Professional Documents
Culture Documents
Amines 2ndPUC PYQs
Uploaded by
Shreedhar KappadiOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Amines 2ndPUC PYQs
Uploaded by
Shreedhar KappadiCopyright:
Available Formats
Amines
(1+5=6)
For: PU Board 2023
MCQs
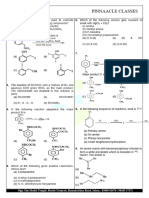
1. Amongst the following the strongest base in aqueous medium is
(a) CH3NH2 (b) (CH)3N (c) (CH3)2NH (d) C6H5NH2 (2023 m)
(5 MARKS)
1. a) i) Explain Hoffmann bromamide degradation for the preparation of Aniline (2014)
ii) Give the IUPAC name of
H3C-N-CH2-CH3
H
b) What is Hinsberg reagent? Between CH3NH2 and C6H5NH2 which is more basic?
2. a) identify the reagent ‘A’ in the following reaction: (2015)
A + 2R – X → R4N+X-
b) Explain Hoffmann’s bromamide degradation reaction for the preparation of methenamine.
c) Which is more basic among aqueous solutions of aniline and ammonia? Give one reason.
3. a) Explain carbylamine reaction
b) What is the action of bromine water on benzenamine (Aniline) at room temp?
c) The pkb value of ammonia methanamine and bezenamine (aniline) are 4.75, 3.38, and
9.38 respectively arrange them in the increasing order of their basic strength. (2016)
4. a) i) Explain the reduction of nitrocompounds to amines with an example.
ii) Why aromatic primary amines cannot be prepared by Gabriel synthesis?
b) How is aniline converted into phenyl isocyanide? Write the equation. (2016s)
5. a) Name the major product formed when nitrous acid is treated with
i) methylamine
ii) aniline at low temperature
b) Explain the Hoffmann’s bromide reaction
c) Write the IUPAC name of (CH3)2N- CH2-CH3 (2015s)
6. a) How primary amine is prepared by Hoffmann bromamide degradation reaction? Write
equation.
b) i) write IUPAC name of CH3CH2NH2.
ii) Arrange the following amines in the order of their increasing basic strength in aqueous
solution. (CH3)3N,(CH3)2NH,CH3NH2
iii) complete the following equation.
273−278 𝐾
C6H5NH2 + NaNO2 + 2HCl → (2017)
7. a) Explain Hoffmann broamide degradation reaction.
b) Name the major organic product formed in the following conversion
i. When nitrous acid is treated with methyl lamine.
ii. Benzene diazonium chloride is treated with KI.
c) Which is more basic among methyl amine and aniline? (2017s)
8. a) Explain carbyl amine reaction with equation.
b) How does nitrobenzene is reduced to aniline? Give equations.
c) Write the IUPAC name of
C6H5 – N – CH3
CH3 (2018)
Download our App “SimplifiedMinds” @SimplifiedMinds
www.simplifiedminds.com
Call Us: 7411-008-008
Whatsapp Us: 8867-008-008
Amines
(1+5=6)
For: PU Board 2023
9. a) How is methanamine prepared by Hoffmann bromamide degradation reaction? Give
equation.
b) How is aniline converted to Benzene diazonium chloride? Give equation.
c) Between ammonia and aniline, which is more basic? (2018s)
10. a) Between CH3NH2 and C6H5NH2 which is more base? Give reason.
b) i) Name the main product when aniline is heated with alcoholic KOH and chloroform.
ii) Give the IUPAC name of (CH3)2N-C2H5.
c) Complete the chemical equation.
𝐵𝑟2 /𝑁𝑎𝑂𝐻
CH3CONH2 → (2019)
11. a) How does Primary amine undergo carbyl amine reaction? Give chemical equations.
b) How aniline is prepared by Hoffmann bromamide degradation reaction? Give equation.
c) Write the general formula of diazonium salt (2019s)
12. a) How does Primary amine undergo carbyl amine reaction? Give chemical equations.
b) How aniline is prepared by Hoffmann bromamide degradation reaction? Give equation.
c) Write the general formula of diazonium salt
13. a) Mention the I.U.P.A.C name of (CH3)2 – N – CH3.
b) How is Aniline is prepared from nitro benzene?
c) Give the equation for the conversion of aniline to 4-Bromo aniline (2020)
14. a) Write the equation and IUPAC name of the product formed when aniline reacts with
bromine water at room temperature. (2021m)
b) Write the increasing order of basicity of the following amines in aqueous solution.
NH3, (C2H5)3N, C2H5NH2, (C2H5)2NH
c) How do you prepare primary amine by Gabriel’s phthalimide synthesis? Give equation.
