Professional Documents
Culture Documents
Entropy and The Ideal Gas
Entropy and The Ideal Gas
Uploaded by
Reuel Rocha0 ratings0% found this document useful (0 votes)
4 views1 pageEntropy and the Ideal Gas
Original Title
Entropy and the Ideal Gas
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentEntropy and the Ideal Gas
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
4 views1 pageEntropy and The Ideal Gas
Entropy and The Ideal Gas
Uploaded by
Reuel RochaEntropy and the Ideal Gas
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 1
Entropy and the Ideal Gas
Ideal gas:
(U
V )T = 0
dU = CV dT
for all processes
dU = dq - P dV
T or V
dq = dU + P dV
dqrev
dT nR
dS = T = CV T + V dV
at constant V
dT
S = CV T
T2
S = CV ln T
1
assume Cv = cst
at constant T
V2
nR
S = V dV = nR ln V
1
dH = dU + PdV + VdP = dqrev PdV + PdV + VdP
dqrev = dH VdP = CP dT VdP
dT nR
dS = CP
dP
T P
at constant P
T2
dT
S = CP T = CP ln T
assume CP = cst
T2
P
S = CP ln T - nR ln 2
P1
1
3
Example: 1 mole ideal monatomic gas: CV = 2 R = 12.5 J/K
double T at cst.V, S = 12.5 J/K ln 2 = 8.6 J/K
double T at cst P , S = 20.8 J/K ln 2 = 14.4 J/K
double V at cst T , S = 8.314 J/K ln 2 = 5.8 J/K
Colby College
Pext = P
You might also like
- Omron PLC Beginner GuideDocument254 pagesOmron PLC Beginner GuideTrịnh Quang100% (6)
- Enthalpy at IsentropicDocument98 pagesEnthalpy at IsentropicAnonymous xmSWrWbUKGNo ratings yet
- Solution Manual for an Introduction to Equilibrium ThermodynamicsFrom EverandSolution Manual for an Introduction to Equilibrium ThermodynamicsNo ratings yet
- Chap 02Document32 pagesChap 02echelon12No ratings yet
- Formula For Engineering Thermodynamics I 254231 Semester 1 2550 Concept andDocument1 pageFormula For Engineering Thermodynamics I 254231 Semester 1 2550 Concept andKenneth MayorNo ratings yet
- Paper Thermo Mechanical EngineeringDocument14 pagesPaper Thermo Mechanical EngineeringAdif HerawanNo ratings yet
- Basic Thermodynamic Processes of Ideal GasDocument6 pagesBasic Thermodynamic Processes of Ideal GasThanh Son100% (1)
- Volumetric Properties of Pure FluidsDocument21 pagesVolumetric Properties of Pure FluidsIR Ika EtyEtyka Dora100% (1)
- PLCBeginnerguide110 177Document68 pagesPLCBeginnerguide110 177Jun CiaoNo ratings yet
- Ideal Gas FormulasDocument2 pagesIdeal Gas FormulasbythekiloNo ratings yet
- Extra Information For Chem 340 Exams:: Transport Temperature (T), Activity/concentration (A) W RT JDocument4 pagesExtra Information For Chem 340 Exams:: Transport Temperature (T), Activity/concentration (A) W RT JZevano C. SibaraniNo ratings yet
- Clase 1 PDFDocument34 pagesClase 1 PDFMafe ArizaNo ratings yet
- Che 254 (Midsem)Document6 pagesChe 254 (Midsem)obumaradonaNo ratings yet
- Volumetric PropertiesDocument36 pagesVolumetric PropertiesRohan BhilkarNo ratings yet
- Thermodynamic Relations PDFDocument10 pagesThermodynamic Relations PDFAnnshun KiatNo ratings yet
- September 25, 2001 Reading: Chapter V Homework: 5.2, 5.4, 5.6, 5.8, 5.10, 5.11, 5.12. More Examples of Use of Maxwell's EquationsDocument5 pagesSeptember 25, 2001 Reading: Chapter V Homework: 5.2, 5.4, 5.6, 5.8, 5.10, 5.11, 5.12. More Examples of Use of Maxwell's EquationsclaudioNo ratings yet
- Uses of Maxwell RelationsDocument17 pagesUses of Maxwell RelationsArun EbenezerNo ratings yet
- ThermodynamicsDocument9 pagesThermodynamicssamir boseNo ratings yet
- September 6, 2001 Reading: Chapter Three Homework: 3.1,3.2,3.4,3.5,3.6Document3 pagesSeptember 6, 2001 Reading: Chapter Three Homework: 3.1,3.2,3.4,3.5,3.6claudioNo ratings yet
- Part 3 - Energy Balance Involving Gases PDFDocument15 pagesPart 3 - Energy Balance Involving Gases PDFHarold SumagaysayNo ratings yet
- DH Du+V DP+PDV Du+V DP H V PDocument4 pagesDH Du+V DP+PDV Du+V DP H V Pbrando1819No ratings yet
- FORMULASDocument26 pagesFORMULASRaymart LaysonNo ratings yet
- Exam Thermo Part1!11!12 2020 FinalDocument11 pagesExam Thermo Part1!11!12 2020 FinalMaarten ElingNo ratings yet
- Thermodynamics CompleteDocument43 pagesThermodynamics Completesutarohit2006No ratings yet
- Adiabatic ChangeDocument2 pagesAdiabatic ChangeYau Ching KoonNo ratings yet
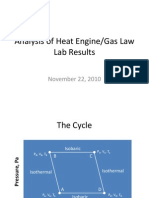
- Analysis of Heat Engine/Gas Law Lab Results: November 22, 2010Document12 pagesAnalysis of Heat Engine/Gas Law Lab Results: November 22, 2010abeast34No ratings yet
