Professional Documents
Culture Documents
Picking Up The Pace of A Slow Heartbeat: Presented By: Insert Physician Name/Title
Picking Up The Pace of A Slow Heartbeat: Presented By: Insert Physician Name/Title
Uploaded by
rohithOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Picking Up The Pace of A Slow Heartbeat: Presented By: Insert Physician Name/Title
Picking Up The Pace of A Slow Heartbeat: Presented By: Insert Physician Name/Title
Uploaded by
rohithCopyright:
Available Formats
Picking up the Pace of a Slow Heartbeat
Presented by: Insert Physician Name/Title
Does This Sound Familiar?
©iStockphoto.com ©iStockphoto.com ©iStockphoto.com
Out of breath Wears you out Makes you dizzy
This isn’t normal and may be the first
signs of a heart rhythm problem.
2 © 2011 Medtronic Content is Confidential and Proprietary
Aging is Inevitable…Slowing Down
Doesn’t Have to Be…
• A healthy heart beats
between 60–100 times
per minute
• A slower heartbeat can’t
pump enough oxygen-rich
blood to the body
• Can cause lethargy, shortness
of breath, dizziness, fainting
• Limits ability to exercise, or
even do normal daily activities.
©iStockphoto.com
3 © 2011 Medtronic Content is Confidential and Proprietary
There’s a Name for It!
Bradycardia:
A slow or irregular heart rhythm, usually
less than 60 beats per minute
– What causes it?
– At what age does this generally set in?
– What are the symptoms?
– How do I find out if I have it?
4 © 2011 Medtronic Content is Confidential and Proprietary
How Can I Pick Up the Pace?
• Lifestyle changes
• Medication therapy
• Implantable pacemakers
– What is a pacemaker?
– How does it work?
– How do I know if it is time
for a pacemaker?
– Living with a pacemaker
5 © 2011 Medtronic Content is Confidential and Proprietary
Can I Lead a Normal Life with a
Pacemaker?
Yes – Let’s address some commonly
you can! asked questions:
Can I go through airport security systems?
Can I use a cell or cordless phone?
Are household appliances safe to use?
Are physical activities safe?
What happens when the battery runs out?
6 © 2011 Medtronic Content is Confidential and Proprietary
What Else Can Happen?
• It’s no secret;
issues with our
health increase as
we age
• Cancer
• Stroke
• Joint or muscle
pain/injuries
©iStockphoto.com
• Neurological diseases
7 © 2011 Medtronic Content is Confidential and Proprietary
Raise Your Hand…
• If you’ve ever undergone an MRI
• Magnetic resonance imaging (MRI) –
is the preferred imaging technology among
physicians to find out “what’s wrong”
• Speed and accuracy of the picture it provides
allows for more rapid diagnosis
8 © 2011 Medtronic Content is Confidential and Proprietary
…And What If I Need a Pacemaker?
Traditional pacemakers are Historically, the FDA and
not compatible with MRI doctors have recommended
because of a risk of harmful that pacemaker patients NOT
interaction between the undergo an MRI
device and the
electromagnetic field1,2,3,4
30,000
>
times
PO
F ©iStockphoto.com
©iStockphoto.com
1. Faris OP, Shein M. Food and Drug Administration perspective: Magnetic resonance imaging of pacemaker and implantable cardioverter‐defibrillator patients. Circulation 2006;114:1232‐1233.
2. Roguin A, Schwitter J, Vahlhaus C, et al. Magnetic resonance imaging in individuals with cardiovascular implantable electronic devices. Europace 2008;10:336 ‐346.
3. Levine GN, Gomes AS, Arai AE, et al. Safety of magnetic resonance imaging in patients with cardiovascular devices: an American Heart Association scientific statement from the Committee on
Diagnostic and Interventional Cardiac Catheterization, Council on Clinical Cardiology, and the Council on Cardiovascular Radiology and Intervention: endorsed by the American College of Cardiology
Foundation, the North American Society for Cardiac Imaging, and the Society for Cardiovascular Magnetic Resonance. Circulation 2007;116:2878‐2891.
