-
tween CD38 and its ligand (17) has been simulated in
Human CD38 is a type II transmembrane glycopro- vitro by means of the specific IB4 mAb, an agonistic
tein prevalently expressed by immature and activated anti-CD38 reagent (19).
T and B lymphocytes, plasma cells, monocytes, periph-
eral blood NK cells, and, at lower epitope density, by MATERIAL AND METHODS
other cells and tissues (1–6). One of its unique charac-
teristics is the ability to perform as coreceptor, featur- Monoclonal antibodies. IB4 (anti-CD38) and CBT3G
ing activation and proliferation signals in lymphocytes (anti-CD3) mAb were prepared (7, 19) and purified by
(1, 3). It is physically associated (and involved in signal affinity chromatography on protein A–Sepharose
transduction) with the surface receptors TcR/CD3, sur- (Pharmacia, Uppsala, Sweden) and HPLC or hydroxyl-
face Ig/CD19, and CD16, which are crucial molecules apatite (19). The batches of mAb used were endotoxin-
for the function of T, B, and NK cells, respectively (6– free, as tested by reaction with the endotoxin kit
10). Second, it has been reported that CD38 mediates (Sigma, St. Louis, MO).
a selectin-like binding to endothelial cells, thus acting Preparation of PBMC and purified T lymphocytes.
as an adhesion molecule (11). PBMC were obtained from heparinized venous periph-
0008-8749/96 $18.00 192
Copyright q 1996 by Academic Press, Inc.
All rights of reproduction in any form reserved.
AID CI 1005 / 6c15$$$221 09-27-96 12:30:12 cia AP: Cell Immuno
CD38 LIGATION AND CYTOKINE PRODUCTION IN T CELLS 193
eral blood samples from healthy donors by centrifuga- at 627C, and 1 min extension at 727C. The PCR cycle
tion on density gradient (Lymphoprep, Nyegaard, Oslo, numbers for each primer pair, optimized to avoid the
Norway). PBMC were washed twice in RPMI 1640 me- PCR plateau effects (23), were 28 for IL-6, GM-CSF,
dium (GIBCO BRL, Life Technologies, Inc., Gaithers- IL-2, and IFN-g and 30 for IL-4, IL-5, and IL-10. The
burg, MD) and resuspended at 2 1 106/ml in RPMI reaction product was visualized by electrophoresis us-
1640 medium supplemented with 5% pooled AB serum ing 10 ml of the reaction mixture. The specificity of
and antibiotics [Penicillin (100 IU/ml), Streptomycin amplified cDNA sequences was validated by correspon-
(0.1 mg/ml); GIBCO], hereafter referred to as complete dence to predicted size of genes and by restriction map-
medium (18). T cells were purified as previously de- ping (25). After electrophoresis, the relative density of
scribed (7, 20). Briefly, PBMC were deprived of adher- the ethidium bromide stained PCR reaction products
ent cells by incubation on nylon wool columns, T cells was determined by using a Pharmacia-LKB Ultrascan
were then purified by positive selection using AET- XL densitometer. The actual densitometric values for
SRBC (experiments 1 and 2) (7) or by immunomagnetic each cytokine mRNA were normalized, using b-actin
negative selection with Dynabeads magnetic particles densitometry as the 100% reference and all other val-
(Dynal, Oslo, Norway) conjugated with the relevant ues expressed as the percentage of this figure (23).
mAb to eliminate B lymphocytes (Dynabeads M-450
Immunofluorescence analysis. Single or double im-
Pan-B (CD19), monocytes (Dynabeads M-450 CD14),
munofluorescence was performed using fluorescein
and NK cells (B73.1 and 3G8 mAb and Dynabeads M-
isothiocyanate (FITC)- or phycoerythrin (PE)-labeled
450 sheep anti-mouse IgG) (experiments 3–6) (20).
anti-CD3, anti-CD14, and anti-CD19 mAb (Becton
Culture conditions and proliferation assays. Cyto- Dickinson, Mountain View, CA). Cells (5000) were ana-
kine mRNA in cultured cells from PBMC and T lym- lysed on a FACScan flow cytometer (Becton Dickinson)
phocytes were measured in the presence of agonistic gated to exclude non viable cells.
mAb, i.e., anti-CD38 (IB4) and anti-CD3 (CBT3G) used
at predetermined optimal doses (20 mg/ml and 50 ng/ RESULTS
ml, respectively) (18). To isolate total RNA, cells were
cultured in 13-ml tubes (Falcon, Becton Dickinson, Lin- Flow cytometric analysis and proliferation of puri-
coln Park, NJ) at 2 1 106 cells/ml in 3 ml complete fied T cell populations. The working hypothesis was
medium for 18 hr at 377C in a 5% CO2 . Culture super- that CD38 triggering is followed by a direct induction
natants were collected and used to measure IFN-g, IL- of cytokine production in purified T cells, even in the
5, IL-6, and IL-10 cytokine secretion by ELISA tests absence of antigen presenting cells. Table 1 shows
(Quantikine, R&D Systems, Minneapolis, MN). ELISA the results of the proliferation of purified T cells and
sensitivities were 1 pg/ml for IL-5, 1.5 pg/ml for IL- PBMC, from the six healthy donors studied, in re-
10, and 3 pg/ml for IL-6 and IFN-g. PBMC and T cell sponse to CD38 or CD3 stimulations. The same table
proliferations were measured in triplicate in 2 1 105 includes the phenotypic characterization of purified
cells in 0.2 ml complete medium in 96-well flat-bottom T cells obtained either by positive selection (experi-
trays (Falcon). Trays were incubated in 5% CO2 at 377C ments 1 and 2) or by negative selection (experiments
and harvested on Day 4, after 18 hr culture in the 3 – 6). Both methods yielded an enrichment in T cells
presence of 0.5 mCi/well of [3H]thymidine (NEN Du of approximately 90% (range 88 – 96%). The ability of
Pont, Dreieich, Germany). Data are expressed as mean CD38 or CD3 triggering to induce proliferation in
cpm 1 1003 { SD (18). purified T cells was correlated to the presence of con-
taminating monocytes in the T cell preparations. For
Polymerase chain reaction (PCR)-assisted mRNA
example, a contamination of 2% of CD14 cells in T
amplification. Total cellular RNA was extracted fol-
cell preparation gives a 30% residual proliferation
lowing the guanidium–isothiocynate method (21).
