Professional Documents
Culture Documents
1 - ( (4-Fluorophenyl) Carbamoyl) Cyclopropane-1-Carboxylic Acid
Uploaded by
Gangadhar Maram0 ratings0% found this document useful (0 votes)
8 views1 pageOriginal Title
New Microsoft Word Document
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
8 views1 page1 - ( (4-Fluorophenyl) Carbamoyl) Cyclopropane-1-Carboxylic Acid
Uploaded by
Gangadhar MaramCopyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 1
1-((4-Fluorophenyl)carbamoyl)cyclopropane-1-carboxylic acid
A suspension of cyclopropane-1,1-dicarboxylic acid CA1 (10.0 g, 0.08 mol) in THF
(56 mL) was stirred in an ice water bath and triethylamine (7.79 g, 0.08 mol) was added
slowly to keep the reaction temperature below 10 ℃. SOCl2 (9.16 g, 0.08 mol) was added
slowly to the reaction mixture to keep the reaction temperature below 8 ℃. When the
addition was complete, the mixture was stirred below 5 ℃ for another 1 h. 4-Fluoroaniline
SI1 (7.77 g, 0.07 mol) in THF (20 mL) was added to the reaction solution slowly to keep the
reaction temperature below 8 ℃, and stirred on the ice water bath for another 3 h. The
reaction solution was then diluted with ethyl acetate (100 mL) and water (100 mL), stirred at
room temperature for 30 min. The organic phase was separated, washed with water (80 mL)
and brine (80 mL). The solution was concentrated under vacuum to give the solid, dried at 50
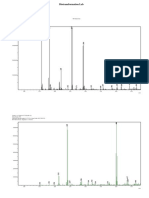
℃ for 4 h to give the product CA2 as a cream solid (9.2 g, 92%). 1 H NMR (DMSO-d6): d
1.41 (s, 4H), 7.14 (t, J ¼ 8.8 Hz, 2H), 7.61 (dd, J ¼ 4.8, 8.8 Hz, 2H), 10.57 (s, 1H). Anal:
Calcd for C11H10FNO3 : C; 59:19; H; 4:52: Found : C; 59:30; H; 4:45
You might also like
- Plant and Animal Bio-Chemistry - Including Information on Amino Acids, Proteins, Pigments and Other Chemical Constituents of Organic MatterFrom EverandPlant and Animal Bio-Chemistry - Including Information on Amino Acids, Proteins, Pigments and Other Chemical Constituents of Organic MatterNo ratings yet
- Shi Epoxidation: Org. Process Res. Dev., 11 (1), 44 - 51, 2007Document9 pagesShi Epoxidation: Org. Process Res. Dev., 11 (1), 44 - 51, 2007kumbham5378No ratings yet
- The Chemistry of Fertilisers and Manure - Including Information on the Chemical Constituents and Types of Fertilisers and ManuresFrom EverandThe Chemistry of Fertilisers and Manure - Including Information on the Chemical Constituents and Types of Fertilisers and ManuresRating: 5 out of 5 stars5/5 (1)
- Piperonal and Safrole From Vanillin and EugenolDocument9 pagesPiperonal and Safrole From Vanillin and EugenolSmokeNo ratings yet
- The Chemistry of Dairy Products - A Chemical Analysis of Milk, Cream and ButterFrom EverandThe Chemistry of Dairy Products - A Chemical Analysis of Milk, Cream and ButterNo ratings yet
- Piperonal and Safrole From Vanillin and EugenolDocument9 pagesPiperonal and Safrole From Vanillin and EugenolMagikFungus100% (4)
- Chemistry at Home - A Collection of Experiments and Formulas for the Chemistry EnthusiastFrom EverandChemistry at Home - A Collection of Experiments and Formulas for the Chemistry EnthusiastNo ratings yet
- New J. Chem., 2011, 35, 1072-1079Document2 pagesNew J. Chem., 2011, 35, 1072-1079Emad AliNo ratings yet
- Advanced Pharmaceutical analysisFrom EverandAdvanced Pharmaceutical analysisRating: 4.5 out of 5 stars4.5/5 (2)
