Professional Documents
Culture Documents
990 PDF
Uploaded by
Neha IkhlaqOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
990 PDF
Uploaded by
Neha IkhlaqCopyright:
Available Formats
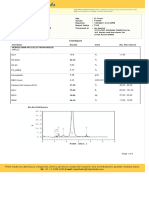
Slip # : 230300990
Pat.Name : BAKHTO MAI Slip Date : 13/03/2023 10:42PM
Age/Gender : 55 Year(s) / Female Reported : 15/03/2023 12:30AM
CNIC No. : Ph.No. : 0 Registered : British Lab Nishtar Road
Specimen : Blood Performed : British Lab
Referred By : CITY LAB ANDDAIGNOSTIC CENTER
PCR HCV RNA (QUANTITATIVE)
Results: This sample is DETECTED for HCV RNA
Concentration: of HCV RNA is 7761 IU/mL
SENSITIVITY:
The test has been shown to be potent to detect at least 30 IU/ml using a HCV positive serum.
Principle:
HCV-diagnosis-by-PCR-is-based-on-the-amplification-of-specific-regions-of-pathogen-genome.-In-Real-Time-PCR-(RT-
PCR)-the-amplified-product-is-detected-via-the-fluorescent-dyes,-which-specifically-binds-to-the-5`-oligonucleotides.
An-Internal-Control-(IC)-is-run-with-each-sample.-The-HCV-and-IC-specific-probes-are-each-labelled-with-different-fluor
ophore,-thus-allowing-for-simultaneous-detection-of-both-amplified-product-at-each-cycle.-The-non-competitive-Internal-
Control-(IC)-is-detected-at-all-HCV-levels.-In-this-way-inhibition-in-the-sample-could-be-observed.-The-homogenous-for
mat-and-sealed-PCR-tubes-eliminate-chances-of-contamination-by-amplified-products.
To-obtain-results-in-copies/ml,-multiply-IU/ml-value-with-1.21.
Formula:-Results-in-IU/ml-`x-1.21-=-copies/ml.
RELIABILITY:
Due to the co-purification and amplification of synthetic internal control RNA maximum reliability of the HCV
quantification kit is granted the absence of PCR inhibitor as well as the correct performance of the extraction can be
detected. Using ready to use virus comp controls a confident quantification of HCV in human serum or plasma samples
is accomplished.
NOTE:
The titer of circulation HCV in blood fluctuates in accordance with the virus latency & variable results may occur in
different phase of viral infection. Therefore clinical correlation is very important Lab to Lab variation may occur due to
difference in sensitivity, specificity and reproducibility of different assay technique used .
Report Electronically Verified By : Unaiza Asim
Electronically verified report, not require any signature.
DR. SHAHID MEHMOOD DR.FAIZA SHAFQAT SADAF SHAHEEN
MBBS (Pb) MBBS, MPhil (Hemat) (M.Phil) Microbiology
Managing Director Consultant Pathologist PHD Microbiology
You might also like
- Infectious Diseases: Smart Study Guide for Medical Students, Residents, and Clinical ProvidersFrom EverandInfectious Diseases: Smart Study Guide for Medical Students, Residents, and Clinical ProvidersNo ratings yet
- InvalidDocument2 pagesInvalidFarrukh JamilNo ratings yet
- RTPCR TestDocument2 pagesRTPCR TestElite EnterprisesNo ratings yet
- Zahida BibiDocument1 pageZahida BibiHabib Ur Rehman BazmiNo ratings yet
- Department of Microbiology: No Motility (No Trichomonas Vaginalis) SeenDocument1 pageDepartment of Microbiology: No Motility (No Trichomonas Vaginalis) SeenMalik Zia AwanNo ratings yet
