Professional Documents
Culture Documents
Nucleophilic Aromatic Substitution
Nucleophilic Aromatic Substitution
Uploaded by
Tairine PimentelOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Nucleophilic Aromatic Substitution
Nucleophilic Aromatic Substitution
Uploaded by
Tairine PimentelCopyright:
Available Formats
Supplementary
Topic
Nucleophilic Aromatic
Substitution
Although most reactions of aromatic compounds occur by way of electrophilic aromatic substitution, aryl halides undergo a limited number of substitution reactions with strong nucleophiles.
General reaction
Nu
Nu
X = F, Cl, Br, I
A = H or electron-withdrawing group
Nucleophilic aromatic substitution results in the substitution of a halogen X on a
benzene ring by a nucleophile (:Nu).
As we learned in Section 7.18, these reactions cannot occur by an SN1 or SN2 mechanism,
which take place only at sp3 hybridized carbons. Instead, two different mechanisms are proposed to explain the results: additionelimination (Section A.1) and eliminationaddition
(Section A.2).
A.1 Nucleophilic Aromatic Substitution
by AdditionElimination
Aryl halides with strong electron-withdrawing groups (such as NO2) on the ortho or para positions react with nucleophiles to afford substitution products. For example, treatment of p-chloronitrobenzene with hydroxide (OH) affords p-nitrophenol by replacement of Cl by OH.
O2N
Cl
p-chloronitrobenzene
OH
O2N
OH
Cl
p-nitrophenol
Nucleophilic aromatic substitution occurs with a variety of strong nucleophiles, including OH,
OR, NH2, SR, and in some cases, neutral nucleophiles such as NH3 and RNH2. The mechanism
of these reactions has two steps: addition of the nucleophile to form a resonance-stabilized carbanion, followed by elimination of the halogen leaving group. Mechanism A.1 is drawn with an
aryl chloride containing a general electron-withdrawing group W.
A-1
smi75624_topicA.indd A-1
6/1/10 12:09 PM
A-2
Topic A
Nucleophilic Aromatic Substitution
Mechanism A.1 Nucleophilic Aromatic Substitution by AdditionElimination
Step [1] Addition of the nucleophile (:Nu) to form a carbanion
Addition of the nucleophile (:Nu ) forms a
Nu
Nu
Cl
Cl
Nu
Cl
Nu
Cl
resonance-stabilized carbanion with a new
C Nu bondthree resonance structures
can be drawn.
Step [1] is rate-determining since the
resonance-stabilized carbanion
aromaticity of the benzene ring is lost.
Step [2] Loss of the leaving group to re-form the aromatic ring
Nu
Cl
In Step [2], loss of the leaving group re-forms
Nu
the aromatic ring. This step is fast because the
aromaticity of the benzene ring is restored.
Cl
In nucleophilic aromatic substitution, the following trends in reactivity are observed.
Increasing the number of electron-withdrawing groups increases the reactivity of the
aryl halide. Electron-withdrawing groups stabilize the intermediate carbanion, and by
the Hammond postulate, lower the energy of the transition state that forms it.
Increasing the electronegativity of the halogen increases the reactivity of the aryl
halide. A more electronegative halogen stabilizes the intermediate carbanion by an
inductive effect, making aryl fluorides (ArF) much more reactive than other aryl halides,
which contain less electronegative halogens.
Thus, aryl chloride B is more reactive than o-chloronitrobenzene (A) because it contains two
electron-withdrawing NO2 groups. Aryl fluoride C is more reactive than B because C contains
the more electronegative halogen, fluorine.
Cl
O2N
Cl
NO2
A
O2N
NO2
B
NO2
C
Increasing reactivity
Note, too, that the location of the electron-withdrawing group greatly affects the rate of
nucleophilic aromatic substitution. When a nitro group is located ortho or para to the halogen,
the negative charge of the intermediate carbanion can be delocalized onto the NO2 group, thus
stabilizing it. With a meta NO2 group, no such additional delocalization onto the NO2 group
occurs.
smi75624_topicA.indd A-2
6/1/10 12:09 PM
A-3
A.1 Nucleophilic Aromatic Substitution by AdditionElimination
Para NO2 group
Nu
Nu
Cl
Cl
O2N
Nu
Cl
Nu
Cl
O
O2N
N
O
O2N
Nu
Cl
additional resonance stabilization
The negative charge is delocalized
on the O atom of the NO2 group.
