Professional Documents
Culture Documents
Laporan Pagi PDF
Laporan Pagi PDF
Uploaded by
Briyya LullaniOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Laporan Pagi PDF
Laporan Pagi PDF
Uploaded by
Briyya LullaniCopyright:
Available Formats
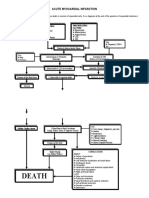
41 Pathophysiology of acute myocardial infarction
Start here Fibrous cap
Macrophages and T-lymphocytes
Stunned myocardium
24 h Lipid core Plaque rupture, platelet aggregation Transmural necrosed zone Infarct appears pale, most cells dead, neutrophils present coagulation necrosis
Mainly dead myocytes and neutrophils
Granulation tissue moves inward and replaces dead tissue with scar tissue 2h Subendocardial necrosed zone Cellular damage progressing, but still partially reversible with reperfusion Thrombus propagates into and along coronary artery 57 days
Endocardium
Cross-section of ventricular wall served by thrombosed artery
Scar thin firm grey
Mixture of living and dead myocytes; substrate for re-entrant arrhythmias
0h > 3 months Finish here
41.1
Infarction is tissue death caused by ischaemia. Acute myocardial infarction (MI) occurs when localized myocardial ischaemia causes the development of a dened region of necrosis. MI is most often caused by rupture of an atherosclerotic lesion in a coronary artery. This causes the formation of a thrombus that plugs the artery, stopping it from supplying blood to the region of the heart that it supplies.
complete thrombotic occlusion of the infarct-related artery. The incidence of total occlusion fell to 65% 1224 h after symptom onset due to spontaneous brinolysis. Fresh thrombi on top of ruptured plaques have also been demonstrated in the infarctrelated arteries in patients dying of MI.
Mechanisms and consequences of plaque rupture
Coronary plaques which are prone to rupture are typically small and nonobstructive, with a large lipid-rich core covered by a thin brous cap. Activated macrophages and T-lymphocytes localized at the site of plaque rupture are thought to release metalloproteases and cytokines which weaken the brous cap, rendering it liable to tear or erode due to the shear stress exerted by the bloodow.
Role of thrombosis in MI
Pivotal studies by DeWood and colleagues showed that coronary thrombosis is the critical event resulting in MI. Of patients presenting within 4 h of symptom onset with ECG evidence of transmural MI, coronary angiography showed that 87% had 88 Pathology and therapeutics
Plaque rupture reveals subendothelial collagen, which serves as a site of platelet adhesion, activation and aggregation. This results in: 1 The release of substances such as thromboxane A2 (TXA2 ), brinogen, 5-hydroxytryptamine (5-HT), platelet activating factor and ADP, which further promote platelet aggregation. 2 Activation of the clotting cascade, leading to brin formation and propagation and stabilization of the occlusive thrombus. The endothelium is often damaged around areas of coronary artery disease. The resulting decit of antithrombotic factors such as thrombomodulin and prostacyclin enhances thrombus formation. In addition, the tendency of several platelet-derived factors (e.g. TXA2, 5-HT) to cause vasoconstriction is increased in the absence of endothelial-derived relaxing factors. This may promote the development of local vasospasm, which worsens coronary occlusion. Sudden death and acute coronary syndrome onset show a circadian variation (daily cycle), peaking at around 9 a.m. with a trough at around 11 p.m. Levels of catecholamines peak about an hour after awakening in the morning, resulting in maximal levels of platelet aggregability, vascular tone, heart rate and blood pressure, which may trigger plaque rupture and thrombosis. Increased physical and mental stress can also cause MI and sudden death, supporting a role for increases in catecholamines in MI pathophysiology. Furthermore, chronic -adrenergic receptor blockade abolishes the circadian rhythm of MI. Autopsies of young subjects killed in road accidents often show small plaque ruptures in susceptible arteries, suggesting that plaque rupture does not always have pathological consequences. The degree of coronary occlusion and myocardial damage caused by plaque rupture probably depends on systemic catecholamine levels, as well as local factors such as plaque location and morphology, the depth of plaque rupture, and the extent to which coronary vasoconstriction occurs. Severe and prolonged ischaemia produces a region of necrosis spanning the entire thickness of the myocardial wall. Such a transmural infarct usually causes ST segment elevation (i.e. STEMI, see Chapter 38). Less severe and protracted ischaemia can arise when: 1 Coronary occlusion is followed by spontaneous reperfusion. 2 The infarct-related artery is not completely occluded. 3 Occlusion is complete, but an existing collateral blood supply prevents complete ischaemia. 4 The oxygen demand in the affected zone of myocardium is smaller. Under these conditions, the necrotic zone may be mainly limited to the subendocardium, typically causing non-ST segment elevation MI. The classication of acute MI according to the presence or absence of ST segment elevation is designed to allow rapid decision-making concerning whether thrombolysis should be initiated (see Chapter 42). This classication replaces the previous one, based on the presence or absence of Q waves on the ECG, which was less useful for guiding immediate therapy.