- Investigation Report: Type of Sample: Nasopharyngeal Swab Test ResultDocument1 pageInvestigation Report: Type of Sample: Nasopharyngeal Swab Test ResultNazim Uddin MahmudNo ratings yet
- C11423 - U'22-Nov-23 - BHUPENDRA B. KANUNGO - U18183 - 24F0F - U - P6565P - 17971 - 8Document4 pagesC11423 - U'22-Nov-23 - BHUPENDRA B. KANUNGO - U18183 - 24F0F - U - P6565P - 17971 - 8khanak bodraNo ratings yet
- Clinical BiochemistryDocument3 pagesClinical BiochemistrySatyam RajNo ratings yet
- CCCCCDocument3 pagesCCCCCmrkazi361No ratings yet
- ReportDocument4 pagesReportruthwik.gmcNo ratings yet
- Azeema Covid ReportDocument2 pagesAzeema Covid ReportAccounts West India GlobalNo ratings yet
- LabReport L14 Mrs - RAJESHWARI60YRS F 06 04 202420240406 2 E38fgDocument3 pagesLabReport L14 Mrs - RAJESHWARI60YRS F 06 04 202420240406 2 E38fgAnkit GairolaNo ratings yet
- Department of Molecular Virology: COVID-19 (Corona) VirusDocument1 pageDepartment of Molecular Virology: COVID-19 (Corona) VirusMuhammad YahyaNo ratings yet
- Jinnah Central Lab: Jinnah Hospital & AIMC Lahore, PakistanDocument2 pagesJinnah Central Lab: Jinnah Hospital & AIMC Lahore, Pakistanafshan liaqatNo ratings yet
- Reportviewinpdf - 2022-07-26T191041.034Document1 pageReportviewinpdf - 2022-07-26T191041.034Usman niazNo ratings yet
- 2020VI75393RDocument2 pages2020VI75393RAhmed Abbas ZaidiNo ratings yet
- Sabera BegumDocument2 pagesSabera BegumRiya ShahNo ratings yet
- Aditi 2Document5 pagesAditi 2princeverma77530No ratings yet
- Wwiy4300 PDFDocument6 pagesWwiy4300 PDFShobhit MishraNo ratings yet
- 2023 10 05 Mrs. SHAILA RASUREDocument2 pages2023 10 05 Mrs. SHAILA RASUREshaileshrasure03No ratings yet
- Specimen Nasopharyngeal Swab & Throat Swab Sars-Cov-2 Rna:: 13/01/2022 08:16 PM 14/01/2022 08:08 AM: 5047VA078778Document2 pagesSpecimen Nasopharyngeal Swab & Throat Swab Sars-Cov-2 Rna:: 13/01/2022 08:16 PM 14/01/2022 08:08 AM: 5047VA078778Harshal Shashikant NagleNo ratings yet
- Chughtai Lab ReportDocument3 pagesChughtai Lab Reportjasimhashmi000No ratings yet
- 1049 PDFDocument3 pages1049 PDFPavan ChaitanyaNo ratings yet
- Rehman Medical Institute: MethodDocument1 pageRehman Medical Institute: MethodWaqas HaleemNo ratings yet
- Covid Test Result Deepti PadteDocument2 pagesCovid Test Result Deepti PadteDeepti PadteNo ratings yet
- R10906124 Rathna U Kuri 270523183758Document8 pagesR10906124 Rathna U Kuri 270523183758suhas umeshNo ratings yet
- Anjali Khapare - 23071171Document3 pagesAnjali Khapare - 23071171dr.menganeNo ratings yet
- Report 4Document2 pagesReport 4Tahira HashmiNo ratings yet
- Test Report: PAVITHRA (25/F)Document3 pagesTest Report: PAVITHRA (25/F)Pavi PavichuNo ratings yet
- ABHIJEETDocument3 pagesABHIJEETKrishNo ratings yet
- Wwiy4300 PDFDocument3 pagesWwiy4300 PDFRani reddy44% (43)
- Shehnaaj BegumDocument2 pagesShehnaaj BegumRiya ShahNo ratings yet
