1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
Science & Tech Biographies Animals & Nature Geography & Travel Arts & Culture Money Videos
Home keyboard_arrow_right Technology keyboard_arrow_right Industry
Science & Tech
stainless steel more_vert Actions
metallurgy
Written and fact-checked by The Editors of Encyclopaedia Britannica
Last Updated: Nov 30, 2023 • Article History
toc Table of Contents
zoom_in
stainless-steel equipment
See all media
Category: Science & Tech
Key People: Georg Jensen • Patrick Joseph Frawley, Jr. • Elwood Haynes
https://www.britannica.com/science/inorganic-compound 1/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
Related Topics: martensitic steel • duplex stainless steel • precipitation-hardening stainless stain •
austenitic steel • ferritic steel ...(Show more)
See all related content →
Top Questions
What are the types of stainless steel? expand_more
Why is chromium used in stainless steel? expand_more
Which type of stainless steel usually has the highest corrosion resistance? expand_more
Stainless steel, any one of a family of alloy steels usually containing 10 to 30 percent
chromium. In conjunction with low carbon content, chromium imparts remarkable
resistance to corrosion and heat. Other elements, such as nickel, molybdenum,
titanium, aluminum, niobium, copper, nitrogen, sulfur, phosphorus, or selenium, may
be added to increase corrosion resistance to specific environments, enhance oxidation
resistance, and impart special characteristics.
Most stainless steels are first melted in electric-arc or basic oxygen furnaces and
subsequently refined in another steelmaking vessel, mainly to lower the carbon content.
In the argon-oxygen decarburization process, a mixture of oxygen and argon gas is
injected into the liquid steel. By varying the ratio of oxygen and argon, it is possible to
remove carbon to controlled levels by oxidizing it to carbon monoxide without also
oxidizing and losing expensive chromium. Thus, cheaper raw materials, such as high-
carbon ferrochromium, may be used in the initial melting operation.
Britannica Quiz
Know Your Chemistry Quiz
https://www.britannica.com/science/inorganic-compound 2/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
zoom_in
stainless steel
The nickel and chromium content of different types of stainless steel.
There are more than 100 grades of stainless steel. The majority are classified into five
major groups in the family of stainless steels: austenitic, ferritic, martensitic, duplex,
and precipitation-hardening. Austenitic steels, which contain 16 to 26 percent
chromium and up to 35 percent nickel, usually have the highest corrosion resistance.
They are not hardenable by heat treatment and are nonmagnetic. The most common
type is the 18/8, or 304, grade, which contains 18 percent chromium and 8 percent
nickel. Typical applications include aircraft and the dairy and food-processing
industries. Standard ferritic steels contain 10.5 to 27 percent chromium and are nickel-
free; because of their low carbon content (less than 0.2 percent), they are not
hardenable by heat treatment and have less critical anticorrosion applications, such as
architectural and auto trim. Martensitic steels typically contain 11.5 to 18 percent
chromium and up to 1.2 percent carbon with nickel sometimes added. They are
hardenable by heat treatment, have modest corrosion resistance, and are employed in
cutlery, surgical instruments, wrenches, and turbines. Duplex stainless steels are a
https://www.britannica.com/science/inorganic-compound 3/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
combination of austenitic and ferritic stainless steels in equal amounts; they contain 21
to 27 percent chromium, 1.35 to 8 percent nickel, 0.05 to 3 percent copper, and 0.05 to
5 percent molybdenum. Duplex stainless steels are stronger and more resistant to
corrosion than austenitic and ferritic stainless steels, which makes them useful in
storage-tank construction, chemical processing, and containers for transporting
chemicals. Precipitation-hardening stainless steel is characterized by its strength, which
stems from the addition of aluminum, copper, and niobium to the alloy in amounts less
than 0.5 percent of the alloy’s total mass. It is comparable to austenitic stainless steel
with respect to its corrosion resistance, and it contains 15 to 17.5 percent chromium, 3
to 5 percent nickel, and 3 to 5 percent copper. Precipitation-hardening stainless steel is
used in the construction of long shafts.
Home keyboard_arrow_right Science keyboard_arrow_right Chemistry
This article was most recently revised and updated by Adam Augustyn.