compared to that induced in PBMC. As already re-
RNA (1 mg in a 20-ml reaction volume) was transcribed
ported (7, 18), CD38 stimulation was able to induce
by using Moloney murine leukemia virus reverse tran-
PBMC to proliferate in all the experiments per-
scriptase, as previously reported (22) and following the
formed. The CD38-stimulated PBMC proliferation
manufacturer’s instructions (GIBCO BRL). PCR was
was at least 38 times the proliferation of unstimu-
carried out in a 20-ml volume, using 2 ml of cDNA, di-
lated cultures. However, the degree of PBMC prolif-
luted 1:10 (GeneAmpkit, Perkin Elmer Cetus, Nor-
eration was equal to or lower than that obtained via
walk, CT) (23). The PCR amplification was carried out
CD3 stimulation in the same donor (Table 1).
starting from as low as 1 ng of original RNA. Cytokine
specific primer pairs were synthesized according to Comparative analysis of cytokine mRNA expression
published sequences (DNA Synthesizer, Applied Bio- in cultured T cells and PBMC. Figure 1 reports a rep-
systems, Inc., Foster City, CA) (24, 25). PCR was per- resentative profile of semiquantitative RT-PCR assays
formed in a 9600 Perkin Elmer thermal cycler, each of the cytokine mRNA expression in cultured purified
cycle with 40 s denaturation at 947C, 40 s annealing T cells and control PBMC challenged with anti-CD38
AID CI 1005 / 6c15$$$222 09-27-96 12:30:12 cia AP: Cell Immuno
194 AUSIELLO ET AL.
TABLE 1
Phenotypic Characterization and Proliferation of Purified Resting T Cells upon Ligation of CD38
T cell proliferationb PBMC proliferationb
Phenotypea [3H]thymidine incorporation [3H]thymidine incorporation
(% positive cells) (cpm { SD 1 103) (cpm { SD 1 103)
Experiment CD3 CD19 CD14 CD38 CD3 Control CD38 CD3 Control
1 95 õ1 1.2 13.3 { 2.1 (32)c 9.6 { 1.1 (23) 0.1 { 0.0 41.0 { 2.8 41.5 { 1.3 0.6 { 0.1
2 95 2 2 12.6 { 0.9 (30) 13.5 { 2.1 (32) 0.1 { 0.0 42.5 { 3.1 42.4 { 2.1 0.6 { 0.1
3 93 õ1 2 9.5 { 3.8 (18) 43.1 { 3.3 (34) 0.1 { 0.0 53.8 { 4.6 125 { 3.3 0.3 { 0.1
4 88 1.5 õ1 0.2 { 0.1 (5) 0.1 { 0.0 (0.0) 0.1 { 0.0 3.8 { 0.3 16.0 { 1.4 0.1 { 0.0
5 93 õ1 0 0.6 { 0.2 (1) 0.8 { 0.0 (0.1) 0.2 { 0.0 41.6 { 2.7 96.2 { 8.5 0.3 { 0.2
6 96 0 0 1.0 { 0.2 (3) 2.5 { 1.1 (7) 0.1 { 0.0 38.1 { 2.5 37.7 { 2.9 0.2 { 0.0
a
Determined by FACS analysis as specified under Materials and Methods.
b
Proliferation was measured after 4-day culture as specified under Materials and Methods.
c
Percentage residual proliferation of T cells as compared to PBMC.
mAb; the control was the analysis of effect induced by based on previous experience which indicated that the
anti-CD3 mAb. These experiments were performed in peak production of the cytokines under analysis is ap-
cells obtained from the same donor. Figure 2 shows the parent at 6 and 24 hr in culture (18).
cumulative results of the comparative analysis in T CD38-mediated signaling constantly induced all T
cells and PBMC, performed on cells derived from the cell cultures to express mRNAs for IL-6, GM-CSF, IFN-
5 individuals included in the RT-PCR analysis. The g, IL-10, and IL-2, the latter at very low level. IL-4 and
data are referred to the intensity of specific transcripts, IL-5 were expressed at low levels in the majority of
expressed as percentage of density relative to that of CD38-activated T cell cultures (four of five and three
b-actin, as determined by densitometry (23). The of five, respectively). As already reported, CD38 trig-
mRNA was extracted after 18 hr of mAb challenge, gering induced all PBMC tested to express IL-6, GM-
CSF, and IL-10, while IFN-g was expressed by PBMC
in the majority of individuals (4/5) activated via CD38.
CD38-activated PBMC of the sample studied displayed
very low levels of mRNA transcripts of IL-2 (4/5), IL-4
(4/5), and IL-5 (3/5).
Comparing relative intensities of the transcripts in
purified T cells and PBMC, it was apparent that CD38-
activated T cells displayed an accumulation of mes-
sages for IFN-g mRNA, while those for IL-6 and IL-10
mRNA were decreased.
After activation via CD3, the messages for GM-CSF,
IFN-g, and IL-10 mRNA were apparent in cultured T
cells of all the donors tested, while the messages for
IL-6, IL-2, IL-4, and IL-5 mRNA were detected in the
majority of the donors (two of three). IL-6, GM-CSF,
IFN-g, IL-2, IL-4, IL-5, and IL-10 were expressed in
CD3-activated PBMC from all the donors tested, con-
firming previous results (18). The analysis of the rela-
tive intensities of the transcripts showed a decrease of
IL-6, IL-2, IL-4, IL-5, and IL-10 mRNA in CD3-acti-
FIG. 1. Profile of cytokine mRNA expression in CD38-activated vated T cells compared to that seen in PBMC samples.
purified T cells and PBMC. Purified T cells and PBMC from a repre-
sentative donor were cultured in the presence of anti-CD38 (20 mg/ No relevant differences were seen in the ability of
ml) or anti-CD3 (50 ng/ml) mAb or unstimulated. After 18 hr, the CD38 ligation to induce cytokines as compared to CD3
cells were harvested and RNA was extracted and reversetranscribed. signaling, except for the expression of IL-6 mRNA,
The cDNA obtained was assayed for the presence of specific cytokine which was more consistent in CD38-activated T cells.