4. Kalin R and Stanton MS. Current clinical issues for MRI scanning of pacemaker and defibrillator patients. PACE 2005;28:326 ‐328.
9 © 2011 Medtronic Content is Confidential and Proprietary
Did You Know…?
200,000 Pacemaker patients in the US annually
are denied access to an MRI scan5
50-75%
probability that cardiac device patients
will be indicated for an MRI over the
lifetime of their devices6
5. Medtronic calculations cited in Rod Gimbel and Ted McKenna, “Safety of Implantable Pacemakers and Cardioverter Defibrillators in the Magnetic Resonance Imaging Environment,”
Business Briefing: Long‐Term Healthcare 2005 (2005) available at www.touchbriefings.co
6. Kalin R and Stanton MS. Current clinical issues for MRI scanning of pacemaker and defibrillator patients. PACE 2005;28:326‐328.
10 © 2011 Medtronic Content is Confidential and Proprietary
Why Is Access To MRI So
Important?
• Provides highly detailed images of soft
tissues (organs, blood vessels, muscles,
joints, tumors, areas of infection, etc.)
• More accurate than X-ray or CAT scan
• Use of MRI* on the rise as the population
ages
• Each year:
‾ 60 million MRIs are performed
worldwide8
‾ 30 million MRI scans performed in the
U.S.9
* Commonly used to diagnose strokes, cancer, Alzheimer’s disease, muscle, bone and back pain – all commonly seen as we age
8. Sutton R, Kanal E, Wilkoff BL, Bello D, et al. Safety of magnetic resonance imaging of patients with a new Medtronic EnRhythm MRI SureScan pacing system: clinical study design. Trials 2008, 9:68.
9. IMV, “Benchmark Report: MRI 2007,” IMV Medical Information Division. Des Plaines, IL. 2008
11 © 2011 Medtronic Content is Confidential and Proprietary
Is There Anything I Can Do?
Anyone needing a pacemaker now has a
Yes – new option that allows MRI access
you can! First and only pacemaker system FDA-approved for use in
an MRI environment
“ I am in the medical field. I am a radiologist
and I didn’t even know that there was an MRI
conditional pacemaker out there when I
needed a pacemaker. It took me having to “
ask my doctor about it
–Dr. Lee Friedman
12 © 2011 Medtronic Content is Confidential and Proprietary
Where To Go for More Information
• Important resources
and informational tools:
• Disease and device
information
• Frequently Asked
Questions
• Quiz: My Pacemaker. My
MRI. My Guide
• Personal stories:
spokesperson Debbie
Allen, pacemaker patients
• Share your story and
become a Pace Maker!
www.JointhePaceMakers.com
13 © 2011 Medtronic Content is Confidential and Proprietary
Take an Active Role in Making
Treatment Decisions with Your Doctor
Ask me
questions!
©iStockphoto.com
10. National Council on Aging (NCOA) Survey on Medical Imaging Safety, February 2011.
14 © 2011 Medtronic Content is Confidential and Proprietary
Important Safety Information
• An implantable pacemaker system relieves symptoms of heart rhythm disturbances. They do this by restoring
normal heart rates. A normal heart rate provides your body with the proper amount of blood circulation. The
pacemaker system is intended for patients who need rate-adaptive pacing or chronic pacing or for patients who
may benefit from synchronizing the pumping of the heart chambers.
• Risks associated with pacemaker system implant include, but are not limited to, infection at the surgical site
and/or sensitivity to the device material, failure to deliver therapy when it is needed, or receiving extra therapy
when it is not needed. After receiving an implantable pacemaker system, you will have limitations with magnetic
and electromagnetic radiation, electric or gas powered appliances, and tools with which you are allowed to be in
contact.