Meta NO2 group
Nu
Nu
Cl
Cl
Nu
Cl
NO2
Nu
Cl
NO2
NO2
NO2
The negative charge is never delocalized on the NO2 group.
Thus, nucleophilic aromatic substitution by an additionelimination mechanism occurs only
with aryl halides that contain electron-withdrawing substituents at the ortho or para position.
Problem A.1
Draw the products of each reaction.
O2N
NaOCH3
a.
c. F
OH
Cl
NO2
b.
NO2
NaSCH2CH3
d. O2N
CH3NH2
NO2
NO2
Problem A.2
Cl
Rank the aryl halides in each group in order of increasing reactivity in nucleophilic aromatic
substitution by an additionelimination mechanism.
a. chlorobenzene, p-fluoronitrobenzene, m-fluoronitrobenzene
b. 1-fluoro-2,4-dinitrobenzene, 1-fluoro-3,5-dinitrobenzene, 1-fluoro-3,4-dinitrobenzene
c. 1-fluoro-2,4-dinitrobenzene, 4-chloro-3-nitrotoluene, 4-fluoro-3-nitrotoluene
Problem A.3
Explain why a methoxy group (CH3O) increases the rate of electrophilic aromatic substitution, but
decreases the rate of nucleophilic aromatic substitution.
Problem A.4
Draw a stepwise mechanism for the following reaction that forms ether D. D can be converted to
the antidepressant fluoxetine (trade name Prozac) in a single step.
N(CH3)2
F3C
KOC(CH3)3
+
Cl
N(CH3)2
F3C
one step
HO
O
D
smi75624_topicA.indd A-3
NHCH3
F3C
O
fluoxetine
6/1/10 12:09 PM
A-4
Topic A
Nucleophilic Aromatic Substitution
A.2 Nucleophilic Aromatic Substitution
by EliminationAddition: Benzyne
Aryl halides that do not contain an electron-withdrawing group generally do not react with
nucleophiles. Under extreme reaction conditions, however, nucleophilic aromatic substitution
can occur with aryl halides. For example, heating chlorobenzene with NaOH above 300 C and
170 atmospheres of pressure affords phenol.
Cl
[1] NaOH, 300 C, 170 atm
[2] H3O+
OH
chlorobenzene
NaCl
phenol
The mechanism proposed to explain this result involves formation of a benzyne intermediate
(C6H4) by eliminationaddition. As shown in Mechanism A.2, benzyne is a highly reactive,
unstable intermediate formed by elimination of HX from an aryl halide.
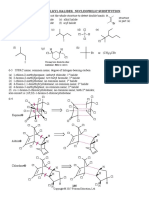
Mechanism A.2 Nucleophilic Aromatic Substitution by EliminationAddition: Benzyne
Part [1] Elimination of HX to form benzyne
Cl
OH
Cl
[1]
[2]
Elimination of H and X from two adjacent carbons forms a
reactive benzyne intermediate (Steps [1] and [2]).
H2O
benzyne
+ Cl
Part [2] Nucleophilic addition to form the substitution product
OH
OH
[3]
OH
[4]
Addition of the nucleophile (OH in this case) and
H OH
protonation form the substitution product (Steps [3] and [4]).
OH
Formation of a benzyne intermediate explains why substituted aryl halides form mixtures of
products. Nucleophilic aromatic substitution by an eliminationaddition mechanism affords
substitution on the carbon directly bonded to the leaving group and the carbon adjacent to
it. As an example, treatment of p-chlorotoluene with NaNH2 forms para- and meta-substitution
products.