Evolution of the infarct
Both infarcted and unaffected myocardial regions undergo progressive changes over the hours, days and weeks following coronary thrombosis. This process of postinfarct myocardial evolution leads to the occurrence of characteristic complications at predictable times after the initial event (see Chapter 42). Ischaemia causes an immediate loss of contractility in the affected myocardium, a condition termed hypokinesis. Necrosis starts to develop in the subendocardium (which is most prone to ischaemia; see Chapter 37), about 1530 min after coronary occlusion. The necrotic region grows outward towards the epicardium over the next 36 h, eventually spanning the entire ventricular wall. In some areas (generally at the edges of the infarct) the myocardium is stunned (reversibly damaged) and will eventually recover if bloodow is restored. Contractility in the remaining viable myocardium increases, a process termed hyperkinesis. A progression of cellular, histological and gross changes develop within the infarct. Although alterations in the gross appearance of infarcted tissue are not apparent for at least 6 h after the onset of cell death, cell biochemistry and ultrastructure begin to show abnormalities within 20 min. Cell damage is progressive, becomingly increasingly irreversible over about 12 h. This period therefore provides a window of opportunity during which thrombolysis and reperfusion may salvage some of the infarct (see Chapter 43). Between 4 and 12 h after cell death starts, the infarcted myocardium begins to undergo coagulation necrosis, a process characterized by cell swelling, organelle breakdown and protein denaturation. After about 18 h, neutrophils (phagocytic lymphocytes) enter the infarct. Their numbers reach a peak after about 5 days, and then decline. After 3 4 days, granulation tissue appears at the edges of the infarct zone. This consists of macrophages, broblasts, which lay down scar tissue, and new capillaries. The infarcted myocardium is especially soft between 4 and 7 days, and is therefore maximally prone to rupturing. This event is usually fatal, may occur at any time during the rst 2 weeks and is responsible for about 10% of MI mortality. As the granulation tissue migrates inward toward the centre of the infarct over several weeks, the necrotic tissue is engulfed and digested by the macrophages. The granulation tissue then progressively matures, with an increase in connective (scar) tissue and loss of capillaries. After 23 months, the infarct has healed, leaving a noncontracting region of the ventricular wall that is thinned, rm and pale grey. Infarct expansion, the stretching and thinning of the infarcted wall, may occur within the rst day or so after a MI, especially if the infarction is large or transmural, or has an anterior location. Over the course of several months, there is progressive dilatation, not only of the infarct zone, but also of healthy myocardium. This process of ventricular remodelling is caused by an increase in end-diastolic wall stress. Infarct expansion puts patients at a sub-stantial risk for the development of congestive heart failure, ventricular arrhythmias, and free wall rupture. Pathophysiology of acute myocardial infarction 89
You might also like