• A complete SureScan pacing system including a Revo MRI SureScan IPG and two CapSureFix MRI SureScan
leads is required for use in the MRI environment. Any other pacing system combination may result in a hazard to
the patient during an MRI scan. When programmed to On, the MRI SureScan feature allows the patient to be
safely scanned while the device continues to provide appropriate pacing. The Revo MRI SureScan pacing system
is MR Conditional. This means the pacing system is designed to allow patients to undergo MRI, when your doctor
determines you meet patient eligibility requirements and the scan is conducted according to Medtronic directions.
• This treatment is prescribed by your physician. This treatment is not for everyone. Please talk to your doctor to
see if it is right for you. Your physician should discuss all potential benefits and risks with you. Although many
patients benefit from the use of this treatment, results may vary.
• For further questions, contact patient services at 1 (800) 551-5544, x41835.
15 © 2011 Medtronic Content is Confidential and Proprietary
You might also like
- Algorithm-PALS Ped Systematic Approach 200615Document1 pageAlgorithm-PALS Ped Systematic Approach 200615Erica Yamamoto100% (2)
- Cardiovascular MCQs For Pharmacology ReviewDocument14 pagesCardiovascular MCQs For Pharmacology ReviewBhopesh Kadian38% (8)
- 2017 Spotlight On Health Winter Edition For HickoryDocument80 pages2017 Spotlight On Health Winter Edition For HickoryCrystal Sutherland DosterNo ratings yet
- CVA Case PresentationDocument8 pagesCVA Case Presentationrichie_chan_2100% (3)
- @MedicalBooksStoreS 2017 Acute IschemicDocument273 pages@MedicalBooksStoreS 2017 Acute IschemicAditya Perdana Dharma Wiguna100% (1)
- CRRT Nursing ManagementDocument14 pagesCRRT Nursing Managementanagh cuphu100% (2)
- CBP Patient Class 4-01Document54 pagesCBP Patient Class 4-01Soarin626No ratings yet
- World Brain Tumor Day, 2021Document40 pagesWorld Brain Tumor Day, 2021shavetaNo ratings yet
- Chiari Malformation: An Overview: Rick Labuda, Executive DirectorDocument18 pagesChiari Malformation: An Overview: Rick Labuda, Executive DirectornsurgeonNo ratings yet
- Electromagnetic Compatibility PDFDocument10 pagesElectromagnetic Compatibility PDFRedouane NaceriNo ratings yet
- Electromagnetic Compatibility PDFDocument10 pagesElectromagnetic Compatibility PDFRedouane NaceriNo ratings yet
- 5 Hand OUTDocument22 pages5 Hand OUTCarol AnnNo ratings yet
- Introduction To Rehabilitation Engineering: Raye Yeow Department of Biomedical EngineeringDocument38 pagesIntroduction To Rehabilitation Engineering: Raye Yeow Department of Biomedical EngineeringJerome WongNo ratings yet
- Braintumor - 7Document31 pagesBraintumor - 7Riju DasNo ratings yet
- Brain Tumor-A Case SudyDocument9 pagesBrain Tumor-A Case SudyJemaikha Hinata100% (1)
- Ems StrokeDocument29 pagesEms StrokeaathulNo ratings yet
- RU - Trauma and Emergency CareDocument32 pagesRU - Trauma and Emergency CareryanNo ratings yet
- Mild Cognitive Impairment FactsheetDocument6 pagesMild Cognitive Impairment FactsheetJoasVinsensiusDavianNo ratings yet
- Management of Patients With Oncologic or Degenerative Neurologic DisordersDocument32 pagesManagement of Patients With Oncologic or Degenerative Neurologic DisordersFeliciaDorghamNo ratings yet