CH3
CH3
NaNH2
NH3
Cl
p-chlorotoluene
CH3
NH2
NH2
p-methylaniline
(p-toluidine)
m-methylaniline
(m-toluidine)
This result is explained by the fact that nucleophilic attack on the benzyne intermediate may
occur at either C3 to form m-methylaniline, or C4 to form p-methylaniline.
smi75624_topicA.indd A-4
6/1/10 12:09 PM
A.2
A-5
Nucleophilic Aromatic Substitution by EliminationAddition: Benzyne
CH3
CH3
CH3
CH3
NH2
NaNH2
NH3
nucleophilic
attack at C3
NH2
two steps
NH2
C3
Cl
p-chlorotoluene
or
m-methylaniline
CH3
CH3
CH3
NH3
NH2
nucleophilic
attack at C4
H
NH2
C4
NH2
p-methylaniline
As you might expect, the triple bond in benzyne is unusual. Each carbon of the six-membered
ring is sp2 hybridized, and as a result, the bond and two bonds of the triple bond are formed
with the following orbitals.
The bond is formed by overlap of two sp2 hybrid orbitals.
One bond is formed by overlap of two p orbitals perpendicular to the plane of the
molecule.
The second bond is formed by overlap of two sp2 hybrid orbitals.
H
sp 2
hybridized
H
Overlap of two sp 2 hybrid
orbitals forms a bond.
Thus, the second bond of benzyne differs from all other bonds seen thus far, because it is
formed by the side-by-side overlap of sp2 hybrid orbitals, not p orbitals. This bond, located
in the plane of the molecule, is extremely weak.
Problem A.5
Draw the products of each reaction.
a.
Cl
NaNH2
NH3
b. CH3O
Cl
NaOH
H2O
c.
Cl
KNH2
NH3
Problem A.6
Draw all products formed when m-chlorotoluene is treated with KNH2 in NH3.
Problem A.7
Explain why 2-chloro-1,3-dimethylbenzene is inert to nucleophilic aromatic substitution by way of
an eliminationaddition mechanism.
Problem A.8
Draw a stepwise mechanism for the following reaction.
NHCH3
Cl
strong base
N
CH3
smi75624_topicA.indd A-5
6/1/10 12:09 PM
You might also like
- Alkyl Halide QuestionsDocument5 pagesAlkyl Halide QuestionsArmelle Keisha MendozaNo ratings yet
- 1H NMR Problem SetDocument5 pages1H NMR Problem Setfishfeeder1No ratings yet
- SET-NET Pericyclic ReactionsDocument61 pagesSET-NET Pericyclic ReactionsBapu ThoratNo ratings yet
- Amines QuestionsDocument5 pagesAmines Questionsamal gainNo ratings yet
- Aldehydes and KetonesDocument9 pagesAldehydes and KetonesCamille AdleNo ratings yet
- Organic Chemistry Nucleophilic SubstitutDocument1 pageOrganic Chemistry Nucleophilic Substitut027 กัญญาภรณ์ ตันกลางNo ratings yet
- Organic Chemistry Experiment 8Document3 pagesOrganic Chemistry Experiment 8Rogeni Misaelle PetinglayNo ratings yet
- 12B Alcohol 2Document11 pages12B Alcohol 2Kasun RatnayakeNo ratings yet
- Homework 3Document13 pagesHomework 3polypeptideNo ratings yet
- Chapter 10 PDFDocument10 pagesChapter 10 PDFKelsi Kyla PeraltaNo ratings yet
- Introduction of Organic Chemistry by Eyes of Ajnish Kumar Gupta (AKG)Document24 pagesIntroduction of Organic Chemistry by Eyes of Ajnish Kumar Gupta (AKG)ajju_208180% (5)
- OrganicChemistryChapter5 PDFDocument19 pagesOrganicChemistryChapter5 PDFJuliet Tatiana CumbeNo ratings yet
- Hybridization 1Document37 pagesHybridization 1Anzari MuhammadNo ratings yet