- 4862 Solutionsxin PDFDocument37 pages4862 Solutionsxin PDFይቴ ስንሻዉNo ratings yet
- ME 331 Thermodynamics II Lecture 3cDocument31 pagesME 331 Thermodynamics II Lecture 3cJosell CaipangNo ratings yet
- Helpful Eqns ME311 F14 PJFDocument2 pagesHelpful Eqns ME311 F14 PJFtarzantrapNo ratings yet
- Mit3 PDFDocument2 pagesMit3 PDFrosendo rojas barraganNo ratings yet
- TD MODULE 4Document12 pagesTD MODULE 4mujeebNo ratings yet
- Formula Sheet Midterm 1Document1 pageFormula Sheet Midterm 1qwqeNo ratings yet
- PC 1 Formulae Sheet 1Document2 pagesPC 1 Formulae Sheet 1Kaaya GodfreyNo ratings yet
- Solution: 1. Refer Eq. (6.28)Document4 pagesSolution: 1. Refer Eq. (6.28)kajal mishrsNo ratings yet
- PHYS 310: Thermodynamics and Statistical Mechanics Final Exam Formula SheetDocument2 pagesPHYS 310: Thermodynamics and Statistical Mechanics Final Exam Formula SheetChristopher ThaiNo ratings yet
- Problem Solving Tips ThermodynamicsDocument5 pagesProblem Solving Tips ThermodynamicsJose FabianNo ratings yet
- Chapter 3 - Section B - Non-Numerical SolutionsDocument10 pagesChapter 3 - Section B - Non-Numerical SolutionsFaris NaufalNo ratings yet
- Chapter 3 - Section B - Non-Numerical SolutionsDocument12 pagesChapter 3 - Section B - Non-Numerical Solutionslight2618No ratings yet
- FORMULAS XNXNDocument23 pagesFORMULAS XNXNRaymart Layson0% (1)
- 1st Law Worked ExamplesDocument4 pages1st Law Worked ExamplesMahir MahmoodNo ratings yet
- Process Calculations For Ideal GasDocument2 pagesProcess Calculations For Ideal GastwiddleapNo ratings yet
- FoundationsDocument1 pageFoundations이소연[ 학부재학 / 화공생명공학과 ]No ratings yet
- 2022 - Chem 7023 - LectureNotes - 0826-1Document7 pages2022 - Chem 7023 - LectureNotes - 0826-1Uche PNo ratings yet
- Termodinamika, Entropy, Dan Energi Dalam 2014Document23 pagesTermodinamika, Entropy, Dan Energi Dalam 2014Deriandra MuhyiddinNo ratings yet
- Chem 132 Cheat Sheet Exam 1-04Document1 pageChem 132 Cheat Sheet Exam 1-04Alvin Tung Kwong ChoongNo ratings yet
- Γ = Dan Cp = Cv +: R.T M - V Dt T R Mcv Dv VDocument2 pagesΓ = Dan Cp = Cv +: R.T M - V Dt T R Mcv Dv VHiLda HayatiiNo ratings yet
- Sistema Cerrado Gas Ideal: V P P V P VDocument2 pagesSistema Cerrado Gas Ideal: V P P V P VSaul OsmarNo ratings yet
- Engine Cycles: - Thermodynamics SupplementDocument23 pagesEngine Cycles: - Thermodynamics SupplementYoonjin HwangNo ratings yet
- Formulas For Thermo 2 QuizDocument1 pageFormulas For Thermo 2 Quizzvikush1No ratings yet
- 2023 CIR310 Sem Test 1 With AnswersDocument7 pages2023 CIR310 Sem Test 1 With Answersu21589969No ratings yet
- 5.60 Thermodynamics & Kinetics: Mit OpencoursewareDocument8 pages5.60 Thermodynamics & Kinetics: Mit OpencoursewarecaptainhassNo ratings yet
- RelationsDocument2 pagesRelationsAditya ShanmukhaNo ratings yet
- Fiqui ParcialDocument7 pagesFiqui ParcialCarlosCristobalNo ratings yet
- Lecture 05 - Chapter 2 - First LawDocument14 pagesLecture 05 - Chapter 2 - First LawHyeon Chang NoNo ratings yet
- Thermal Summary SheetDocument2 pagesThermal Summary SheetAhsan Habib TanimNo ratings yet
- Quiz RemedialDocument1 pageQuiz RemedialkargovilNo ratings yet
- Unit 2 First Law-Closed System ProblemsDocument11 pagesUnit 2 First Law-Closed System Problemspiravi66No ratings yet
- Quiz 1 - MA SolutionsDocument6 pagesQuiz 1 - MA SolutionsabullaNo ratings yet
- Lesson Plan 7Document13 pagesLesson Plan 7khandaker raiyanNo ratings yet
- Constants, Data, Definitions Diffusion and Heat Conduction Physics 213 Formula SheetDocument1 pageConstants, Data, Definitions Diffusion and Heat Conduction Physics 213 Formula SheetBenjamin ChiangNo ratings yet
- Chapter 3Document6 pagesChapter 3Joshua EspirituNo ratings yet
- ADAM, Intelligent Integrated Self-Enhanced Photovoltaic Panel With Rainwater Harvesting For Irrigation, Unit Cooling and CleaningDocument4 pagesADAM, Intelligent Integrated Self-Enhanced Photovoltaic Panel With Rainwater Harvesting For Irrigation, Unit Cooling and CleaningJun CiaoNo ratings yet
- Session Layer: Osi ModelDocument11 pagesSession Layer: Osi ModelJun CiaoNo ratings yet
- SolucionarioDocument16 pagesSolucionariopedro75223No ratings yet
- SolucionarioDocument16 pagesSolucionariopedro75223No ratings yet