- A Window Into The Brain: Neuroimaging and Mental IllnessDocument8 pagesA Window Into The Brain: Neuroimaging and Mental IllnessvaibswebNo ratings yet
- MRI ReportDocument47 pagesMRI ReportRizell MangubatNo ratings yet
- Koroshetz Stroke 1Document36 pagesKoroshetz Stroke 1vladNo ratings yet
- SaferDocument57 pagesSaferah0507No ratings yet
- HPD Group 10Document20 pagesHPD Group 10Shruti SinghNo ratings yet
- Brain DeathDocument19 pagesBrain DeathWira WicaksanaNo ratings yet
- Brain Injury BasicsDocument64 pagesBrain Injury BasicsSilvanaPutri100% (2)
- Intro (MVA)Document36 pagesIntro (MVA)dustin_camposNo ratings yet
- Advanced Clinical Coaching: For NES Health PractitionersDocument15 pagesAdvanced Clinical Coaching: For NES Health PractitionersLearnItLive100% (1)
- Explaining Stroke 101Document29 pagesExplaining Stroke 101Christopher AdhisasmitaNo ratings yet
- Seizures: Risk Awareness Tool (RAT)Document22 pagesSeizures: Risk Awareness Tool (RAT)Ak AkNo ratings yet
- EpilepsyDocument37 pagesEpilepsyNader Smadi100% (7)
- Acquired Brain Injury Early Rehabilitation and Long Term OutcomeDocument46 pagesAcquired Brain Injury Early Rehabilitation and Long Term OutcomeVeena RaigangarNo ratings yet
- Geriatric Trauma: Top 10 Lessons From Older Patients: Nursing2010Document4 pagesGeriatric Trauma: Top 10 Lessons From Older Patients: Nursing2010Reychelle ChecheNo ratings yet
- Adult Routine Head CTDocument17 pagesAdult Routine Head CTAvan IchsanNo ratings yet
- Bidmc GraceDocument55 pagesBidmc GracemelissamattisonNo ratings yet
- Acoustic Neuromas: Information For Patients and FamiliesDocument8 pagesAcoustic Neuromas: Information For Patients and FamiliesMrgNo ratings yet
- Life After Stroke Guide - 7819Document36 pagesLife After Stroke Guide - 7819Dr. Victor A. RochaNo ratings yet
- Spinal Cord Trauma - Harvard HealthDocument1 pageSpinal Cord Trauma - Harvard Healthstephanealves17No ratings yet
- Brain TumorsDocument30 pagesBrain TumorsDagmawi BahiruNo ratings yet
- MLP Summer2012webDocument32 pagesMLP Summer2012webEditorial CapitalNo ratings yet
- Rehabilitation in Tbi: by DR Khiu Fu Lung MDDocument35 pagesRehabilitation in Tbi: by DR Khiu Fu Lung MDQ90No ratings yet
- Geriatric Trauma: Florence A MDocument19 pagesGeriatric Trauma: Florence A MflorenceNo ratings yet
- The Code Stroke Handbook Approach To The Acute Stroke Patient 1St Edition Andrew Micieli Full Download ChapterDocument51 pagesThe Code Stroke Handbook Approach To The Acute Stroke Patient 1St Edition Andrew Micieli Full Download Chaptersue.duron301100% (4)
- Peripheral Nerve InjuriesDocument8 pagesPeripheral Nerve InjuriesAli AftabNo ratings yet
- Brain Tumor (Group B)Document29 pagesBrain Tumor (Group B)Srush AliNo ratings yet
- Consensus Statement On Brain Death - Malaysia - 2003Document25 pagesConsensus Statement On Brain Death - Malaysia - 2003docpunit100% (1)
- Brain CancerDocument12 pagesBrain CancerERROR WEBNo ratings yet
- PocketCard Assessment and Management of Stroke July 2020Document12 pagesPocketCard Assessment and Management of Stroke July 2020chellczyNo ratings yet