- Challenge Problems in David Klein Chap 7-17Document32 pagesChallenge Problems in David Klein Chap 7-17Ling LingNo ratings yet
- Reaction Maps v1.0 - LinksDocument15 pagesReaction Maps v1.0 - LinksCATALINA FERNANDA JARAMILLO ARAYANo ratings yet
- Huckel Rule of Aromaticity 2 PDFDocument25 pagesHuckel Rule of Aromaticity 2 PDFUmar Farooq100% (1)
- Benzopinacol PhotochemistryDocument3 pagesBenzopinacol PhotochemistryJulius Victorius Aragon SaluriaNo ratings yet
- Aldol Notes PDFDocument8 pagesAldol Notes PDFAna100% (1)
- Paterno Buchi ReactionDocument35 pagesPaterno Buchi ReactionHarish Chopra100% (1)
- Aldol CondensationDocument71 pagesAldol CondensationJeyanthiNo ratings yet
- Aromaticity NotesDocument6 pagesAromaticity NotesSLIMBOI OpNo ratings yet
- Reviewer AnachemDocument13 pagesReviewer AnachemAurora CelesteNo ratings yet
- IB Chemistry Notes - Stoichiometry and The Mole ConceptDocument1 pageIB Chemistry Notes - Stoichiometry and The Mole ConceptKefin TajebNo ratings yet
- CH 6 Answers (All) PDFDocument29 pagesCH 6 Answers (All) PDFAhmed SideegNo ratings yet
- Hückel's MO Treatment of BenzeneDocument12 pagesHückel's MO Treatment of BenzeneRichard Allen0% (1)
- CHM145L - Exp 1 - Melting Point and Boiling Point of Organic CompoundsDocument6 pagesCHM145L - Exp 1 - Melting Point and Boiling Point of Organic CompoundsBiancaTardecillaNo ratings yet
- Pinacol Pinacolone RearrangementDocument13 pagesPinacol Pinacolone RearrangementsachindhodreNo ratings yet
- Elimination Reactions Mechanism Lecture NotesDocument17 pagesElimination Reactions Mechanism Lecture NotesveluselvamaniNo ratings yet
- Aromaticity NotesDocument10 pagesAromaticity NotesVirendra Singh Rajput100% (1)
- Qoi0809t1 ConfDocument13 pagesQoi0809t1 ConfTahirat NasiruNo ratings yet
- Unit 2 Organic ChemistryDocument18 pagesUnit 2 Organic Chemistrydeepashashikumar10100% (1)
- Course Title: Organic Chemistry-I Course Code: Chm-553, Chm-507 Semester: MSC 1, Bs 5Document18 pagesCourse Title: Organic Chemistry-I Course Code: Chm-553, Chm-507 Semester: MSC 1, Bs 5Mian Naveed AhmedNo ratings yet
- Alkenes and AlkynesDocument2 pagesAlkenes and AlkynesLewis AlfonsoNo ratings yet
- Pinacol RearrangementDocument2 pagesPinacol RearrangementkarinadegomaNo ratings yet
- Alkyl HalidesDocument15 pagesAlkyl Halidesutkarsh jain100% (4)
- Recrystallization in Organic Chemistry LabDocument5 pagesRecrystallization in Organic Chemistry Labrc865No ratings yet
- 19 Enolates EnaminesDocument59 pages19 Enolates EnaminesTwas Anassin100% (2)
- Organic Exam Answer.Document11 pagesOrganic Exam Answer.S JNo ratings yet
- Organic Chemistry ExperimentDocument7 pagesOrganic Chemistry ExperimentKayla RhodesNo ratings yet
- Module 2.3 NGP PDFDocument3 pagesModule 2.3 NGP PDFIshaan ChaturvediNo ratings yet
- Aromaticity CompleteDocument104 pagesAromaticity Completewahidalwahdi100% (1)
- Aromaticity With Huckle's RuleDocument7 pagesAromaticity With Huckle's RuleSk ZNo ratings yet
- Mass Spectroscopy Part 2 Lecture4 Solved ProblemsDocument8 pagesMass Spectroscopy Part 2 Lecture4 Solved ProblemsMvita HenocNo ratings yet