Science & Tech
inorganic compound more_vert Actions
chemical compound
Written by Steven S. Zumdahl
Fact-checked by The Editors of Encyclopaedia Britannica
Last Updated: Jan 4, 2024 • Article History
toc Table of Contents
https://www.britannica.com/science/inorganic-compound 4/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
zoom_in
crystal structure of nickel arsenide
See all media
Category: Science & Tech
Key People: Carl Wilhelm Scheele • Joseph Loschmidt • Viktor Meyer • Jean-Charles Galissard de Marignac
Related Topics: organometallic compound • oxyacid • salt • silicone • hydrogen cyanide
On the Web: Demo Pressbooks Network - Boundless Anatomy and Physiology - Inorganic Compounds
(Jan. 04, 2024) ...(Show more)
See all related content →
Inorganic compound, any substance in which two or more chemical elements
(usually other than carbon) are combined, nearly always in definite proportions.
Compounds of carbon are classified as organic when carbon is bound to hydrogen.
Carbon compounds such as carbides (e.g., silicon carbide [SiC2]), some carbonates (e.g.,
https://www.britannica.com/science/inorganic-compound 5/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
calcium carbonate [CaCO3]), some cyanides (e.g., sodium cyanide [NaCN]), graphite,
carbon dioxide, and carbon monoxide are classified as inorganic.
Read about the differences between inorganic compounds and organic compounds in
the article chemical compound.
More From Britannica
chemical compound: Inorganic compounds
zoom_in
structure of the three-centre, two-electron bond in a B―H―B fragment of a diborane
molecule
Borane is an example of an inorganic compound.
Inorganic compounds include compounds that are made up of two or more elements
other than carbon, as well as certain carbon-containing compounds that lack carbon-
carbon bonds, such as cyanides and carbonates. Inorganic compounds are most often
classified in terms of the elements or groups of elements that they contain. Oxides, for
example, can be either ionic or molecular. Ionic oxides contain O2− (oxide) ions and
metal cations, whereas molecular oxides contain molecules in which oxygen (O) is
covalently bonded to other nonmetals such as sulfur (S) or nitrogen (N). When ionic
https://www.britannica.com/science/inorganic-compound 6/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
oxides are dissolved in water, the O2− ions react with water molecules to form hydroxide
ions (OH−), and a basic solution results. Molecular oxides react with water to produce
oxyacids, such as sulfuric acid (H2SO4) and nitric acid (HNO3). In addition, inorganic
compounds include hydrides (containing hydrogen atoms or H− ions), nitrides
(containing N3− ions), phosphides (containing P3− ions), and sulfides (containing S2−
ions).
Transition metals form a great variety of inorganic compounds. The most important of
these are coordination compounds in which the metal atom or ion is surrounded by two
to six ligands. Ligands are ions or neutral molecules with electron pairs that they can
donate to the metal atom to form a coordinate-covalent bond.
The resulting covalent bond is given a special name because one entity (the ligand)
furnishes both of the electrons that are subsequently shared in the bond. An example of
a coordination compound is [Co(NH3)6]Cl3, which contains the Co(NH3)63+ ion, a
cobalt ion (Co3+) with six ammonia molecules (NH3) attached to it, acting as ligands.
Get a Britannica Premium subscription and gain access to exclusive content.
Subscribe Now
In the early days of the science of chemistry, there was no systematic approach to
naming compounds. Chemists coined names such as sugar of lead, quicklime, milk of
magnesia, Epsom salts, and laughing gas to describe familiar compounds. Such names
are called common or trivial names. As chemistry advanced, it became evident that, if
common names were used for all known compounds, which number in the millions,
great confusion would result. It clearly would be impossible to memorize trivial names
for such a large number of compounds. Therefore a systematic nomenclature (naming
process) has been developed. There are, however, certain familiar compounds that are
always referred to by their common names. The systematic names for H2O and NH3, for
https://www.britannica.com/science/inorganic-compound 7/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
example, are never used; these vital compounds are known only as water and ammonia,
respectively.
The simplest chemical compounds are binary compounds—those consisting of two
elements. Different rules apply for the nomenclature of binary ionic compounds and
binary molecular (covalent) compounds, and so they will be considered separately.
Binary compounds
Binary ionic compounds
The nomenclature for binary ionic compounds simply entails naming the ions according
to the following rules:
1. The positive ion (called a cation) is named first and the negative ion (anion)
second.