15. a) How do you convert methyl cyanide into ethyl amine by reduction by catalytic
hydrogenation method?
b) When aniline is treated with HNO2 at 273K-278K, benzene diazonium chloride is formed.
c) Write the equation and name the reaction.
d) Give reason: Methyl amine is more basic than ammonia. (2021m)
16. a) Explain carbylamine reaction with suitable example
b) Write chemical equation for the conversion of Nitro-benzene to aniline
c) Why primary amines have higher boiling point than tertiary amines? (2023 m)
What you get on our app?
1. Detailed explanation of every topic and all PYQs
2. Lecture pdfs and Hand written notes for important formulas and derivations for PU Board.
3. In app chat feature for all your doubts directly with the faculty
Download our App “SimplifiedMinds” @SimplifiedMinds
www.simplifiedminds.com
Call Us: 7411-008-008
Whatsapp Us: 8867-008-008
You might also like
- Class 12 Chemistry PYQs Shobhit NirwanDocument68 pagesClass 12 Chemistry PYQs Shobhit NirwanDron Pande83% (12)
- CPP AminesDocument13 pagesCPP AminesDivyansh purohitNo ratings yet
- Erich FrommDocument2 pagesErich FrommTina NavarroNo ratings yet
- Coles Recipe MagazineDocument68 pagesColes Recipe MagazinePhzishuang TanNo ratings yet
- Quinta RuedaDocument20 pagesQuinta RuedaArturo RengifoNo ratings yet
- Studovaný Okruh: Physical Therapist Sample Test Questions (G5+)Document8 pagesStudovaný Okruh: Physical Therapist Sample Test Questions (G5+)AndreeaNo ratings yet
- Ainsworth, The One-Year-Old Task of The Strange SituationDocument20 pagesAinsworth, The One-Year-Old Task of The Strange SituationliliaNo ratings yet
- Amines 2ndPUC PYQs - 43f50ccd 882e 41e8 A2ff 15f151fec8c5Document3 pagesAmines 2ndPUC PYQs - 43f50ccd 882e 41e8 A2ff 15f151fec8c5nandinandisht4No ratings yet
- Amines 2ndPUC PYQsDocument2 pagesAmines 2ndPUC PYQsDIKSHITH GOWDANo ratings yet
- Chemistry Test Series Chapter Name: Amines Class-Xii MM: 35 Time: 2 HoursDocument2 pagesChemistry Test Series Chapter Name: Amines Class-Xii MM: 35 Time: 2 HoursOm MishraNo ratings yet
- Amine - QTN - Bank (1) JonishDocument6 pagesAmine - QTN - Bank (1) Jonishjeba kingNo ratings yet
- Worksheet On AminesDocument3 pagesWorksheet On Aminesloginrandom9No ratings yet
- AminesDocument3 pagesAminesgarvitNo ratings yet
- CHAPTER - 12 (Amines)Document5 pagesCHAPTER - 12 (Amines)Forzen flamesNo ratings yet
- UNIT 13 AMINES QN Bank PDFDocument6 pagesUNIT 13 AMINES QN Bank PDFDharaneesh S.k.No ratings yet
- Amines PreparatoryDocument4 pagesAmines Preparatoryevelynziggyada77No ratings yet
- All District Preparatory Questions FinalDocument39 pagesAll District Preparatory Questions Finalvarshithajadhav6No ratings yet
- Aminines Markswise QuestionsDocument5 pagesAminines Markswise QuestionsSachin GuptaNo ratings yet
- Unit 13 AminesDocument3 pagesUnit 13 AminesArinath DeepaNo ratings yet
- Alcohols, Phenols and Ethers 2nd PUC PYQsDocument3 pagesAlcohols, Phenols and Ethers 2nd PUC PYQsdagar2790No ratings yet
- Amines PyqDocument10 pagesAmines PyqEr.Punit MohanNo ratings yet
- Xii - Ch-Amines-QsDocument6 pagesXii - Ch-Amines-Qskaushiksarathi89No ratings yet
- 12 Chemistry Impq CH13 Amines 01Document5 pages12 Chemistry Impq CH13 Amines 01nikhilporwal84No ratings yet
- AminesDocument6 pagesAminesatharvbaghel4444No ratings yet
- Hsslive-Xii-Chemistry-Qb-Ans-Anil-13. AminesDocument9 pagesHsslive-Xii-Chemistry-Qb-Ans-Anil-13. AminesSneha Rose BennyNo ratings yet
- Haloalkanes and Haloarenes 2ndPUC PYQsDocument3 pagesHaloalkanes and Haloarenes 2ndPUC PYQssparkysanthosh69No ratings yet