- Renal Block 2Document52 pagesRenal Block 2Maya LaPradeNo ratings yet
- Acute Myocardial Infarction - CSDocument49 pagesAcute Myocardial Infarction - CSMASII94% (17)
- Acute Myocardial InfarctionDocument20 pagesAcute Myocardial InfarctionDavid Christian CalmaNo ratings yet
- Coronary Artery Disease - Case StudyDocument19 pagesCoronary Artery Disease - Case StudyJulieteeySarania100% (3)
- Freeman Refining Your Brand PersonalityDocument15 pagesFreeman Refining Your Brand Personalityjafrihusnain4035No ratings yet
- Non-St Segment Elevation Myocardial Infarction (Nstemi)Document24 pagesNon-St Segment Elevation Myocardial Infarction (Nstemi)MHIEMHOINo ratings yet
- Cardiac TamponadeDocument3 pagesCardiac TamponadeKimberly SolisNo ratings yet
- Bhaswati Bhattacharya Everyday Ayurveda DaiDocument305 pagesBhaswati Bhattacharya Everyday Ayurveda DaiRohit KumarNo ratings yet
- Cardiogenic Shock PDFDocument14 pagesCardiogenic Shock PDFAdreiTheTripleANo ratings yet
- Acute Respiratory Distress SyndromeDocument3 pagesAcute Respiratory Distress SyndromeJorie Roco100% (1)
- Divine Jyotish Com 2014-12-31 Recommended BooksDocument15 pagesDivine Jyotish Com 2014-12-31 Recommended BooksJatinder SainiNo ratings yet
- Pathophysiology CAP and HTNDocument5 pagesPathophysiology CAP and HTNLeonaPunzalanNo ratings yet
- Pathophysiology of Myocardial Infarction (STEMI)Document2 pagesPathophysiology of Myocardial Infarction (STEMI)michaela100% (3)
- Anatomy Myocardial InfarctionDocument5 pagesAnatomy Myocardial InfarctionLyka Milo AvilaNo ratings yet
- Pa Tho Physiology of Hemorrhagic StrokeDocument2 pagesPa Tho Physiology of Hemorrhagic StrokeMerlash MerlaNo ratings yet
- Patho of MIDocument2 pagesPatho of MIInchan Montesines0% (1)
- Case Study NSTEMIDocument25 pagesCase Study NSTEMImichpadua100% (2)
- Tetanus Case StudyDocument41 pagesTetanus Case StudyFAt Ty100% (1)
- Micu Case Study d1Document28 pagesMicu Case Study d1jmarc_2180% (5)
- Final Myocardial Infarction Pathophysiology PDFDocument3 pagesFinal Myocardial Infarction Pathophysiology PDFDave JoshuaNo ratings yet
- Myocardial Infarction Pathophysiology - Schematic DiagramDocument3 pagesMyocardial Infarction Pathophysiology - Schematic DiagramAbi Habiling100% (3)
- pg36-37 of Pneumothorax Case StudyDocument2 pagespg36-37 of Pneumothorax Case Studyikemas100% (1)
- Patho (Lung Cancer)Document1 pagePatho (Lung Cancer)k.n.e.d.No ratings yet
- Spinal Cord InjuriesDocument51 pagesSpinal Cord Injurieswanglee2000No ratings yet
- August - 2022 Commercial RentDocument26 pagesAugust - 2022 Commercial Rentmonty.rajputNo ratings yet
- Myocardial InfarctionDocument21 pagesMyocardial InfarctionasdnofalNo ratings yet
- Rheumatic Heart DiseaseDocument4 pagesRheumatic Heart DiseaseAlfrin Antony100% (3)
- Myocardial InfarctionDocument45 pagesMyocardial InfarctionGopal SinghNo ratings yet
- Acute Respiratory Distress Syndrome - PathophysiologyDocument5 pagesAcute Respiratory Distress Syndrome - PathophysiologyJoann67% (3)
- Pulmonary EmbolismDocument8 pagesPulmonary EmbolismspoilttbrattNo ratings yet
- Blackbuck Marks The Beginning of A New Path in Trucking. A Path That Is Organized and Makes Trucking Simple For EveryDocument5 pagesBlackbuck Marks The Beginning of A New Path in Trucking. A Path That Is Organized and Makes Trucking Simple For EveryArjun Sharma100% (1)