2. A simple cation (obtained from a single atom) takes its name from its parent
element. For example, Li+ is called lithium in the names of compounds
containing this ion. Similarly, Na+ is called sodium, Mg2+ is called
magnesium, and so on.
3. A simple anion (obtained from a single atom) is named by taking the root of
the parent element’s name and adding the suffix -ide. Thus, the F− ion is
called fluoride, Br− is called bromide, S2− is called sulfide, and so on.
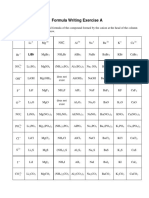
The following examples illustrate the nomenclature rules for binary ionic compounds:
compound ions present name
NaCl Na+, Cl− sodium chloride
KI K + , I− potassium iodide
CaS Ca2+, S2− calcium sulfide
CsBr Cs+, Br− cesium bromide
MgO Mg2−, O2− magnesium oxide
https://www.britannica.com/science/inorganic-compound 8/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
In the formulas of ionic compounds, simple ions are represented by the chemical
symbol for the element: Cl means Cl−, Na means Na+, and so on. When individual ions
are shown, however, the charge is always included. Thus, the formula of potassium
bromide is given as KBr, but, when the potassium and bromide ions are shown
individually, they are written K+ and Br−.
When a given metal atom can form more than one type of cation, the charge on the
particular cation present must be specified in the name of the compound. For example,
lead (Pb) can exist as Pb2+ or Pb4+ ions in ionic compounds. Also, iron (Fe) can form
Fe2+ or Fe3+ ions, tin (Sn) can form Sn2+ or Sn4+ ions, gold (Au) can form Au+ or Au3+
ions, and so on. Therefore, the names of binary compounds containing metals such as
these must include a Roman numeral to specify the charge on the ion. For example, the
compound FeCl3, which contains Fe3+, is named iron(III) chloride. On the other hand,
the compound FeCl2, which contains Fe2+, is designated as iron(II) chloride. In each
case, the Roman numeral in the name specifies the charge of the metal ion present.
Common simple cations and anions
cation name anion name
H+ hydrogen H− hydride
Li+ lithium F− fluoride
Na+ sodium Cl− chloride
K+ potassium Br− bromide
Cs+ cesium I− iodide
Be2+ beryllium O2− oxide
Mg2+ magnesium S2− sulfide
Ca2+ calcium
Ba2+ barium
Al3+ aluminum
Ag+ silver
https://www.britannica.com/science/inorganic-compound 9/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
An alternative system for naming compounds containing metals that form only two ions
is sometimes seen, especially in older literature. The ion with the higher charge has a
name ending in -ic, and the one with the lower charge has the suffix -ous. For example,
Fe3+ is called the ferric ion, and Fe2+ is called the ferrous ion. The names for FeCl3 and
FeCl2 are then ferric chloride and ferrous chloride, respectively.
Common ions that form multiple cations
ion systematic name alternate name
Fe3+ iron(III) ferric
Fe2+ iron(II) ferrous
Cu2+ copper(II) cupric
Cu+ copper(I) cuprous
Co3+ cobalt(III) cobaltic
Co2+ cobalt(II) cobaltous
Sn4+ tin(IV) stannic
Sn2+ tin(II) stannous
Pb4+ lead(IV) plumbic
Pb2+ lead(II) plumbous
Hg2+ mercury(II) mercuric
Hg22+(*) mercury(I) mercurous
*Mercury(I) ions always occur bound together to form Hg22+.
Binary molecular (covalent) compounds
Binary molecular (covalent) compounds are formed as the result of a reaction between
two nonmetals. Although there are no ions in these compounds, they are named in a
similar manner to binary ionic compounds. The nomenclature of binary covalent
compounds follows these rules:
1. The first element in the formula is given first, using the element’s full name.
https://www.britannica.com/science/inorganic-compound 10/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
2. The second element is named as if it were an anion.
3. Prefixes are used to denote the numbers of atoms present. If the first element
exists as a single atom, the prefix mono- is omitted. For example, CO is called
carbon monoxide rather than monocarbon monoxide.