- Sigmatropic Rearrangement ReactionDocument18 pagesSigmatropic Rearrangement ReactionSuman ChauhanNo ratings yet
- Alkyl Halide and Aryl HalideDocument68 pagesAlkyl Halide and Aryl HalideAniruddha KawadeNo ratings yet
- Practice Questions-Conformational AnalysisDocument4 pagesPractice Questions-Conformational AnalysisHarry Zgambo100% (1)
- Huckel Theory For Conjugated Systems: CH 105: Organic ChemistryDocument72 pagesHuckel Theory For Conjugated Systems: CH 105: Organic ChemistryRaunaq Bhirangi100% (1)
- Chemistry of Aromatics CompoundsDocument16 pagesChemistry of Aromatics CompoundsOfudje Edwin AndrewNo ratings yet
- Intro To Organic Reactions CHM457Document73 pagesIntro To Organic Reactions CHM457Zafrel ZaffNo ratings yet
- Carboranes AgapinDocument17 pagesCarboranes AgapinJulienne Stephanie Fabie100% (1)
- Difficult Questions On Organic ChemistryDocument5 pagesDifficult Questions On Organic Chemistrytarunbirbanga100% (1)
- Comparing The SN1 and SN2 Reactions - Master Organic ChemistryDocument5 pagesComparing The SN1 and SN2 Reactions - Master Organic Chemistryprince ranaNo ratings yet
- Essential Organic Chemistry: Aromaticity: Reactions of Benzene and Substituted BenzenesDocument75 pagesEssential Organic Chemistry: Aromaticity: Reactions of Benzene and Substituted BenzeneschurvaloooNo ratings yet
- Aromatic Anti Aromatic Non AromaticDocument2 pagesAromatic Anti Aromatic Non AromaticRitesh SonawaneNo ratings yet
- Lecture Notes Chapter-12-Aldehydes, Ketones & Carboxylic AcidsDocument26 pagesLecture Notes Chapter-12-Aldehydes, Ketones & Carboxylic AcidsSHUBHAMNo ratings yet
- Nucleophlic Aromatic SubstitutionDocument10 pagesNucleophlic Aromatic SubstitutionHalala HajiNo ratings yet
- Electrophilic Aromatic Substitution + The Chemistry of BenzeneDocument28 pagesElectrophilic Aromatic Substitution + The Chemistry of BenzeneTrescia Mae EstilloreNo ratings yet
- Benzene and Aromatic Compounds-fazli-In ClassDocument57 pagesBenzene and Aromatic Compounds-fazli-In Classjokowi123No ratings yet
- Electrophilic Substitution ReactionDocument33 pagesElectrophilic Substitution ReactionPraveen Kumar SNo ratings yet
- Code - #Include Using Namespace STD Int Main (Int Arr (95) Int Arrno (95) Int I, J Int Even 0, Odd 0 For (Int I 0 I 95 I++) (Document4 pagesCode - #Include Using Namespace STD Int Main (Int Arr (95) Int Arrno (95) Int I, J Int Even 0, Odd 0 For (Int I 0 I 95 I++) (njwaghmare7392No ratings yet
- Atomic Structure Type 1Document20 pagesAtomic Structure Type 1Sudhakar ChollangiNo ratings yet
- Leave RulesDocument1 pageLeave Rulesnjwaghmare7392No ratings yet
- Access Control SystemDocument5 pagesAccess Control Systemnjwaghmare7392No ratings yet
- Access Control SystemDocument5 pagesAccess Control Systemnjwaghmare7392No ratings yet
- Synthetic Strategies in ChemistryDocument361 pagesSynthetic Strategies in ChemistrynadjibwassimNo ratings yet
- Aryl Halides As Precursors of Electrogenerated Bases. Utilization in Coupling Reactions of Acetonitriie With Various Electrophilic CompoundsDocument8 pagesAryl Halides As Precursors of Electrogenerated Bases. Utilization in Coupling Reactions of Acetonitriie With Various Electrophilic CompoundsWalid Ebid ElgammalNo ratings yet