- Oxycodone From CodeineDocument7 pagesOxycodone From CodeineJi ChemNo ratings yet
- Kimia Organologam: Kristianus Raidra Rustina SeptianiDocument30 pagesKimia Organologam: Kristianus Raidra Rustina SeptianiRustina SeptianiNo ratings yet
- Glucono Lactone Dextrose MonohydrateDocument2 pagesGlucono Lactone Dextrose MonohydrateSilvia Melendez HNo ratings yet
- ParacetamolDocument11 pagesParacetamolAjitha AzhakesanNo ratings yet
- Synthesis of Sulphonated CalixiranesDocument6 pagesSynthesis of Sulphonated CalixiranesreddygrNo ratings yet
- Sintesis Nitro FenantrolinaDocument15 pagesSintesis Nitro FenantrolinasgpizarroNo ratings yet
- Media For ColiDocument2 pagesMedia For ColiKathleen PowellNo ratings yet
- Ffee 430000004Document2 pagesFfee 430000004Anonymous FigYuONxuuNo ratings yet
- Procedure MavDocument6 pagesProcedure MavyjgjbhhbNo ratings yet
- The Synthesis, Characterization, and Application of Polysaccharides Based Additives To Increase The Dry Strength of PaperDocument1 pageThe Synthesis, Characterization, and Application of Polysaccharides Based Additives To Increase The Dry Strength of PaperDavina NaidooNo ratings yet
- Synthesis of Adrafinil & ModafinilDocument2 pagesSynthesis of Adrafinil & ModafinildjavahkNo ratings yet
- Aminophylline (Aminophyllinum) : The International Pharmacopoeia - Sixth Edition, 2016Document2 pagesAminophylline (Aminophyllinum) : The International Pharmacopoeia - Sixth Edition, 2016MarkusNo ratings yet
- Reduction of Phenylalanine To AmphetamineDocument2 pagesReduction of Phenylalanine To AmphetamineFlorian FischerNo ratings yet
- FerroceneDocument16 pagesFerroceneshoeb321No ratings yet
- Sertralina SínteseDocument4 pagesSertralina SínteseRaphael AlvarengaNo ratings yet
- Yazılı MetinDocument1 pageYazılı MetinPhoneix reisNo ratings yet
- Synthesis of Acetophenone DerivativesDocument6 pagesSynthesis of Acetophenone DerivativesAwad SaidNo ratings yet
- Naphazoline HCL Synthesis Written ReportDocument15 pagesNaphazoline HCL Synthesis Written ReportFranco BayleNo ratings yet
- STEP 1: Synthesis of N-Phenyl-1,2-Benzenediamine: Experimental ProdedureDocument2 pagesSTEP 1: Synthesis of N-Phenyl-1,2-Benzenediamine: Experimental ProdedureKonstantina MsNo ratings yet
- 5.1.6. Preparation of A Phenol-Formaldehyde Resin and Fabrication of A MouldingDocument2 pages5.1.6. Preparation of A Phenol-Formaldehyde Resin and Fabrication of A MouldingA MahmoodNo ratings yet
- 5328-5337 Volumetric SolutionsDocument10 pages5328-5337 Volumetric Solutionsismail khurshidNo ratings yet
- Preparation and Assay of Acetyl PhosphateDocument4 pagesPreparation and Assay of Acetyl PhosphatecataawwwNo ratings yet
- Cell IsolationDocument3 pagesCell IsolationJitendra SinghNo ratings yet
- Protecting GroupsDocument45 pagesProtecting GroupsSait MalkonduNo ratings yet
- Ferric Citrate: Test SolutionDocument2 pagesFerric Citrate: Test SolutionLalit ModiNo ratings yet
- Seasonal Dynamics in The Nutritional and AntinutritionalDocument9 pagesSeasonal Dynamics in The Nutritional and Antinutritionaludaysing patilNo ratings yet
- Laboratory Course On Organic AnalysisDocument22 pagesLaboratory Course On Organic Analysissoumitrasoni100% (2)
- INDENTIFICATIONDocument6 pagesINDENTIFICATIONMomina HaiderNo ratings yet