- Neurosurgery: A Case-Based Approach Christos M. Tolias Anastasios Giamouriadis Florence Rosie Avila Hogg Prajwal GhimireDocument225 pagesNeurosurgery: A Case-Based Approach Christos M. Tolias Anastasios Giamouriadis Florence Rosie Avila Hogg Prajwal GhimireMystero Rasico100% (1)
- ContentServer PDFDocument17 pagesContentServer PDFReiza IndraNo ratings yet
- Brain Tumours IntroductionDocument11 pagesBrain Tumours IntroductionsonyNo ratings yet
- Brain Tumors: What You Need To Know AboutDocument51 pagesBrain Tumors: What You Need To Know AboutIndahNo ratings yet
- CASE PRES Cva.Document22 pagesCASE PRES Cva.pamelaideaNo ratings yet
- 05 Cognitive Changes With AgeingDocument29 pages05 Cognitive Changes With AgeingAdeel AhmadNo ratings yet
- How Long Does It Take To Get MRI ResultsDocument15 pagesHow Long Does It Take To Get MRI ResultsAubrey RazonNo ratings yet
- FC Back To Sports Meeting Presentation Slides and Script With Video UCM - 490125Document53 pagesFC Back To Sports Meeting Presentation Slides and Script With Video UCM - 490125redaNo ratings yet
- CP AssessmentDocument10 pagesCP AssessmentMunna KendreNo ratings yet
- Introduction To Cerebral PalsyDocument23 pagesIntroduction To Cerebral PalsyVenkata Nagaraj Mummadisetty100% (1)
- Fall Alarm Monitoring NFDDocument17 pagesFall Alarm Monitoring NFDcody whiteNo ratings yet
- Lect 1 (Ask)Document33 pagesLect 1 (Ask)Personal growthNo ratings yet
- Personalized Medicine and Psychiatry - Dream or RealityDocument6 pagesPersonalized Medicine and Psychiatry - Dream or RealityAwais QureshiNo ratings yet
- Dementia Marc AntonioDocument55 pagesDementia Marc AntonioAgitha Melita PutriNo ratings yet
- Islamic University of Technology (IUT) Organisation of Islamic Cooperation (OIC)Document2 pagesIslamic University of Technology (IUT) Organisation of Islamic Cooperation (OIC)rohithNo ratings yet
- Nonrenewable AND Renewable ResourcesDocument16 pagesNonrenewable AND Renewable ResourcesrohithNo ratings yet
- OBE Course Outline - EEE 4895Document4 pagesOBE Course Outline - EEE 4895rohithNo ratings yet
- Mousum Kabir Rabbi: ContactDocument4 pagesMousum Kabir Rabbi: ContactrohithNo ratings yet
- Syllabus: Diploma in Technical EducationDocument7 pagesSyllabus: Diploma in Technical EducationrohithNo ratings yet
- Defibrillator: DR Sumanth ReddyDocument39 pagesDefibrillator: DR Sumanth Reddyrohith100% (1)
- Fossil Fuels Coal: Power Lines From Coal Burning Power Plant in Pekin, ILDocument52 pagesFossil Fuels Coal: Power Lines From Coal Burning Power Plant in Pekin, ILrohithNo ratings yet
- Renewable Energy Resources: Textile ProcessingDocument17 pagesRenewable Energy Resources: Textile ProcessingrohithNo ratings yet
- Educational Technology: Meaning, Nature and ScopeDocument19 pagesEducational Technology: Meaning, Nature and ScoperohithNo ratings yet
- 6 DefibrillationDocument22 pages6 DefibrillationrohithNo ratings yet
- Well Come To My Power Point Presentation Greetings!: Islamic University of Technology EEEDocument28 pagesWell Come To My Power Point Presentation Greetings!: Islamic University of Technology EEErohithNo ratings yet
- Lesson 8 - Et332b (2) .PPSXDocument28 pagesLesson 8 - Et332b (2) .PPSXrohithNo ratings yet
- ScvO2 Pocket Card - 2017 PDFDocument3 pagesScvO2 Pocket Card - 2017 PDFghg sddNo ratings yet
