Professional Documents
Culture Documents
7.5 Water Potential
Uploaded by
emanuel coates0 ratings0% found this document useful (0 votes)
13 views7 pagesCopyright
© © All Rights Reserved
Available Formats
PPTX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
13 views7 pages7.5 Water Potential
Uploaded by
emanuel coatesCopyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
You are on page 1of 7
Water Potential
What is water potential?
• The measure of the relative tendency of water to
move from one area to another, and
• It is commonly represented by the Greek letter
Ψ (Psi).
What is pressure potential?
• Both pressure and solute concentration affect
water potential.
• Ψw = Ψp + Ψs
• Pressure potential is the physical pressure on
a solution and can be positive or negative. Ψs
What is solute potential?
• The solute potential Ψs (or osmotic potential) of a
solution is proportional to the number of dissolved solute
molecules.
• By definition, the solute potential of pure water is 0.
• The addition of solutes lowers the water potential
because the solutes bind water molecules, which have less
freedom to move than they do in pure water.
• Any solution at atmospheric pressure has a negative water
potential.
• Water potential affects the uptake and loss of
water in plant cells.
▫ In a flaccid cell, Ψp = 0 and the cell is limp.
▫ If this cell is placed in a solution with a higher
solute concentration (and, therefore, a lower Ψw),
water will leave the cell by osmosis.
▫ Eventually, the cell will plasmolyze by shrinking
and pulling away from its wall.
• If a flaccid cell is placed in pure water (Ψw = 0), the
cell will have lower water potential than pure water
due to the presence of solutes, and water will enter
the cell by osmosis.
• As the cell begins to swell, it will push against the cell
wall, producing turgor pressure.
• The partially elastic wall will push back until this
pressure is great enough to offset the tendency for
water to enter the cell because of solutes.
Aquaporins affect the rate of water
transport across membranes.
• Both plant and animal membranes have specific
transport proteins, aquaporins, which facilitate
the passive movement of water across a
membrane.
• Aquaporins do not affect the water potential
gradient or the direction of water flow, but
rather increase the rate at which water diffuses
down its water potential gradient.
You might also like
- Proteins 1 - Solutions FinalDocument2 pagesProteins 1 - Solutions Finalemanuel coatesNo ratings yet
- 6 Proteins - IntroDocument25 pages6 Proteins - Introemanuel coatesNo ratings yet
- Name: Emanuel Charles Coates Registration #: 2018010033 Topic: Case Inguinal HerniaDocument11 pagesName: Emanuel Charles Coates Registration #: 2018010033 Topic: Case Inguinal Herniaemanuel coatesNo ratings yet
- CAPE Chemistry Unit 2 Paper 2 2017 AnswersDocument12 pagesCAPE Chemistry Unit 2 Paper 2 2017 Answersemanuel coates100% (9)
- Summary - Module 3 - Unit2Document5 pagesSummary - Module 3 - Unit2emanuel coatesNo ratings yet
- 3 Functional GroupsDocument24 pages3 Functional Groupsemanuel coatesNo ratings yet
- 1 Chemical & Physical PhenomonenDocument62 pages1 Chemical & Physical Phenomonenemanuel coatesNo ratings yet
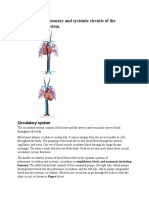
- Compare The Pulmonary and Systemic Circuits of The Cardiovascular SystemDocument6 pagesCompare The Pulmonary and Systemic Circuits of The Cardiovascular Systememanuel coatesNo ratings yet
- Chemistry Unit 2 Paper 2 (Solutions) - 1 PDFDocument66 pagesChemistry Unit 2 Paper 2 (Solutions) - 1 PDFDe'arra Brooks90% (10)
- A Introduction To Medical TerminologyDocument29 pagesA Introduction To Medical Terminologyemanuel coatesNo ratings yet
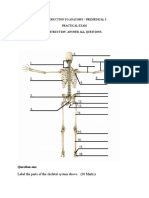
- Introduction To Anatomy - Premedical 3 Practical Exam Instruction-Answer All QuestionsDocument5 pagesIntroduction To Anatomy - Premedical 3 Practical Exam Instruction-Answer All Questionsemanuel coatesNo ratings yet
- Body OrganizationDocument74 pagesBody Organizationemanuel coatesNo ratings yet
- Medical Terminology WK 2Document32 pagesMedical Terminology WK 2emanuel coatesNo ratings yet
- Notes On THE NERVOUS SYSTEMDocument15 pagesNotes On THE NERVOUS SYSTEMemanuel coatesNo ratings yet
- Metric Conversion WorksheetDocument1 pageMetric Conversion Worksheetemanuel coates0% (1)
- Kinematics in Two DiamensionsDocument34 pagesKinematics in Two Diamensionsemanuel coatesNo ratings yet
- Kinematics in One DimensionDocument61 pagesKinematics in One Dimensionemanuel coates33% (3)
- Overcoming AnxietyDocument22 pagesOvercoming Anxietyemanuel coatesNo ratings yet
- General Chemistry Assignment #1Document2 pagesGeneral Chemistry Assignment #1emanuel coatesNo ratings yet
- Metric Conversion WorksheetDocument1 pageMetric Conversion Worksheetemanuel coates0% (1)
- Application Form: 1) Basic InformationDocument4 pagesApplication Form: 1) Basic Informationemanuel coatesNo ratings yet
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (400)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (345)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (121)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)