- Unit 11 Alcohols & Ethers - 230206 - 114927-1 - 230703 - 162856Document7 pagesUnit 11 Alcohols & Ethers - 230206 - 114927-1 - 230703 - 162856jigneshkavybinaNo ratings yet
- C13 - ORGANIC COMPOUND CONTAINING NITROGEN (1) .9d8e384Document4 pagesC13 - ORGANIC COMPOUND CONTAINING NITROGEN (1) .9d8e384akashkishore363No ratings yet
- Seth Anandram Jaipuria School Revision Test (2021-22), Class-Xii (Topic - Amines)Document2 pagesSeth Anandram Jaipuria School Revision Test (2021-22), Class-Xii (Topic - Amines)vedang agarwalNo ratings yet
- Amines Final On Print WITH StampDocument8 pagesAmines Final On Print WITH StampAhmedAdelIbrahimNo ratings yet
- CH 13Document6 pagesCH 13Himanshu SangwanNo ratings yet
- 6441 Topper 21 129 510 2 8550 Amines and Aromatic Compounds Containing Nitrogen Up201612091904 1481290486 9301 PDFDocument45 pages6441 Topper 21 129 510 2 8550 Amines and Aromatic Compounds Containing Nitrogen Up201612091904 1481290486 9301 PDFSakethNo ratings yet
- AMINES Quiz FormatDocument8 pagesAMINES Quiz FormatSankar KumarasamyNo ratings yet
- AminesDocument2 pagesAminesVed patelNo ratings yet
- Worksheet:1 AminesDocument2 pagesWorksheet:1 AminesRaj PandeyNo ratings yet
- Unit 09Document9 pagesUnit 09ZackNo ratings yet
- 13 AminesDocument2 pages13 AminesChemGuru 4 UNo ratings yet
- Question PaprDocument3 pagesQuestion PapruvsNo ratings yet
- CH7 Alcoholsphenols and Ethers HHW WorksheetDocument4 pagesCH7 Alcoholsphenols and Ethers HHW WorksheetAaditya SharmaNo ratings yet
- AMINES - Obj.Document1 pageAMINES - Obj.Aparna ArunkumarNo ratings yet
- Amines Revision 2022Document3 pagesAmines Revision 2022Dêêpák Sîñgh ÑîtwálNo ratings yet
- Unit 11 Alcohols & EthersDocument6 pagesUnit 11 Alcohols & EthersDeepesh kumarNo ratings yet
- ALCOHOL ChapterDocument6 pagesALCOHOL ChapterSharon Shymala LewisNo ratings yet
- Chemistry AminesDocument8 pagesChemistry AminesVanshika LudhaniNo ratings yet
- PYQ of AminesDocument11 pagesPYQ of AminesankitaNo ratings yet
- Unit 11 Alcohols & EthersDocument6 pagesUnit 11 Alcohols & EthersLet's play Guitar with AshmitaNo ratings yet
- Class Xii Chemistry Assignment Unit: AminesDocument4 pagesClass Xii Chemistry Assignment Unit: AminesMeet P TrivediNo ratings yet
- Question Based On Name ReactionDocument4 pagesQuestion Based On Name ReactionSelcouth elysianNo ratings yet
- DPT 33 Centre Rasi Iit Jee Che Key 09-12-23Document4 pagesDPT 33 Centre Rasi Iit Jee Che Key 09-12-23Deena chemistNo ratings yet
- CBSE Class 12 Chemistry Chapter 13 - Amines Important Questions 2023-24Document18 pagesCBSE Class 12 Chemistry Chapter 13 - Amines Important Questions 2023-24Afzal MohamedNo ratings yet
- 12th Class Chapter Amines Wise QP 2022-23Document6 pages12th Class Chapter Amines Wise QP 2022-23Gunjan BisenNo ratings yet
- Hsslive-Xii-Chem-9. AminesDocument4 pagesHsslive-Xii-Chem-9. Aminesnr249488No ratings yet
- Amines Question PaperDocument1 pageAmines Question PaperPrajwal DixitNo ratings yet
- Delhi Public School: Nacharam/ Mahendra Hills/ NadergulDocument3 pagesDelhi Public School: Nacharam/ Mahendra Hills/ Naderguleeshwar saagarNo ratings yet
- Amines Important Questions.Document16 pagesAmines Important Questions.Rockz RockzzNo ratings yet
- 13 AminesDocument2 pages13 Aminesmystical moonbeamNo ratings yet
- Question Bank Unit 12 Aldehydes, Ketones & Carboxylic Acids CLASS-12 (CBSE)Document8 pagesQuestion Bank Unit 12 Aldehydes, Ketones & Carboxylic Acids CLASS-12 (CBSE)Dharshitha100% (1)
- 12 Pre Board 1 1222Document5 pages12 Pre Board 1 1222geyeyo9277No ratings yet
- Bhu Pet 2020Document14 pagesBhu Pet 2020iftikar hussainNo ratings yet