- 3 Vs Apical PulseDocument2 pages3 Vs Apical Pulse잔잔No ratings yet
- JAAD CME Aug 2023 - Cutaneous Manifestations of Cardiovascular and Vascular DiseaseDocument39 pagesJAAD CME Aug 2023 - Cutaneous Manifestations of Cardiovascular and Vascular DiseasejchengwangNo ratings yet
- Point-Of-Care Ultrasound in Cardiac ArrestDocument13 pagesPoint-Of-Care Ultrasound in Cardiac ArrestEgorSazhaevNo ratings yet
- BPJ Vol 14 No 2 P 961-969Document9 pagesBPJ Vol 14 No 2 P 961-969Sifa ShopingNo ratings yet
- Federal Employee Program Incentive For Using CPT Category Ii CodesDocument2 pagesFederal Employee Program Incentive For Using CPT Category Ii CodesngNo ratings yet
- BP ChartDocument1 pageBP Chartabu ubaidahNo ratings yet
- Wolff-Parkinson-White Syndrome: Anatomy, Epidemiology, Clinical Manifestations, and Diagnosis - UpToDateDocument46 pagesWolff-Parkinson-White Syndrome: Anatomy, Epidemiology, Clinical Manifestations, and Diagnosis - UpToDateKarla ChongNo ratings yet
- Ecg Ospe QuizDocument26 pagesEcg Ospe Quizsivakamasundari pichaipillaiNo ratings yet
- Tony Cross CDU Low Risk ACS Proforma - V3.212 Review Due June 2014Document4 pagesTony Cross CDU Low Risk ACS Proforma - V3.212 Review Due June 2014ED@HEFTNo ratings yet
- Catheter Ablation in End Stage Heart FailureDocument10 pagesCatheter Ablation in End Stage Heart FailureJose Gomez MorenoNo ratings yet
- Company Canon Canon Canon Ge Healthcge Healthcge Healthcge Healthcge Healthcneusoft MeDocument27 pagesCompany Canon Canon Canon Ge Healthcge Healthcge Healthcge Healthcge Healthcneusoft MeAnguschowNo ratings yet
- Pearls in Neonatal ResuscitationDocument2 pagesPearls in Neonatal ResuscitationAdam ParadiseNo ratings yet
- Digital Ischemic Compression Technique Aims at Evoking Temporary IschaemicDocument2 pagesDigital Ischemic Compression Technique Aims at Evoking Temporary IschaemicNAvi SinghNo ratings yet
- ELSO Interim Guidelines For Venoarterial.2Document18 pagesELSO Interim Guidelines For Venoarterial.2BiancaPancuNo ratings yet
- LercanidipineDocument8 pagesLercanidipineddandan_2No ratings yet
- Mitral Valve ProapseDocument8 pagesMitral Valve ProapseEgorSazhaevNo ratings yet
- Topic 9 Cardiovascular SystemDocument36 pagesTopic 9 Cardiovascular SystemAlejandra CastilloNo ratings yet
- American Society of ExtraCorporeal Technology Standards and GuidelinesDocument22 pagesAmerican Society of ExtraCorporeal Technology Standards and GuidelinesErkanNo ratings yet
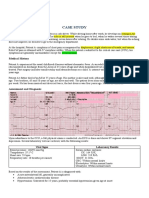
- Case Study With ECG Reading OcañaDocument3 pagesCase Study With ECG Reading OcañaNicole Chloe OcanaNo ratings yet
- Introduction Definition: Types of PacemakersDocument8 pagesIntroduction Definition: Types of PacemakersPrasann RoyNo ratings yet
- Circulatory System NotesDocument8 pagesCirculatory System Notesthe lillyNo ratings yet
- FHC Data EntryDocument19 pagesFHC Data EntryLittle DevilNo ratings yet
- Tier 1 CVSDocument23 pagesTier 1 CVSCHETHAN K SNo ratings yet
- Conginital Heart DiseaseDocument19 pagesConginital Heart DiseaseSanthosh.S.UNo ratings yet
- Heart Awareness Paula Cervera Joana Villanueva Regina IbarraDocument2 pagesHeart Awareness Paula Cervera Joana Villanueva Regina IbarraPaula CerveraNo ratings yet