These examples show how the rules are applied for the covalent compounds formed by
nitrogen and oxygen:
compound systematic name common name
N 2O dinitrogen monoxide nitrous oxide (laughing gas)
NO nitrogen monoxide nitric oxide
NO2 nitrogen dioxide
N 2O 3 dinitrogen trioxide
N 2O 4 dinitrogen tetroxide
N 2O 5 dinitrogen pentoxide
To avoid awkward pronunciations, the final o or a of the prefix is often dropped when
the element name begins with a vowel. For example, N2O4 is referred to as dinitrogen
tetroxide, not dinitrogen tetraoxide, and CO is called carbon monoxide, not carbon
monooxide.
https://www.britannica.com/science/inorganic-compound 11/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
Prefixes used in
chemical nomenclature
prefix number of atoms
mono- 1
di- 2
tri- 3
tetra- 4
penta- 5
hexa- 6
hepta- 7
octa- 8
Nonbinary compounds
Ionic compounds containing polyatomic ions
A special type of ionic compound is exemplified by ammonium nitrate (NH4NO3),
which contains two polyatomic ions, NH4+ and NO3−. As the name suggests, a
polyatomic ion is a charged entity composed of several atoms bound together.
Polyatomic ions have special names that are used in the nomenclature of the
compounds containing them.
https://www.britannica.com/science/inorganic-compound 12/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
Common polyatomic ions
ion name ion name
NH4+ ammonium CO32− carbonate
NO2− nitrite HCO3− hydrogen carbonate**
NO3− nitrate ClO− hypochlorite
SO32− sulfite ClO2− chlorite
SO42− sulfate ClO3− chlorate
HSO4− hydrogen sulfate* ClO4− perchlorate
OH− hydroxide C2H 3O 2− acetate
CN− cyanide MnO4− permanganate
PO43− phosphate Cr2O72− dichromate
HPO42− hydrogen phosphate CrO42− chromate
H2PO4− dihydrogen phosphate O22− peroxide
*Bisulfate and **bicarbonate are widely used common names for hydrogen sulfate
and hydrogen carbonate, respectively.
Several series of polyatomic anions exist that contain an atom of a given element in
combination with different numbers of oxygen atoms. Such anions are called oxy
anions. When the series contains only two members, the name of the ion with fewer
oxygen atoms ends in -ite, and the name of the other ion ends in -ate. For example,
SO32− is called sulfite and SO42− is called sulfate. In those cases where more than two
oxy anions constitute the series, hypo- (less than) and per- (more than) are used as
prefixes to name the members of the series with the smallest and the largest number of
oxygen atoms, respectively. The chlorine-containing oxy anions provide an example:
https://www.britannica.com/science/inorganic-compound 13/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
ClO− hypochlorite
ClO2− chlorite
ClO3− chlorate
ClO4− perchlorate
Naming ionic compounds that contain polyatomic ions is similar to naming binary ionic
compounds. For example, the compound NaOH is called sodium hydroxide, because it
contains the Na+ (sodium) cation and the OH− (hydroxide) anion. As in binary ionic
compounds, when a metal that can form multiple cations is present, a Roman numeral
is required to specify the charge on the cation. For example, the compound FeSO4 is
called iron(II) sulfate, because it contains Fe2+.
Acids
An acid can be thought of as a molecule containing at least one hydrogen cation (H+)
attached to an anion. The nomenclature of acids depends on whether the anion contains
oxygen. If the anion does not contain oxygen, the acid is named with the prefix hydro-
and the suffix -ic. For example, HCl dissolved in water is called hydrochloric acid.
Likewise, HCN and H2S dissolved in water are called hydrocyanic and hydrosulfuric
acids, respectively.
If the anion of the acid contains oxygen, the name is formed by adding the suffix -ic or -
ous to the root name of the anion. If the anion name ends in -ate, the -ate is replaced by
-ic (or sometimes -ric). For example, H2SO4 contains the sulfate anion (SO42−) and is
called sulfuric acid; H3PO4 contains the phosphate anion (PO43−) and is called
phosphoric acid; and HC2H3O2, which contains the acetate ion (C2H3O2−), is called
acetic acid. For anions with an -ite ending, the -ite is replaced by -ous in naming the
acid. For example, H2SO3, which contains sulfite (SO32−), is called sulfurous acid; and
HNO2, which contains nitrite (NO2−), is named nitrous acid. The acids of the oxy anions
of chlorine are used here to illustrate the rules for naming acids with oxygen-containing
cations.