mRNA by PCR, as described under Materials and Methods. Reaction
products were run on 1% agarose gel in the presence of appropriate
This finding confirms previous experience with PBMC
molecular weight markers (arrow). b-actin was used as the positive cultures (18).
control. Untreated T cell cultures showed a constitutive
AID CI 1005 / 6c15$$$222 09-27-96 12:30:12 cia AP: Cell Immuno
CD38 LIGATION AND CYTOKINE PRODUCTION IN T CELLS 195
general concordance in the analyses of the cytokines
evaluated in terms of mRNA expression or release in
the supernatants. IL-6, IL-10, and IFN-g were consis-
tently found in culture supernatants of CD38-activated
purified T cells and the control PBMC. Similar results
were found in CD3-activated T cells and PBMC. IL-5
levels were almost not detectable in supernatants of
CD38-activated T cells or PBMC, but approached the
lower detection limits of the assay in CD3-activated
PBMC.
More striking differences were noted after compara-
tive analysis of released IL-6 and IL-10, cytokines
which are produced by monocytes and T cells. A
marked reduction in IL-6 and IL-10 secretion was in-
deed observed in CD38- or CD3-activated T cells as
compared to PBMC challenged in similar ways. Other
differences included an increased release of IFN-g in
CD38-activated T cells, as compared to PBMC acti-
vated in similar fashions. The comparison with the
same cell preparations in the absence of any ligation
showed that purified T cells display detectable levels
of IL-6 and IFN-g, while PBMC were characterized by
the presence of IL-6 and IL-10 in the supernatants.
DISCUSSION
CD38 stimulation specifically induces purified rest-
FIG. 2. Cumulative results of cytokine mRNA expression in puri-
ing T lymphocytes to express the messages for cyto-
fied T cells upon CD38 ligation. Purified T cells and PBMC from five kines. The profile of cytokines induced in T cells via
donors were cultured in the presence of anti-CD38 (20 mg/ml) or anti- CD38 was similar to that observed after CD3 signaling:
CD3 (50 ng/ml) mAb or unstimulated. After 18 hr, the cells were one relevant difference was the higher secretion of IL-
harvested and RNA was extracted and reverse transcribed. The 6 in T cells, confirming the results obtained in PBMC
cDNA obtained was assayed for the presence of specific cytokine
mRNA by PCR, as described under Materials and Methods. Reaction
(18). Cytokine secretion was characterized by higher
products were run on 1% agarose gel in the presence of appropriate levels of IFN-g in T cell cultures upon CD38-ligation
molecular weight markers (arrow). b-Actin was used as the positive than in PBMC similarly treated.
control. Data are expressed as percentage absorbance (mean { SEM) This study reveals two important facets of human T
using b-actin as comparative reference. lymphocytes activated via CD38. First, cytokine pro-
duction is not associated with T cell proliferation, indi-
cating a dissociation of the two events. Second, CD38
mRNA expression of GM-CSF and IFN-g even if only activation, like CD3 activation, induces a cytokine se-
in very low amounts. PBMC, in similar conditions, dis- cretion profile, which includes Th1- and Th2-type cyto-
played a constitutive expression of IL-6, IFN-g, IL-4, kines (26).
and IL-10 mRNA in the majority of cultures tested, but Approximately 5% of freshly isolated T cells are
the expression of these cytokines was clearly lower as CD38/ (6, 13) and these are usually CD45RA/ (11).
compared to the messages implemented by CD38 and Thus CD38 cells are actually mature resting T cells,
CD3 ligation. which respond poorly in vitro to stimuli acting via TcR/
CD3 (27, 28). The expression of CD38 in T cells is ap-
Comparative analysis of cytokine secretion by puri- proximately 10-fold lower than in activated T lympho-
fied T cells and PBMC in culture. The secretion of IL- cytes and plasma cells (11).
6, IL-5, IL-10, and IFN-g was measured by ELISA in A possible working hypothesis for explaining the in-
supernatants of CD38- and CD3-stimulated purified T duction of cytokine secretion via CD38 activation of
cells from the subjects studied in PCR-assisted mRNA resting mature T cells stems from similar experiences
amplification using PBMC as reference (Table 2). The derived from CD27–CD70 interaction (29, 30). CD27 is
supernatants were collected after 18 hr of culture, and a transmembrane glycoprotein expressed by peripheral
the data are expressed either as picograms per millili- T cells, medullary T lymphocytes, B cells, and NK cells.
ter or as the number of positive cultures over the total Similar to CD38, CD27 is expressed preferentially in
cultures examined. One conclusion is that there is a CD45RA/ subsets of naive CD4/ T cells (29). The ligand
AID CI 1005 / 6c15$$$222 09-27-96 12:30:12 cia AP: Cell Immuno
196 AUSIELLO ET AL.
TABLE 2
Cytokine Release in Purified T Cell upon CD38 Ligationa
CD38 CD3 None
T cells PBMC T cells PBMC T cells PBMC
IL-6 430 { 226 2/2 654 { 151 2/2 172 { 14** 2/2 514 { 30** 2/2 81 { 68 2/2 225 { 174 2/2
IFN-g 1048 { 370 4/4 894 { 402 4/4 766 { 361 3/4 1363 { 124 4/4 83 { 66 2/3 7{6 2/4
IL-5 0 0/2 0.6 1/2 0.3 1/2 4 2/2 0 0/2 0 0/2
IL-10 53 { 33* 2/2 519 { 102* 2/2 99 { 73 2/2 306 { 97 2/2 14 { 5 2/2 79 { 63 2/2
a
Immunoenzymatic assay of cytokine production in supernatants (18 hr) of CD38-activated T cells. PBMC and CD3 activation were
included as the control reference, while background was evaluated by addition of culture medium alone. Data are expressed as pg/ml (mean
{ SEM) and as the number of positive supernatants over total cultures examined.
* Student t test P Å 0.02.