- YEAR 2022: (I) (Ii) (Iii)Document3 pagesYEAR 2022: (I) (Ii) (Iii)Anurag DubeyNo ratings yet
- AminesDocument4 pagesAminesveerlocusNo ratings yet
- C Module 8ADocument43 pagesC Module 8ASundareshwar SNo ratings yet
- Differentiation of Chiral Compounds Using NMR SpectroscopyFrom EverandDifferentiation of Chiral Compounds Using NMR SpectroscopyNo ratings yet
- Carbon Nanomaterials for Advanced Energy Systems: Advances in Materials Synthesis and Device ApplicationsFrom EverandCarbon Nanomaterials for Advanced Energy Systems: Advances in Materials Synthesis and Device ApplicationsWen LuNo ratings yet
- Sonesse 30 Databook NewestDocument32 pagesSonesse 30 Databook Newestsunii19847908No ratings yet
- S:/admin/mpi/MP1169 - Amaia Skies Samat/000 - ACTIVE DOCUMENTS/09 - SPECS/2013-07-23 - Design Development/04-Plumbing/15050Document19 pagesS:/admin/mpi/MP1169 - Amaia Skies Samat/000 - ACTIVE DOCUMENTS/09 - SPECS/2013-07-23 - Design Development/04-Plumbing/15050Lui TCC BariaNo ratings yet
- CL Analyzer: Coagulometric, Chromogenic and Immunological AssaysDocument2 pagesCL Analyzer: Coagulometric, Chromogenic and Immunological AssaysEdwinNo ratings yet
- Sesion 2 - Copia-1Document14 pagesSesion 2 - Copia-1Maeva FigueroaNo ratings yet
- Aseptic TechniquesDocument3 pagesAseptic TechniquesMacy MarianNo ratings yet
- API 650 10th Edition ErrataDocument6 pagesAPI 650 10th Edition ErrataJosé Ramón GutierrezNo ratings yet
- API 614-Chapter 4 DATA SHEET Dry Gas Seal Module Si Units: System Responsibility: (2.1.2)Document10 pagesAPI 614-Chapter 4 DATA SHEET Dry Gas Seal Module Si Units: System Responsibility: (2.1.2)tutuionutNo ratings yet
- Compensation ManagementDocument2 pagesCompensation Managementshreekumar_scdlNo ratings yet
- Presentation of DR Rai On Sahasrara Day Medical SessionDocument31 pagesPresentation of DR Rai On Sahasrara Day Medical SessionRahul TikkuNo ratings yet
- Sargent Catalog CutsDocument60 pagesSargent Catalog CutssmroboNo ratings yet
- Weekly Meal Prep GuideDocument7 pagesWeekly Meal Prep Guideandrew.johnson3112No ratings yet
- Chapter 11-15Document172 pagesChapter 11-15Mansoor AhmadNo ratings yet
- Stereochemistry Chiral Molecules QuizDocument3 pagesStereochemistry Chiral Molecules QuizSean McDivittNo ratings yet
- Assignment 2 - CE 669A - 22-23Document1 pageAssignment 2 - CE 669A - 22-23Sonam MeenaNo ratings yet
- Pengaruh Penambahan Lateks Pada Campuran Asphalt Concrete Binder Course (AC-BC)Document10 pagesPengaruh Penambahan Lateks Pada Campuran Asphalt Concrete Binder Course (AC-BC)Haris FirdausNo ratings yet
- High CarbonDocument2 pagesHigh CarbonKarisoNo ratings yet
- The Power of PositivityDocument5 pagesThe Power of PositivityYorlenis PintoNo ratings yet
- Brain Slides SEMINAR 1 - 220606 - 142811 - 220606 - 223805Document32 pagesBrain Slides SEMINAR 1 - 220606 - 142811 - 220606 - 223805pang pangNo ratings yet
- UK FreshTECH Jammer RecipeBook 0Document24 pagesUK FreshTECH Jammer RecipeBook 0Temet NoscheNo ratings yet
- Effect of Moisture Content On The Extraction Rate of Coffee Oil From Spent Coffee Grounds Using Norflurane As SolventDocument8 pagesEffect of Moisture Content On The Extraction Rate of Coffee Oil From Spent Coffee Grounds Using Norflurane As SolventMega MustikaningrumNo ratings yet
- Macroscopic Physics Chemistry HW #1Document11 pagesMacroscopic Physics Chemistry HW #1Akash ModyNo ratings yet
- ODocument11 pagesOMihaela CherejiNo ratings yet
- TDS Shell Spirax s6 Gxme 75w-80Document2 pagesTDS Shell Spirax s6 Gxme 75w-80rstec pyNo ratings yet
- Laws and Regulation Related To FoodDocument33 pagesLaws and Regulation Related To FoodDr. Satish JangraNo ratings yet