- Halides (Q.B) AdvancedDocument26 pagesHalides (Q.B) AdvancedRaj ModiNo ratings yet
- Alkyl Halides LectureDocument16 pagesAlkyl Halides LectureKoki KingNo ratings yet
- 14L3Document30 pages14L3Hafizszulfeyzul FeyzulNo ratings yet
- Organic Chemistry Sn2 Sn1 E2 E1Document4 pagesOrganic Chemistry Sn2 Sn1 E2 E1vishve2015No ratings yet
- Organic Chemistry Crash Reviewer PDFDocument11 pagesOrganic Chemistry Crash Reviewer PDFmaganda akoNo ratings yet
- Haloalkanes and Haloarenes-Imp QNSDocument3 pagesHaloalkanes and Haloarenes-Imp QNSjamesNo ratings yet
- Organic Chemistry For Chemical EngineersDocument13 pagesOrganic Chemistry For Chemical EngineersavixNo ratings yet
- HSC Chemistry II Board Paper 2013Document2 pagesHSC Chemistry II Board Paper 2013Vaibhav GaikwadNo ratings yet
- Chemistry: NEET - 2020-21Document1 pageChemistry: NEET - 2020-21pratikNo ratings yet
- Chemistry Vol.2 Figures Class 12Document60 pagesChemistry Vol.2 Figures Class 12Sasidaran SasiNo ratings yet
- GOC Neet Key NotesDocument40 pagesGOC Neet Key Notesiampriyatiwarii890No ratings yet
- Aldehyde and KetonesDocument45 pagesAldehyde and KetonesShivam GuptaNo ratings yet
- Chem 331Document11 pagesChem 331Satya KamNo ratings yet
- An Extended Model of The S 2 Reaction Between A Substituted Alkane and NucleophileDocument10 pagesAn Extended Model of The S 2 Reaction Between A Substituted Alkane and NucleophileSubhabrata MabhaiNo ratings yet
- Reaksi Substitusi Dan EliminasiDocument28 pagesReaksi Substitusi Dan Eliminasianita hasyimNo ratings yet
- Chapter 6-Alkyl Halides Nucleophilic Substitution: You May Have Drawn The Other Enantiomer. Either Is CorrectDocument23 pagesChapter 6-Alkyl Halides Nucleophilic Substitution: You May Have Drawn The Other Enantiomer. Either Is Correct張湧浩No ratings yet
- Aromatic Amines As Nucleophiles in The Bargellini Reaction PDFDocument4 pagesAromatic Amines As Nucleophiles in The Bargellini Reaction PDFdavidNo ratings yet
- CBQ Haloalkanes and HaloarenesDocument20 pagesCBQ Haloalkanes and HaloarenesAnish dekaNo ratings yet
- Organic ChemDocument116 pagesOrganic ChemNidhi Sisodia100% (1)
- Reaction Mechanism DDocument36 pagesReaction Mechanism Dprajaktac506No ratings yet
- 11 Chemistry PP ch12 Organic Chemistry Principle TechniquesDocument5 pages11 Chemistry PP ch12 Organic Chemistry Principle TechniquesSwayamNo ratings yet
- Chem BookDocument34 pagesChem BookmuskanNo ratings yet
- 24 Organic Review 2023Document37 pages24 Organic Review 2023Elyssa En YingNo ratings yet
- Chapter 12 Organic Chemistry Some Basic PrinciplesDocument29 pagesChapter 12 Organic Chemistry Some Basic PrinciplesYash PlayNo ratings yet
- Comparing The SN1 and SN2 Reactions - Master Organic ChemistryDocument5 pagesComparing The SN1 and SN2 Reactions - Master Organic Chemistryprince ranaNo ratings yet
- GOC AllenDocument33 pagesGOC AllenAshish Ranjan100% (5)
- Carboxylic AcidsDocument3 pagesCarboxylic AcidsDian Agus SetyawatiNo ratings yet
- Alkyl HalidesDocument87 pagesAlkyl HalidesDidi LeraNo ratings yet