** Student t test P Å 0.004.
for CD27 has been recently identified in the CD70 mole- nal transduction (12, 36). High levels of soluble CD38
cule. CD70 is induced after activation on the CD45RO/ molecules are found in biological fluids of myeloma pa-
T cell. Morimoto et al. (30) provided evidence for di- tients (12). IL-6 is also reported to play a role in HIV
rect cellular communication between CD45RO/ and pathophysiology: increased levels of the cytokine are
CD45RA/ T cell subsets through CD27/CD70 ligation. detected in the serum of patients (37–39) while in-
The contact is direct and leads to activation of creased intratumoral expression of this cytokine is
CD45RA/ T cells, as indicated by the upmodulation of found in AIDS lymphomas (40). Another symptom in
CD25 (30). In the culture conditions adopted in the AIDS patients with poor prognosis is the presence of
present study, CD38 ligation might mimic the single or high levels of soluble CD38 in the serum (14). Shedding
multiple ligands and directly activate CD45RA/ cells, of CD38 is related to cell activation or interaction with
promoting the activation of CD45RO/ cells. cytokines, and it can be an indication that this soluble
The secretion of significant amounts of cytokines form serves as a receptor. The finding that elevated
upon CD38 ligation either by Th1 (i.e., IL-2, IFN-g) or amounts of IL-6 and soluble CD38 molecules are simul-
by Th2 subsets (i.e., IL-6, IL-10) suggests the broad taneously presents in different pathological situations
activation of the molecule in T cells, a characteristic can be an indication of a connection between the two
shared with CD3. Actually, the ability of a T cell to events. However, further studies are needed to confirm
secrete Th1 or Th2 cytokines upon a same signal is not and explain this work hypothesis.
surprising. In fact, it was recently reported that IL-12
is able to directly activate CD45RO//CD30/ cells and ACKNOWLEDGMENTS
to induce the secretion of IFN-g (31). Nonetheless,
CD30 T cells produce IL-5, which is generally consid- This work was supported by grants from the Special Project ACRO
ered to be a Th2 cytokine, thus indicating that CD30/ (Consiglio Nazionale delle Ricerche, Rome, Italy), AIDS and ‘‘Pro-
cells exhibit properties of both Th1 and Th2 cells (32). getto Nazionale Tubercolosi’’ (Istituto Superiore di Sanita’, Rome,
These data coincide with recent views that consider the Italy), Telethon (Rome, Italy), and AIRC (Milano, Italy).
clear-cut division of cytokine producing T cells into Th1
and Th2 subsets as an oversimplification (33). There is REFERENCES
strong evidence that a single naive peripheral T cell
can yield progeny of either Th1 or Th2 type, depending 1. Malavasi, F., Funaro, A., Roggero, S., Horenstein, A., Calosso,
upon the signals received during and after activation L., and Mehta, K., Immunol. Today 15, 95, 1994.
(34, 35). 2. Jackson, D. G., and Bell, J. I., J. Immunol. 144, 2811, 1990.
A constant finding in our studies on cytokine induc- 3. Lund, F. Solvason, N., Grimaldi, J. C., Parkhouse, R. M., and
tion in immunocompetent cells activated via CD38 is Howard, M., Immunol. Today 16, 469, 1995.
the overinduction of IL-6 gene expression. IL-6 is classi- 4. Mizuguchi, M., Otsuka, N., Sato, M., Ishii, Y., Kon, S., Yamada,
M., Nishina, H., Katada, T., and Ikeda, K., Brain. Res. 697,
cally secreted by Th2 cells and among its many pleio- 235, 1995.
tropic effects, IL-6 is also a myeloma cell growth factor 5. Ramaschi, G., Torti, M., Tolnai Festetics, E., Sinigaglia, F., Ma-
in vitro and in vivo (12). This cytokine is overproduced lavasi, F., and Balduini, C., Blood 87, 2308, 1996.
in association with a poor prognosis in myeloma pa- 6. Mehta, K., Shahid, U., and Malavasi, F., FASEB J., in press.
tients. In such patients the differentiation of B cells 7. Funaro, A., Spagnoli, G. C., Ausiello, C. M., Alessio, M., Rog-
into high rate Ig secreting mature plasma cells is im- gero, S., Delia, D., Zaccolo, M., and Malavasi, F., J. Immunol.
paired, likely due to abnormality in IL-6-mediated sig- 145, 2390, 1990.
AID CI 1005 / 6c15$$$222 09-27-96 12:30:12 cia AP: Cell Immuno
CD38 LIGATION AND CYTOKINE PRODUCTION IN T CELLS 197
8. Zupo, S., Rugarli, E., Dono, M., Tamborelli, G., Malavasi, F., 23. La Sala, A., Urbani, F., Lande, R., and Ausiello, C. M., Minerva
and Ferrarini, M., Eur. J. Immunol. 24, 1218, 1994. Biotec. 7, 207, 1995.
9. Funaro, A., DeMonte, L. B., Dianzani, U., Forni, M., and Mala- 24. Ehlers, S., and Smith, K. A., J. Exp. Med. 173, 25, 1991.
vasi F., Eur. J. Immunol. 23, 2407, 1993. 25. Spagnoli, G. C., Juretic, A., Schultz-Thater, E., Dellabona, P.,
10. Kumagai, M., Coustan-Smith, E., Murry, D. J., Silvennoinen, Filgueira, L., Horig, H., Zuber, M., Garotta, G., and Herberer,
O., Murti, G. P., Evans, D. J., Malavasi, F., and Campana, D., M., Cell. Immunol. 146, 391, 1992.
J. Exp. Med. 181, 1101, 1995. 26. Fraser, J. D., Straus, D., and Weiss, A., Immunol. Today 14,
11. Dianzani, U., Funaro, A., DiFranco, D., Garbarino, G., Bra- 357, 1993.
gardo, M., Redoglia, V., Buonfiglio, D., DeMonte, L., Pileri, A., 27. Beverley, P. C. L. Semin. Immunol. 4, 35, 1992.
and Malavasi, F., J. Immunol. 153, 952, 1994. 28. Bell, E. B., Semin. Immunol. 4, 43, 1992.