- AnginaDocument12 pagesAnginaHermiie Joii Galang Maglaquii0% (1)
- Case Study RheumaticDocument46 pagesCase Study RheumaticJill Anne Balderosa91% (11)
- 2016 List of DoctorsDocument29 pages2016 List of DoctorsKuch Baate Aisi BhiNo ratings yet
- 2016 Advanced Timber StructuresDocument241 pages2016 Advanced Timber Structuresjack.condori100% (1)
- Rheumatic FeverDocument3 pagesRheumatic FeverKhalid Mahmud Arifin100% (3)
- Acute Myocardial InfarctionDocument53 pagesAcute Myocardial InfarctionYew Ping Hee100% (1)
- Daily Lesson Log Weekly - Oct 28-31Document3 pagesDaily Lesson Log Weekly - Oct 28-31tin100% (1)
- Acute Myocardial InfarctionDocument32 pagesAcute Myocardial InfarctionListya Normalita100% (1)
- Hypovolemic Shock 09Document58 pagesHypovolemic Shock 09Joanne Bernadette Aguilar100% (2)
- Myocardial Infarction: Nonmodifiable Factors: Modifiable FactorsDocument4 pagesMyocardial Infarction: Nonmodifiable Factors: Modifiable FactorsHearty ArriolaNo ratings yet
- Myocardial InfarctionDocument6 pagesMyocardial InfarctionMaicie Rose Bautista VallesterosNo ratings yet
- Human Diseases Case Study 18 ADocument4 pagesHuman Diseases Case Study 18 Aairickann100% (1)
- Pathophysiology of Brain Abscess Secondary To Chronic Otitis MediaDocument5 pagesPathophysiology of Brain Abscess Secondary To Chronic Otitis Mediafufulabrador100% (1)
- Acs Nstemi Vs Ua - PathoDocument2 pagesAcs Nstemi Vs Ua - PathoJerom YamatNo ratings yet
- Acute Myocardial InfarctionDocument2 pagesAcute Myocardial InfarctionhailleyannNo ratings yet
- Cardiac TamponadeDocument3 pagesCardiac Tamponadescremo_xtreme100% (2)
- Buerger's DiseaseDocument13 pagesBuerger's DiseaseJoan JuradoNo ratings yet
- DB13 - Pathophysiology of AtherosclerosisDocument2 pagesDB13 - Pathophysiology of Atherosclerosisi_vhie03No ratings yet
- BUERGER's DiseaseDocument2 pagesBUERGER's DiseasechemnikoNo ratings yet
- Concept MapDocument4 pagesConcept Mapdejosep_informaticsNo ratings yet
- NCP HeadDocument11 pagesNCP Headann-lisel-manahan-7670100% (2)
- POLYCYTHEMIADocument24 pagesPOLYCYTHEMIAYanne BarridoNo ratings yet
- Case Study GonorrheaDocument19 pagesCase Study GonorrheaErika ThereseNo ratings yet
- Cardiogenic Shock: Cardiogenic Shock Is Based Upon An Inadequate Circulation ofDocument5 pagesCardiogenic Shock: Cardiogenic Shock Is Based Upon An Inadequate Circulation ofhabeeb23No ratings yet
- Pathophysiology of Angina PectorisDocument1 pagePathophysiology of Angina PectorisCheryl100% (1)
- Pathophysiology Diagram of Congestive Heart FailureDocument3 pagesPathophysiology Diagram of Congestive Heart Failurea_samiane64% (11)
- Common Cause or Etiology of Pott's DiseaseDocument5 pagesCommon Cause or Etiology of Pott's DiseaseStan Aves GarciaNo ratings yet
- The Pathophysiology of Acute Coronary SyndromesDocument6 pagesThe Pathophysiology of Acute Coronary SyndromesTulisan NonaNo ratings yet
- BA Blok 4.1 Cardiovascular 2018Document97 pagesBA Blok 4.1 Cardiovascular 2018Ilga ZanNo ratings yet
- Cardiovascular Disorders SlidesDocument9 pagesCardiovascular Disorders SlidesCenonina Paola TapiaNo ratings yet
- 13 - Atherosclerotic Plaque HealingDocument12 pages13 - Atherosclerotic Plaque HealingAndres Felipe Hurtado BautistaNo ratings yet