- Monografi JECFA Additive-390-M11 SALP BasicDocument2 pagesMonografi JECFA Additive-390-M11 SALP BasicM. Amalul AhliNo ratings yet
- Suggested Methods For Coated Gold NanoparticlesDocument2 pagesSuggested Methods For Coated Gold NanoparticlesJinan JinanNo ratings yet
- Supplementary Material: Azole-Based Inhibitors of P. Falciparum LDHDocument21 pagesSupplementary Material: Azole-Based Inhibitors of P. Falciparum LDHsarabinduroyNo ratings yet
- Ethene Homo - and Ethenepropene Copolymerization 2000Document5 pagesEthene Homo - and Ethenepropene Copolymerization 2000mayamaruguerraNo ratings yet
- Harada 1984Document5 pagesHarada 1984julianpellegrini860No ratings yet
- Disodium Salt Tests For Detecting Fe%Document4 pagesDisodium Salt Tests For Detecting Fe%NIKHIL SHINDENo ratings yet
- Additive 091 m1Document2 pagesAdditive 091 m1Patel HitendraNo ratings yet
- Ap-Magnesium Stearate-1Document7 pagesAp-Magnesium Stearate-1Ade YuLianiNo ratings yet
- SFC Monograph - D344 in Japan's Food StandardsDocument2 pagesSFC Monograph - D344 in Japan's Food StandardsNitinPrachiJainNo ratings yet
- Estimation Protocol Antinutrional Factors in OilseedsDocument33 pagesEstimation Protocol Antinutrional Factors in OilseedsnarayananNo ratings yet
- 2438.2413 DNA Prep BloodDocument3 pages2438.2413 DNA Prep BloodGAURAVNo ratings yet
- Synthesis of Tryptamine From TryptophanDocument5 pagesSynthesis of Tryptamine From TryptophanAnaiza EchelonNo ratings yet
- Phytic Acid: 1.1. Nature, Mechanism of Action, and Biological EffectsDocument5 pagesPhytic Acid: 1.1. Nature, Mechanism of Action, and Biological EffectsBrachim AnsariNo ratings yet
- Lactic AcidDocument2 pagesLactic Acidhediana sandiNo ratings yet
- Calcium CarbonateDocument4 pagesCalcium CarbonateUtibe EdemNo ratings yet
- Preparation of n-Isopropylidene-n'-2-Nitrobenzenesulfonyl Hydrazine (IPNBSH) and Its Use in Palladium-Catalyzed Synthesis of Monoalkyl Diazenes. Synthesis of 9-AllylanthraceneDocument22 pagesPreparation of n-Isopropylidene-n'-2-Nitrobenzenesulfonyl Hydrazine (IPNBSH) and Its Use in Palladium-Catalyzed Synthesis of Monoalkyl Diazenes. Synthesis of 9-AllylanthraceneYami BaudelaireNo ratings yet
- Chem 31.1 Experiment 9 Synthesis of Organic CompoundsDocument68 pagesChem 31.1 Experiment 9 Synthesis of Organic Compoundshello87623100% (1)
- TLC Colour GuideDocument22 pagesTLC Colour GuideMogeshSababathyNo ratings yet
- Synthesis of NitrazepamDocument3 pagesSynthesis of Nitrazepamjiskate77100% (1)
- Synthesis of Novel Fluorobenzothiazole Linked Thiadiazole Compounds: As Possible Anti-Tubercular AgentsDocument2 pagesSynthesis of Novel Fluorobenzothiazole Linked Thiadiazole Compounds: As Possible Anti-Tubercular AgentsVINODNo ratings yet
- Nitrazepam (Synth)Document4 pagesNitrazepam (Synth)clyro27No ratings yet
- Synthesis of A Potential Flame Retardant - HexachlorocyclotriphosphazineDocument2 pagesSynthesis of A Potential Flame Retardant - HexachlorocyclotriphosphazineDaniel RodmanNo ratings yet
- (I) Synthesis of 2,2',4,4',5,5'-Hexaphenyl-4h, 4h'-4,4'-BiimidazoleDocument2 pages(I) Synthesis of 2,2',4,4',5,5'-Hexaphenyl-4h, 4h'-4,4'-BiimidazoleBowie ChongNo ratings yet
- Acs Joc 1c01555Document14 pagesAcs Joc 1c01555Gangadhar MaramNo ratings yet
- Apr N4 213Document2 pagesApr N4 213Gangadhar MaramNo ratings yet