12. Klein B., Zhang X. G., Lu, Z. Y., and Bataille, R., Blood 85, 863, 29. Maurer, D. W., Holter, W., Majdic, O., Fisher, G. F., and Knapp,
1995. W., Eur. J. Immunol. 20, 2679, 1990.
13. Funaro, A., Horestein, A. L., and Malavasi, F., Fundam. Clin. 30. Agematsu, K., Kobata, T., Sugita, K., Hirose, T., Scholossman,
Immunol. 3, 101, 1995. S. F., and Morimoto, C., J. Immunol. 154, 3627, 1995.
14. Howard, M., Grimaldi, J. C., Baxer, F. J., Lund, F. E., Santos- 31. Alzona, M., Jack, H.-M., Fisher, R. I., and Ellis, T. M., J. Immu-
Argumedo, L., Parkhouse, R. M. E., and Walseth, T. F., Science nol. 153, 2861, 1994.
262, 1056, 1993. 32. Mosmann, T. R., Cherwinski, H., Bond, M. W., Giedlin, M. A.,
15. Zocchi, E., Franco, L., Guida, L., Benatti, U., Bargellesi, A., and Coffman, R. L., J. Immunol. 136, 2348, 1990.
Malavasi, F., Lee, H. C., and De Flora, A., Biochem. Biophys. 33. Kelso, A., Immunol. Today 16, 374, 1995.
Res. Commun. 196, 1459, 1993. 34. Rocken, M., Saurat, J-K., and Hauser, C., J. Immunol. 148,
16. Gallione, A., Science 259, 325, 1993. 1031, 1992.
35. Sad, S., and Mosman, T. R., J. Immunol. 153, 3514, 1994.
17. Deaglio, S., Dianzani, U., Horestein, A. L., Fernandez, J. E.,
Van Kooten, C., Bragardo, M., Garbarino, G., Funaro, A., DiVir- 36. Reibnegger, G., Krainer, M., Herold, M., Ludwig, H., Watcher,
gilio, F., Banchereau, J., and Malavasi, F., J. Immunol. 156, H., and Huber, H., Cancer Res. 51, 6250, 1991.
727, 1996. 37. Birx, D. L., Redfield, R. R., Tencer, K., Fowler, A., Burke,
18. Ausiello, C. M., Urbani, F., laSala, A., Funaro, A., and Malavasi, D. S., and Tosato, G., Blood 76, 2303, 1990.
F., Eur. J. Immunol. 25, 1477, 1995. 38. Marfaing-Koka, A., Aubin, J. T., Grangeot-Keros, L., Portier,
A., Benattar, C., Merrien, D., Agut, H., Aucouturier, P., Autran,
19. Alessio, M., Roggero, S., Funaro, A., De Monte, L., Peruzzi, L.,
B., Wijdenes, J., Galanaud, P., and Emilie, D., J. AIDS Hum.
Geuna, M., and Malavasi, F., J. Immunol. 145, 878, 1990.
Retrovirus 11, 59, 1996.
20. Arancia, G., Molinari, A., Crateri, P., Stringaro, A., Ramoni, C., 39. Breen, E. C., Rezai, A. R., Nakajima, K., Beall, G. N., Mitsuy-
Dupuis, M. L., Gomez MJ., Torosantucci A., and Cassone, A., asu, R. T., Hirano, T., Kishimoto, T., and Martinez-Maza, O.,
Infect. Immun. 63, 280, 1995. J. Immunol. 144, 480, 1990.
21. Chomcznski, P., and Sacchi, N., Anal. Biochem. 162, 156, 1987. 40. Emile, D., Fior, R., Llorente, L., Marfaing-Koke, A. I., Peuch-
22. Ausiello, C. M., Urbani, F., Gessani, S., Spagnoli, G. C., Gomez, maur, M., Devergne, O., Jarrousse, B., Wijdenes, J., Bove, F.,
M. J., and Cassone, A., Infect. Immun. 61, 4105, 1993. and Galanaud, P., Immunol. Rev. 140(5), 1994.
AID CI 1005 / 6c15$$$223 09-27-96 12:30:12 cia AP: Cell Immuno
You might also like
- Co-signal Molecules in T Cell Activation: Immune Regulation in Health and DiseaseFrom EverandCo-signal Molecules in T Cell Activation: Immune Regulation in Health and DiseaseMiyuki AzumaNo ratings yet
- CTLA4 Is A Second Receptor For The B Cell Activation Antigen B7Document9 pagesCTLA4 Is A Second Receptor For The B Cell Activation Antigen B7Fernanda RibeiroNo ratings yet
- J Exp Med 1991 Linsley 721 30Document10 pagesJ Exp Med 1991 Linsley 721 30Fernanda RibeiroNo ratings yet
- C-Rel Is Crucial For Lymphocyte Proliferation But Dispensable For T Cell Effector FunctionDocument11 pagesC-Rel Is Crucial For Lymphocyte Proliferation But Dispensable For T Cell Effector FunctionFrontiersNo ratings yet
- 13 Full PDFDocument18 pages13 Full PDFWillyPeñaNo ratings yet
- Hogg 1999 (c.817G-A)Document10 pagesHogg 1999 (c.817G-A)Syed Irfan RazaNo ratings yet
- Ek 2006Document6 pagesEk 2006ᄋᄋNo ratings yet
- 9747-Supplementary Appendix-74841-1-10-20210329 PDFDocument19 pages9747-Supplementary Appendix-74841-1-10-20210329 PDFTianliang GuoNo ratings yet
- Spittler Et Al. - 1997 - A Glutamine Deficiency Impairs The Function of CulDocument3 pagesSpittler Et Al. - 1997 - A Glutamine Deficiency Impairs The Function of CulMATHILDE MAGRONo ratings yet
- 135 FullDocument9 pages135 FullDhila FadhilaNo ratings yet
- 1 s2.0 S1043466607001494 MainDocument6 pages1 s2.0 S1043466607001494 MainKevin MaiseyNo ratings yet
- CD Marker HandbookDocument47 pagesCD Marker HandbookSteph Vee100% (1)
- CD Markers HandbookDocument48 pagesCD Markers HandbookOmair Riaz100% (2)
- CD27 Ligand Regulates Lymphocyte FunctionDocument10 pagesCD27 Ligand Regulates Lymphocyte FunctionactivnetNo ratings yet
- IL-2 Trans-Presentation by DCs Revealed by Daclizumab TherapyDocument12 pagesIL-2 Trans-Presentation by DCs Revealed by Daclizumab TherapydavdavdavdavdavdavdaNo ratings yet
- 2010 ER Stress Induction in T CellDocument6 pages2010 ER Stress Induction in T CellRaul ReyesNo ratings yet
