Professional Documents
Culture Documents
4.4 Air Standard Otto Cycle
4.4 Air Standard Otto Cycle
Uploaded by
Rahul PundoraOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
4.4 Air Standard Otto Cycle
4.4 Air Standard Otto Cycle
Uploaded by
Rahul PundoraCopyright:
Available Formats
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
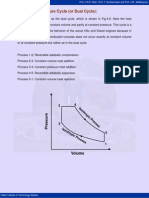
4.4 Air Standard Otto Cycle:
The air-standard-Otto cycle is the idealized cycle for the spark-ignition internal
combustion engines. This cycle is shown above on p-v and T-s diagrams. The Otto
cycle 1-2-3-4 consists of following four process:
Process 1-2: Reversible adiabatic compression of air.
Process 2-3: Heat addition at constant volume.
Process 3-4: Reversible adiabatic expansion of air.
Process 4-1: Heat rejection at constant volume.
2
4
Volume
Indian Institute of Technology Madras
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
2
4
1
Entropy
Fig.4.4. Otto cycle on p-v and T-s diagrams
Air Standard Efficiency:
th =
Net workdone
Net heat added
Since processes 1-2 and 3-4 are adiabatic processes, the heat transfer during the cycle
takes place only during processes 2-3 and 4-1 respectively. Therefore, thermal
efficiency can be written as,
th =
Heat added - Heat rejected
Heat added
Consider m kg of working fluid,
Heat added = mCV ( T3 - T2 )
Heat Rejected = mCV ( T4 - T1 )
th =
Indian Institute of Technology Madras
mCV ( T3 - T2 ) - mCV ( T4 - T1 )
mCV ( T3 - T2 )
=1-
T4 - T1
T3 - T2
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
For the reversible adiabatic processes 3-4 and 1-2, we can write,
v
T4
= 3
T3
v4
-1
V
T
and 1 = 2
T2
V1
-1
v 2 = v3 and v 4 = v1
V
T4
T
T -T
= 1 = 4 1 = 2
T3
T2
T3 - T2
V1
th
The ratio
V
T1
= 1- 2
= 1 T2
V1
-1
V1
is called as compression ratio, r.
V2
th
1
=1-
r
-1
From the above equation, it can be observed that the efficiency of the Otto cycle is
mainly the function of compression ratio for the given ratio of Cp and Cv. If we plot the
variations of the thermal efficiency with increase in compression ratio for different
gases, the curves are obtained as shown in Fig.4.4.1. Beyond certain values of
compression ratios, the increase in the thermal efficiency is very small, because the
curve tends to be asymptotic. However, practically the compression ratio of petrol
engines is restricted to maximum of 9 or 10 due to the phenomenon of knocking at high
compression ratios.
Indian Institute of Technology Madras
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
=1.67
=1.40
=1.30
Compression ratio,r
Effect of CR and on efficiency for Otto cycle.
Fig.4.4.1. Variation of thermal efficiency with compression ratio
Mean Effective Pressure:
Generally, it is defined as the ratio of the net workdone to the displacement volume of
the piston.
Let us consider m kg of working substance.
Net work done = m Cv {( T3 - T2 ) - ( T4 - T1 )}
Displacement Volume =
( V1
- V2 )
m R T1 r - 1
1
= V1 1 - =
r
P1
since, R = Cv ( - 1)
Indian Institute of Technology Madras
m C v ( - 1) T1 r - 1
P1
r
Gas Power Cycles
Prof. U.S.P. Shet , Prof. T. Sundararajan and Prof. J.M . Mallikarjuna
mep =
m C v ( T3 - T2 )
m C v ( -1) T1
P1
1 p1 r
- 1 T1 r - 1
Now,
T2 = T1 ( r )
Let,
rp =
T4
- T2 ) - ( T4 - T1
-1
P3
T2 = rp T2 = rp r -1 T1
P2
1
= T3
r
mep =
-1
= rp r
-1
1
T1
r
P1 r
( r - 1) ( - 1)
{( r
(for V = C)
-1
= rp T1
r -1 - r -1
) - ( rp - 1)}
) (
)(
r -1 r - 1 - r - 1
p
p
= P1 r
( - 1) ( r - 1)
r -1 - 1 r - 1
p
mep = P1 r
( r - 1) ( - 1)
Indian Institute of Technology Madras
)}
P3 T3
=
= Pressure ratio
P2 T2
T3 =
So,
{( T3
- ( T4 - T1 )
r - 1
You might also like
- Fuel Injection & TurbochargingDocument4 pagesFuel Injection & Turbocharging6 avinashNo ratings yet
- Wind Tunnel InroductionDocument38 pagesWind Tunnel Inroductionmich48chinNo ratings yet
- Forces and MomentsDocument13 pagesForces and Momentsanon_287164994No ratings yet
- Shrinkage Cracking in Restrained Reinforced Concrete MembersDocument93 pagesShrinkage Cracking in Restrained Reinforced Concrete MembersPeter MsomaNo ratings yet
- Aircraft PropulsionDocument158 pagesAircraft Propulsionbiswajit prustyNo ratings yet
- Design of Plate GirderDocument85 pagesDesign of Plate Girdersamirbendre1100% (2)
- Unmanned Aircraft SystemsFrom EverandUnmanned Aircraft SystemsElla AtkinsNo ratings yet