- Department of Molecular Pathology: Corona Virus (Covid-19) Rna by PCRDocument1 pageDepartment of Molecular Pathology: Corona Virus (Covid-19) Rna by PCRMuhammad AbdullahNo ratings yet
- Fully Automated Lab Accurate Reports Reports Verified by PathologistDocument11 pagesFully Automated Lab Accurate Reports Reports Verified by PathologistRavindranatha AnNo ratings yet
- 2020MB6953RDocument1 page2020MB6953RFaizan RasoolNo ratings yet
- 109db8e0 3eaDocument3 pages109db8e0 3eaJabeen NigarNo ratings yet
- Test Report: Mrs - UMA (46/F)Document3 pagesTest Report: Mrs - UMA (46/F)KanjamNo ratings yet
- Siddhartha PDFDocument9 pagesSiddhartha PDFArnav kumarNo ratings yet
- BSFH 12Document9 pagesBSFH 12jinop79764No ratings yet
- Specimen Nasopharyngeal Swab & Throat Swab Sars-Cov-2 Rna Detected CT Value of Confirmatory Gene: Target (S)Document2 pagesSpecimen Nasopharyngeal Swab & Throat Swab Sars-Cov-2 Rna Detected CT Value of Confirmatory Gene: Target (S)Apache RTRNo ratings yet
- Max Lab ReportDocument8 pagesMax Lab ReportKallu PrasadNo ratings yet
- Specimen Nasopharyngeal Swab & Throat Swab Sars-Cov-2 Rna CT Value of Confirmatory Gene: Target (S)Document2 pagesSpecimen Nasopharyngeal Swab & Throat Swab Sars-Cov-2 Rna CT Value of Confirmatory Gene: Target (S)Ankit SuraNo ratings yet
- TyphoidDocument3 pagesTyphoidShivam Tomar83% (6)
- Lab Report NewDocument5 pagesLab Report Newsamirghosal19No ratings yet
- Department of Pathology: Molecular BiologyDocument1 pageDepartment of Pathology: Molecular BiologyHussain AliNo ratings yet
- DR. Neelanjana SHARMADocument5 pagesDR. Neelanjana SHARMADr. Neelanjana SharmaNo ratings yet
- 001 230277754 CC2 117 1Document1 page001 230277754 CC2 117 1irshad72No ratings yet
- Specimen Nasopharyngeal Swab & Throat Swab Sars-Cov-2 Rna Target (S)Document2 pagesSpecimen Nasopharyngeal Swab & Throat Swab Sars-Cov-2 Rna Target (S)manwanimuki12No ratings yet
- 23-Jan-2020 5:15 PM 36 Year(s) /male Brought From Outside 1196 26-Jan-2020 10:12 PM Mashallah Homephty Hospital Mandi YazmanDocument1 page23-Jan-2020 5:15 PM 36 Year(s) /male Brought From Outside 1196 26-Jan-2020 10:12 PM Mashallah Homephty Hospital Mandi YazmanFast ComputersNo ratings yet
- SHAFNA Female34 Years 207392Document2 pagesSHAFNA Female34 Years 207392Muhammed DilshanNo ratings yet
- Report of Mr. RAJA PDFDocument3 pagesReport of Mr. RAJA PDFraja.tyagi2125No ratings yet
- Rit ReportDocument4 pagesRit Reportadarshranjansingh1903No ratings yet
- Quality ReportDocument2 pagesQuality ReportUSMAN ....No ratings yet
- Department of Molecular Virology: COVID-19 (Corona) VirusDocument1 pageDepartment of Molecular Virology: COVID-19 (Corona) VirusShahzad Ali100% (1)
- Mangal Cook ReportDocument2 pagesMangal Cook ReportSanjeev SharmaNo ratings yet
- BSFH 13Document9 pagesBSFH 13jinop79764No ratings yet
- Pedia LabDocument6 pagesPedia LabMary Grace VillegasNo ratings yet
- SAGARIKA SAHOO-Female30 Years-6143Document3 pagesSAGARIKA SAHOO-Female30 Years-6143J. K. MuduliNo ratings yet
- Xdue6378 2Document2 pagesXdue6378 2S Abedi50% (2)
- Report ViewerDocument1 pageReport ViewerHammad ur Rehman100% (1)