- A Three-Symbol Code For Organized Proteomes Based On Cyclical Imaging of Protein LocationsDocument9 pagesA Three-Symbol Code For Organized Proteomes Based On Cyclical Imaging of Protein LocationsJulio YarNo ratings yet
- Cells 09 00417 PDFDocument36 pagesCells 09 00417 PDFMilos CuculovicNo ratings yet
- Antibody Binding Profile of Purified and Cell-Bound CD26 1993Document14 pagesAntibody Binding Profile of Purified and Cell-Bound CD26 1993heyligenNo ratings yet
- Pnas 201717789siDocument11 pagesPnas 201717789siygilad9139No ratings yet
- Activation of CD8 T Cells Induces Expression of CD4, Which Functions As A Chemotactic ReceptorDocument6 pagesActivation of CD8 T Cells Induces Expression of CD4, Which Functions As A Chemotactic ReceptorKevin MaiseyNo ratings yet
- CD107 NK Cell 2004Document8 pagesCD107 NK Cell 2004Lu LizNo ratings yet
- A Role For Dicer in Immune Regulation: ArticleDocument9 pagesA Role For Dicer in Immune Regulation: ArticleSwati2013No ratings yet
- Construction and functional research of fourth-generation CAR-T cells targeting CD138Document10 pagesConstruction and functional research of fourth-generation CAR-T cells targeting CD138Tianliang GuoNo ratings yet
- Alter 2004Document8 pagesAlter 2004Ivan VazquezNo ratings yet
- Lentiviral Vector-Mediated Correction of A Mouse Model of Leukocyte Adhesion Deficiency Type IDocument11 pagesLentiviral Vector-Mediated Correction of A Mouse Model of Leukocyte Adhesion Deficiency Type IDeede DarkoNo ratings yet
- Flow Cytometry Analysis of Intracellular Antigens Using Saponin PermeabilizationDocument10 pagesFlow Cytometry Analysis of Intracellular Antigens Using Saponin PermeabilizationMinh TriếtNo ratings yet
- Cdi2012 820827Document9 pagesCdi2012 820827Dian ParamitaNo ratings yet
- 1 s2.0 S0014482702955813 MainDocument12 pages1 s2.0 S0014482702955813 MainKevin MaiseyNo ratings yet
- Arneth 2015 Early Activation of CD and CD T LymDocument5 pagesArneth 2015 Early Activation of CD and CD T LymCarla Pabón FloresNo ratings yet
- Flow Cytometry: An Introduction and Application To Cytology: December 2018Document12 pagesFlow Cytometry: An Introduction and Application To Cytology: December 2018ABHINABA GUPTANo ratings yet
- Differential Expression of Cyclin-Dependent Kinase 6 in Cortical Thymocytes and T-Cell Lymphoblastic Leukemia.Document9 pagesDifferential Expression of Cyclin-Dependent Kinase 6 in Cortical Thymocytes and T-Cell Lymphoblastic Leukemia.Wenhui ZhouNo ratings yet
- Agematsu 1997Document7 pagesAgematsu 1997Lizeth Rincon DelgadoNo ratings yet
- Transgenic Expression of IL 7 Regulates CAR T Cell Metabolism and Enhances in Vivo Persistence Against Tumor CellsDocument14 pagesTransgenic Expression of IL 7 Regulates CAR T Cell Metabolism and Enhances in Vivo Persistence Against Tumor Cells송애용No ratings yet
- Data SupplementDocument17 pagesData SupplementManovriti ThakurNo ratings yet
- Expression of High Amounts of The CD117 Molecule in A Case of B-Cell Non-Hodgkin's Lymphoma Carrying The T (14:18) TranslocationDocument4 pagesExpression of High Amounts of The CD117 Molecule in A Case of B-Cell Non-Hodgkin's Lymphoma Carrying The T (14:18) Translocationawdafeagega2r3No ratings yet
- 2023 Article 9940Document10 pages2023 Article 9940FCO JAVIER SANCHEZ PEÑANo ratings yet
- Effect of Cannabinoids On Anti-HIV-Tat-induced Antibody ProductionDocument1 pageEffect of Cannabinoids On Anti-HIV-Tat-induced Antibody ProductiondoctortmNo ratings yet
- Pic RenderDocument9 pagesPic Renderkora_c995No ratings yet
- Gpc3 Generate de Celule Dendritice 2010Document11 pagesGpc3 Generate de Celule Dendritice 2010grigmihNo ratings yet
- The Human Cathelicidin, LL-37, Induces Granzyme-Mediated Apoptosis in Regulatory T CellsDocument7 pagesThe Human Cathelicidin, LL-37, Induces Granzyme-Mediated Apoptosis in Regulatory T Cellsek_rzepkaNo ratings yet
- Immunology00120 0028Document7 pagesImmunology00120 0028carlitoseverectNo ratings yet
- Analysis of CD16þCD56dim NK Cells From CLL Patients Evidence Supporting ADocument9 pagesAnalysis of CD16þCD56dim NK Cells From CLL Patients Evidence Supporting Aimmunologie cellulaireNo ratings yet
- Ajtr 0151665Document12 pagesAjtr 0151665Muhammad ImranNo ratings yet
- Toll-Like Receptor 2-Mediated Dendritic Cell Activation by A Porphyromonas Gingivalis Synthetic LipopeptideDocument7 pagesToll-Like Receptor 2-Mediated Dendritic Cell Activation by A Porphyromonas Gingivalis Synthetic Lipopeptideapi-26071302No ratings yet
- tmp10C4 TMPDocument6 pagestmp10C4 TMPFrontiersNo ratings yet
- Document 1Document7 pagesDocument 1xaero1234No ratings yet
- Parcial 3 Inmunología T Cell DevelopmentDocument4 pagesParcial 3 Inmunología T Cell Developmentjuan mondaNo ratings yet
- 1995 JicrDocument9 pages1995 JicrAndrea la SalaNo ratings yet