https://www.britannica.com/science/inorganic-compound 14/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
Names of less common acids
formula name
H3BO3 orthoboric acid*
H2CO3 carbonic acid
H3PO4 orthophosphoric acid**
H 4 P2 O7 pyrophosphoric acid
H5P3O10 triphosphoric acid
(HPO3)n metaphosphoric acid
(HPO3keyboard_arrow_right
Home )3 Visual trimetaphosphoric acid
Arts keyboard_arrow_right Decorative Art
H3PO3 phosphorous acid
Arts & Culture
H PO hypophosphorous acid
3 2
Georg Jensen more_vert Actions
H2SO5 silversmith
Danish peroxosulfuric acid
Written and fact-checked by The Editors of Encyclopaedia Britannica
H2Updated:
Last S2 O6 dithionic
Article acid
History
H 2 S2 O3 thiosulfuric acid
HMnO4 permanganic acid
toc Table of Contents
*Often called boric acid.
**Often called phosphoric acid.
https://www.britannica.com/science/inorganic-compound 15/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
Names of common acids
formula name
HF hydrofluoric acid
HCl hydrochloric acid
HBr hydrobromic acid
HI hydroiodic acid
HCN hydrocyanic acid
H2S hydrosulfuric acid
zoom_in
HNO3 nitric acid
HNO2 nitrous acid
H2SO4 sulfuric acid
H2SO3 sulfurous acid
HC2H3O2 acetic acid
acid formula anion name
Georg Jensen
HClO4 perchlorate perchloric acid
See all media
HClO3 chlorate chloric acid
Category: Artschlorite
HClO & Culture chlorous acid
2
Born: August 31, 1866, Raadvad, Denmark
HClO hypochlorite hypochlorous acid
Died: October 2, 1935, Copenhagen (aged 69)
Compounds with complex ions
See all related content →
A coordination compound is composed of one or more complex structural units, each of
which has a central atom bound directly to a surrounding set of groups called ligands.
The nomenclature of coordination compounds is based on these structural
Georg Jensen, (born August 31, 1866, Raadvad, Denmark—died October 2, 1935,
relationships.
Copenhagen), Danish silversmith and designer who achieved international prominence
for his commercial application of modern metal design. The simple elegance of his
Stevenand
works S. Zumdahl
their emphasis on fine craftsmanship, hallmarks of Jensen’s products, are
recognized around the world.
https://www.britannica.com/science/inorganic-compound 16/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
Jensen was apprenticed to a goldsmith at age 14. His artistic talents were briefly focused
on sculpture, but he returned to metalwork, primarily jewelry and silver pieces,
produced in the workshop he opened in Copenhagen in 1904. Jensen exhibited his
works at several major foreign exhibitions (winning a gold medal at the Brussels
Exhibition of 1910) and quickly built a reputation as an outstanding and highly original
silversmith. He moved to a larger workshop in 1912 and acquired his first factory
building in 1919.
Jensen’s silverware achieved immediate popularity and commercial success. He was, in
fact, the first silvermaker to realize a profit from the manufacture of modern silver.
Until Jensen’s time virtually all successful silverware producers had relied on a standard
repertory of popular traditional designs. Jensen, however, found that the market for his
Load Next Page
keyboard_arrow_down
sleek, simple pieces was larger than anyone had predicted. His firm grew rapidly,
expanding throughout Europe and opening branches in London and New York City. On
both continents Jensen’s work set trends for contemporary tableware. He was among
the first designers to fashion steel—formerly considered fit only for low-quality,
inexpensive flatware—into handsome, serviceable cutlery.
By 1935 the firm had branches all over the world and carried more than 3,000 patterns
in open stock. Out of fairness to his customers, Jensen refused to discontinue old
patterns, a practice common among manufacturers of fine silver and tableware. After
his death the business was carried on by his son, Søren Georg Jensen. In 1973 the
company became part of the Royal Scandinavia Group.
https://www.britannica.com/science/inorganic-compound 17/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
https://www.britannica.com/science/inorganic-compound 18/19
�1/25/24, 3:45 PM Inorganic compound | Definition & Examples | Britannica
https://www.britannica.com/science/inorganic-compound 19/19