- Acs Joc 2c02398Document10 pagesAcs Joc 2c02398Gangadhar MaramNo ratings yet
- 6 Phenyl 4,5 Dihydro 3 (2H) PyridazinonDocument9 pages6 Phenyl 4,5 Dihydro 3 (2H) PyridazinonGangadhar MaramNo ratings yet
- Available Online Through: Various Approaches For Synthesis of Oxadiazole DerivativesDocument10 pagesAvailable Online Through: Various Approaches For Synthesis of Oxadiazole DerivativesGangadhar MaramNo ratings yet
- ICH Quality Guidelines: An Implementation GuideFrom EverandICH Quality Guidelines: An Implementation GuideAndrew TeasdaleNo ratings yet
- Chemistry for Breakfast: The Amazing Science of Everyday LifeFrom EverandChemistry for Breakfast: The Amazing Science of Everyday LifeRating: 4.5 out of 5 stars4.5/5 (14)
- Periodic Tales: A Cultural History of the Elements, from Arsenic to ZincFrom EverandPeriodic Tales: A Cultural History of the Elements, from Arsenic to ZincRating: 3.5 out of 5 stars3.5/5 (137)
- It's Elemental: The Hidden Chemistry in EverythingFrom EverandIt's Elemental: The Hidden Chemistry in EverythingRating: 4 out of 5 stars4/5 (10)
- The Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsFrom EverandThe Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsRating: 5 out of 5 stars5/5 (3)
- Taste: Surprising Stories and Science About Why Food Tastes GoodFrom EverandTaste: Surprising Stories and Science About Why Food Tastes GoodRating: 3 out of 5 stars3/5 (20)
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactFrom EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactRating: 5 out of 5 stars5/5 (5)
- AP® Chemistry Crash Course, For the 2020 Exam, Book + Online: Get a Higher Score in Less TimeFrom EverandAP® Chemistry Crash Course, For the 2020 Exam, Book + Online: Get a Higher Score in Less TimeRating: 5 out of 5 stars5/5 (1)
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactFrom EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactRating: 5 out of 5 stars5/5 (1)
- Monkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeFrom EverandMonkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeRating: 4 out of 5 stars4/5 (1)
- The Production of Volatile Oils and Perfumery Plants in the United StatesFrom EverandThe Production of Volatile Oils and Perfumery Plants in the United StatesNo ratings yet
- Guidelines for Defining Process Safety Competency RequirementsFrom EverandGuidelines for Defining Process Safety Competency RequirementsRating: 3 out of 5 stars3/5 (1)
- AP Chemistry Flashcards, Fourth Edition: Up-to-Date Review and PracticeFrom EverandAP Chemistry Flashcards, Fourth Edition: Up-to-Date Review and PracticeNo ratings yet
- Handbook of Formulating Dermal Applications: A Definitive Practical GuideFrom EverandHandbook of Formulating Dermal Applications: A Definitive Practical GuideNo ratings yet
- Chemistry for Breakfast: The Amazing Science of Everyday LifeFrom EverandChemistry for Breakfast: The Amazing Science of Everyday LifeRating: 4.5 out of 5 stars4.5/5 (90)
- Essential Chemistry for Formulators of Semisolid and Liquid DosagesFrom EverandEssential Chemistry for Formulators of Semisolid and Liquid DosagesRating: 5 out of 5 stars5/5 (2)
- Is That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeFrom EverandIs That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeRating: 5 out of 5 stars5/5 (4)