- Cancer Immunotherapy 2020Document76 pagesCancer Immunotherapy 2020stupid.graphsNo ratings yet
- Preston1991Document5 pagesPreston1991Alberto TerronesNo ratings yet
- Expansion of NK Cells Using Genetically Engineered K562 Feeder CellsDocument8 pagesExpansion of NK Cells Using Genetically Engineered K562 Feeder Cellshoangphuong08101992No ratings yet
- 2022 Article 210Document13 pages2022 Article 210uttamNo ratings yet
- J Exp Med 1995 Krummel 459 65Document7 pagesJ Exp Med 1995 Krummel 459 65Fernanda RibeiroNo ratings yet
- LL 37Document9 pagesLL 37Te PeNo ratings yet
- Mechanism of Macrophage-Derived Chemokine Production by KeratinocytesDocument5 pagesMechanism of Macrophage-Derived Chemokine Production by Keratinocytesoscarbio2009No ratings yet
- CCL18 TextoDocument13 pagesCCL18 TextoQuimico Inmunologia GeneticaNo ratings yet
- CD Antigen Cellular Expression Functions Other NamesDocument16 pagesCD Antigen Cellular Expression Functions Other NamesrahulbafnaeNo ratings yet
- The Immune System, Fourth Edition Chapter 8: T Cell-Mediated ImmunityDocument18 pagesThe Immune System, Fourth Edition Chapter 8: T Cell-Mediated Immunitylina lopezNo ratings yet
- Pnas 2304319120 SappDocument15 pagesPnas 2304319120 SappfilymascoloNo ratings yet
- Ammar K. Daoud: Haneen Ibrahim Hamza Al-SleatiDocument10 pagesAmmar K. Daoud: Haneen Ibrahim Hamza Al-SleatiInnocent L NdambakuwaNo ratings yet
- Tanuvas ICAR GuideDocument45 pagesTanuvas ICAR GuideSanjay Kumar100% (1)
- MRCPUK Part 1 SyllabusDocument62 pagesMRCPUK Part 1 Syllabusthargyikyaw100% (5)
- Repost Train Your Brain To Boost Your Immune System Jennifer WolkinDocument4 pagesRepost Train Your Brain To Boost Your Immune System Jennifer WolkinHaisheng ZhangNo ratings yet
- CytokineDocument26 pagesCytokinePro KartuNo ratings yet
- SN4 Question Paper Sum 2006Document16 pagesSN4 Question Paper Sum 2006EmmaNo ratings yet
- 1final MCQSDocument56 pages1final MCQSahmed100% (4)
- Solastra Tech Spec Sheet BrochureDocument5 pagesSolastra Tech Spec Sheet BrochurecandiddreamsNo ratings yet
- Ebook Cellular and Molecular Immunology 9Th Edition Abbas Test Bank Full Chapter PDFDocument8 pagesEbook Cellular and Molecular Immunology 9Th Edition Abbas Test Bank Full Chapter PDFDavidLeekemt100% (11)
- Risk Factors For Herpes Zoster Infection: A Meta-Analysis: Review ArticleDocument8 pagesRisk Factors For Herpes Zoster Infection: A Meta-Analysis: Review Articleyenny handayani sihiteNo ratings yet
- Immune System: Basic ImmunityDocument14 pagesImmune System: Basic ImmunityPLANARIANo ratings yet
- Lymphatic System and Immunity - Seeleys - NotesDocument9 pagesLymphatic System and Immunity - Seeleys - NotesLol lol100% (2)
- Pathophysiology of IBDDocument11 pagesPathophysiology of IBDOktarina Heni SunandarNo ratings yet
- Kuby Immunology 7e TBDocument134 pagesKuby Immunology 7e TBStarrx71494% (18)
- The Clinical Complications of Asthma and Its Pharmacotherapy PDFDocument30 pagesThe Clinical Complications of Asthma and Its Pharmacotherapy PDFHow ToNo ratings yet
- Rh incompatibility causes and preventionDocument10 pagesRh incompatibility causes and preventionVernie Dinglasa GeligNo ratings yet
- Systemic Response To Injury and Metabolic Support, Chapter 2 of Schwartz by Anas MK HindawiDocument99 pagesSystemic Response To Injury and Metabolic Support, Chapter 2 of Schwartz by Anas MK HindawiAnas Mk Hindawi81% (16)
- BCH444 PDFDocument232 pagesBCH444 PDFYaseen AhmadNo ratings yet
- Tissue Culture Technology, 242013Document63 pagesTissue Culture Technology, 242013Chornelia DemesNo ratings yet
- Physiology ImmunityDocument75 pagesPhysiology Immunitymohani maiyaNo ratings yet
- End of Course Assessment Review PracticeDocument194 pagesEnd of Course Assessment Review PracticeyoniNo ratings yet
- Clinical Manifestation of Oral TuberculosisDocument6 pagesClinical Manifestation of Oral TuberculosisSasa AprilaNo ratings yet
- Curcumin Role in ParaproteinemiaDocument18 pagesCurcumin Role in ParaproteinemiaINDIRAKALYANINo ratings yet
- The Pathogenesis of Immune Thrombocytopaenic Purpura: ReviewDocument11 pagesThe Pathogenesis of Immune Thrombocytopaenic Purpura: ReviewNaomi ThamNo ratings yet
- Vaccines 11 00206 v2Document31 pagesVaccines 11 00206 v2sesiaNo ratings yet
- CD4+ T უჯრედების მრავალფეროვნებაDocument1 pageCD4+ T უჯრედების მრავალფეროვნებაEMD GROUPNo ratings yet
- Fungal Vaccines and ImmunotherapeuticsDocument13 pagesFungal Vaccines and Immunotherapeuticshamza aliNo ratings yet
- Blood & Immunology McqsDocument6 pagesBlood & Immunology McqsAbdullah TheNo ratings yet
- The Impact of Heat Stress On The Immune System in Dairy Cattle A ReviewDocument9 pagesThe Impact of Heat Stress On The Immune System in Dairy Cattle A Reviewciro ortizNo ratings yet