- Ischemic Heart Disease NotesDocument8 pagesIschemic Heart Disease NotesDr. Tarush DhawanNo ratings yet
- Myocardial InfarctionDocument11 pagesMyocardial Infarctionkinzay batoolNo ratings yet
- Clinical Cardiology: New FrontiersDocument8 pagesClinical Cardiology: New Frontiersayuhati siregarNo ratings yet
- 2020 Atherosclerotic Plaque HealingDocument12 pages2020 Atherosclerotic Plaque HealingErnesto ArellanoNo ratings yet
- Macroscopic Diagnostic For Pathoanathomy and Cytopathology ExamDocument21 pagesMacroscopic Diagnostic For Pathoanathomy and Cytopathology Examkapil pancholiNo ratings yet
- Atherosclerosis: Yang Haibo MD Department of Cardiology, 1 Affiliated Hospital of ZzuDocument22 pagesAtherosclerosis: Yang Haibo MD Department of Cardiology, 1 Affiliated Hospital of Zzuapi-19916399No ratings yet
- Ralph LaurenDocument11 pagesRalph LaurenAnushkaNo ratings yet
- Integrated Review For MS 2023Document11 pagesIntegrated Review For MS 2023Joen SinamagNo ratings yet
- Sales Management Course Panda Sachdev 2259Document1,408 pagesSales Management Course Panda Sachdev 2259raachyeta_sharmaNo ratings yet
- Astorga V VillegasDocument7 pagesAstorga V Villegasamolyrehs24No ratings yet
- Activity Design Leaders Camp Final 1Document7 pagesActivity Design Leaders Camp Final 1Jayson B. OmañaNo ratings yet
- Chitral Brief Report 2020Document3 pagesChitral Brief Report 2020fahad tahirNo ratings yet
- Strategic Management Individual Assignment: Critique On Disney Goes OTT: The Launch That Defined A Major Industry ShiftDocument8 pagesStrategic Management Individual Assignment: Critique On Disney Goes OTT: The Launch That Defined A Major Industry ShiftShivam TiwariNo ratings yet
- Training MeasurLink 8Document17 pagesTraining MeasurLink 8Cristian RoblesNo ratings yet
- Nurul Ayu AlikaDocument16 pagesNurul Ayu AlikaNurul Ayu Alika SaharaNo ratings yet
- EXP-1000HD - 167-090gb-B-Manual-Exp-1000-Hd-EuDocument26 pagesEXP-1000HD - 167-090gb-B-Manual-Exp-1000-Hd-EuMihai Popa100% (1)
- EIL 6-79-0026 Insulation MaterialDocument70 pagesEIL 6-79-0026 Insulation MaterialSagarBabuNo ratings yet
- Pronunciation Bank: Activities: Unit 1Document1 pagePronunciation Bank: Activities: Unit 1Maria Da Conceição MoraisNo ratings yet
- Venturina NCPSDocument5 pagesVenturina NCPSYsabelle VenturinaNo ratings yet
- Flying High: Unit 5Document10 pagesFlying High: Unit 5ducNo ratings yet
- Reverberation Time Problems SolvedDocument23 pagesReverberation Time Problems SolvedVincent Emman SalibioNo ratings yet
- Sap PP ConfigurationDocument32 pagesSap PP ConfigurationcvalentNo ratings yet
- Coldplay - Every Teardrop Is A WaterfallDocument2 pagesColdplay - Every Teardrop Is A WaterfallfelipepaupitzNo ratings yet
- 93049v00 Simulink Real-Time Whitepaper PDFDocument6 pages93049v00 Simulink Real-Time Whitepaper PDFAnonymous gH8rfDNo ratings yet
- Gmma Relationships: by Daryl GuppyDocument5 pagesGmma Relationships: by Daryl GuppyMoed D'lhoxNo ratings yet
- Environmental Principles and ConceptsDocument7 pagesEnvironmental Principles and ConceptsMary Rose BaluranNo ratings yet
- Chennai Public School: English Class - ViiiDocument2 pagesChennai Public School: English Class - ViiiAbishek ThiyagarajanNo ratings yet
- MFCI Company Profile (English)Document26 pagesMFCI Company Profile (English)florencio morenomfciNo ratings yet