- COVID-19: Unanswered Questions On Immune Response and PathogenesisDocument5 pagesCOVID-19: Unanswered Questions On Immune Response and PathogenesisDyah PerwaNo ratings yet
- Tales from Both Sides of the Brain: A Life in NeuroscienceFrom EverandTales from Both Sides of the Brain: A Life in NeuroscienceRating: 3 out of 5 stars3/5 (18)
- When the Body Says No by Gabor Maté: Key Takeaways, Summary & AnalysisFrom EverandWhen the Body Says No by Gabor Maté: Key Takeaways, Summary & AnalysisRating: 3.5 out of 5 stars3.5/5 (2)
- Why We Die: The New Science of Aging and the Quest for ImmortalityFrom EverandWhy We Die: The New Science of Aging and the Quest for ImmortalityRating: 4 out of 5 stars4/5 (3)
- All That Remains: A Renowned Forensic Scientist on Death, Mortality, and Solving CrimesFrom EverandAll That Remains: A Renowned Forensic Scientist on Death, Mortality, and Solving CrimesRating: 4.5 out of 5 stars4.5/5 (397)
- A Brief History of Intelligence: Evolution, AI, and the Five Breakthroughs That Made Our BrainsFrom EverandA Brief History of Intelligence: Evolution, AI, and the Five Breakthroughs That Made Our BrainsRating: 4.5 out of 5 stars4.5/5 (4)
- The Molecule of More: How a Single Chemical in Your Brain Drives Love, Sex, and Creativity--and Will Determine the Fate of the Human RaceFrom EverandThe Molecule of More: How a Single Chemical in Your Brain Drives Love, Sex, and Creativity--and Will Determine the Fate of the Human RaceRating: 4.5 out of 5 stars4.5/5 (515)
- 10% Human: How Your Body's Microbes Hold the Key to Health and HappinessFrom Everand10% Human: How Your Body's Microbes Hold the Key to Health and HappinessRating: 4 out of 5 stars4/5 (33)
- Gut: the new and revised Sunday Times bestsellerFrom EverandGut: the new and revised Sunday Times bestsellerRating: 4 out of 5 stars4/5 (392)
- Inside of a Dog: What Dogs See, Smell, and KnowFrom EverandInside of a Dog: What Dogs See, Smell, and KnowRating: 4 out of 5 stars4/5 (390)
- The Ancestor's Tale: A Pilgrimage to the Dawn of EvolutionFrom EverandThe Ancestor's Tale: A Pilgrimage to the Dawn of EvolutionRating: 4 out of 5 stars4/5 (811)
- Gut: The Inside Story of Our Body's Most Underrated Organ (Revised Edition)From EverandGut: The Inside Story of Our Body's Most Underrated Organ (Revised Edition)Rating: 4 out of 5 stars4/5 (378)
- A Series of Fortunate Events: Chance and the Making of the Planet, Life, and YouFrom EverandA Series of Fortunate Events: Chance and the Making of the Planet, Life, and YouRating: 4.5 out of 5 stars4.5/5 (62)
- Wayfinding: The Science and Mystery of How Humans Navigate the WorldFrom EverandWayfinding: The Science and Mystery of How Humans Navigate the WorldRating: 4.5 out of 5 stars4.5/5 (18)
- Who's in Charge?: Free Will and the Science of the BrainFrom EverandWho's in Charge?: Free Will and the Science of the BrainRating: 4 out of 5 stars4/5 (65)
- Moral Tribes: Emotion, Reason, and the Gap Between Us and ThemFrom EverandMoral Tribes: Emotion, Reason, and the Gap Between Us and ThemRating: 4.5 out of 5 stars4.5/5 (115)
- Fast Asleep: Improve Brain Function, Lose Weight, Boost Your Mood, Reduce Stress, and Become a Better SleeperFrom EverandFast Asleep: Improve Brain Function, Lose Weight, Boost Your Mood, Reduce Stress, and Become a Better SleeperRating: 4.5 out of 5 stars4.5/5 (15)
- The Other Side of Normal: How Biology Is Providing the Clues to Unlock the Secrets of Normal and Abnormal BehaviorFrom EverandThe Other Side of Normal: How Biology Is Providing the Clues to Unlock the Secrets of Normal and Abnormal BehaviorNo ratings yet
- Undeniable: How Biology Confirms Our Intuition That Life Is DesignedFrom EverandUndeniable: How Biology Confirms Our Intuition That Life Is DesignedRating: 4 out of 5 stars4/5 (11)
- Buddha's Brain: The Practical Neuroscience of Happiness, Love & WisdomFrom EverandBuddha's Brain: The Practical Neuroscience of Happiness, Love & WisdomRating: 4 out of 5 stars4/5 (215)
- Crypt: Life, Death and Disease in the Middle Ages and BeyondFrom EverandCrypt: Life, Death and Disease in the Middle Ages and BeyondRating: 4 out of 5 stars4/5 (4)
- Human: The Science Behind What Makes Your Brain UniqueFrom EverandHuman: The Science Behind What Makes Your Brain UniqueRating: 3.5 out of 5 stars3.5/5 (38)
- The Lives of Bees: The Untold Story of the Honey Bee in the WildFrom EverandThe Lives of Bees: The Untold Story of the Honey Bee in the WildRating: 4.5 out of 5 stars4.5/5 (44)
- Good Without God: What a Billion Nonreligious People Do BelieveFrom EverandGood Without God: What a Billion Nonreligious People Do BelieveRating: 4 out of 5 stars4/5 (66)
- The Dog Who Couldn't Stop Loving: How Dogs Have Captured Our Hearts for Thousands of YearsFrom EverandThe Dog Who Couldn't Stop Loving: How Dogs Have Captured Our Hearts for Thousands of YearsNo ratings yet