- Aerospace Actuators V3: European Commercial Aircraft and Tiltrotor AircraftFrom EverandAerospace Actuators V3: European Commercial Aircraft and Tiltrotor AircraftNo ratings yet
- Lift Line TheoryDocument19 pagesLift Line TheoryMuzakkir Sharieff100% (1)
- Questions For Gas Turbine EngineDocument120 pagesQuestions For Gas Turbine EngineJay Ganesh Murale DaranNo ratings yet
- Aircraft Design QuestionsDocument2 pagesAircraft Design QuestionsVijay Gorfad100% (2)
- AER 504 Aerodynamics: Ryerson University Department of Aerospace EngineeringDocument8 pagesAER 504 Aerodynamics: Ryerson University Department of Aerospace EngineeringbatmanbittuNo ratings yet
- Design of Grit & Sag Rods As Per Is CodeDocument3 pagesDesign of Grit & Sag Rods As Per Is CodeDarsHan MoHan100% (1)
- PRC - Aircraft PowerplantDocument24 pagesPRC - Aircraft PowerplantMommyPokaNo ratings yet
- IC Presentation1Document2 pagesIC Presentation1Angielou Sialana100% (6)
- B1, Timber Beam Analysis & Design (AS1720)Document3 pagesB1, Timber Beam Analysis & Design (AS1720)Saiful IslamNo ratings yet
- Rocket and Missiles Important Question PaperDocument3 pagesRocket and Missiles Important Question PaperSundaraPandi100% (3)
- Classroom Activities in Aerodynamics: © 2000 Dr. Charley Rodriguez, PH.DDocument21 pagesClassroom Activities in Aerodynamics: © 2000 Dr. Charley Rodriguez, PH.DAerocse EceitmechNo ratings yet
- EASA Mod 15 BK 9 Heli EngsDocument45 pagesEASA Mod 15 BK 9 Heli EngsaviNo ratings yet
- Cooling Water Pump Data Sheet FinalDocument8 pagesCooling Water Pump Data Sheet FinalShahzaibUsmanNo ratings yet
- Propeller FundamentalDocument7 pagesPropeller FundamentalSharath UpadhyaNo ratings yet
- Radial TurbinesDocument46 pagesRadial TurbinesarorabbNo ratings yet
- Fan Dry Coil UnitDocument5 pagesFan Dry Coil UnitHiei ArshavinNo ratings yet
- E5 Lab Sheet Measurement of Jet ForcesDocument9 pagesE5 Lab Sheet Measurement of Jet ForcesHoongNo ratings yet
- YhhjjDocument52 pagesYhhjjSam CunananNo ratings yet
- Distillation Control TheoryDocument12 pagesDistillation Control Theoryinstrutech0% (1)
- 08rr410306 Jet Propulsion and Rocket EngineeringDocument6 pages08rr410306 Jet Propulsion and Rocket EngineeringsmijusNo ratings yet
- Reviewer 5Document59 pagesReviewer 5KleberNo ratings yet
- How To Disassemble Jet EnginesDocument1 pageHow To Disassemble Jet Enginesmamo santosoNo ratings yet
- Incompressible Flow Over Finite Wings-IntroDocument11 pagesIncompressible Flow Over Finite Wings-IntroJanuarSianiparNo ratings yet
- Propulsion and Fuel System IntegrationDocument36 pagesPropulsion and Fuel System IntegrationNouman Nadeem0% (1)
- Total Question OutDocument31 pagesTotal Question OutJoe Ho100% (2)
- AEMR-Unit - II & III, 2 Marks & 16 Marks Questions With Anwers-IV Yr AeroDocument39 pagesAEMR-Unit - II & III, 2 Marks & 16 Marks Questions With Anwers-IV Yr AeroRAJASUDHAKAR SNo ratings yet
- AE221 - Aerodynamics Xfoil Project (SC18B096) PDFDocument21 pagesAE221 - Aerodynamics Xfoil Project (SC18B096) PDFRahul Muvvala100% (1)
- Unit V - Rocket PropulsionDocument5 pagesUnit V - Rocket PropulsionKeerthi VarmanNo ratings yet
- Experimental Stress AnalysisDocument194 pagesExperimental Stress AnalysisJEEVA KUMAR M100% (1)
- Chapter7 Lecture 24-26performance Analysis III - Range and EnduranceDocument10 pagesChapter7 Lecture 24-26performance Analysis III - Range and EnduranceChegrani AhmedNo ratings yet
- IC EnginesDocument14 pagesIC EnginesPradeep N B100% (1)
- Aerodynamics II Unit 3 Notes (Part 1Document14 pagesAerodynamics II Unit 3 Notes (Part 1anirudh pNo ratings yet
- Wind Tunnel Pope PDFDocument20 pagesWind Tunnel Pope PDFKrmxl Tdre100% (1)
- Module 3 IC EnginesDocument70 pagesModule 3 IC EnginesAnkitNo ratings yet
- Lecture Notes Aircraft Navigation (Lecture 1,2)Document9 pagesLecture Notes Aircraft Navigation (Lecture 1,2)VIRAJ HADKARNo ratings yet
- Module 17 Propellers Test AnswerDocument4 pagesModule 17 Propellers Test AnswercamapnhiNo ratings yet
- Aircraft Pneumatic SystemsDocument46 pagesAircraft Pneumatic SystemsSasiKumar100% (1)
- Aero Engine Maintenance and RepairDocument21 pagesAero Engine Maintenance and RepairDurai Raj KumarNo ratings yet
- AE8302-Elements of Aeronautical Engineering: Reg. NoDocument2 pagesAE8302-Elements of Aeronautical Engineering: Reg. NoViswanath ViswaNo ratings yet
- Industrial AerodynamicsDocument78 pagesIndustrial AerodynamicsAnuNo ratings yet
- AS1 FormatDocument97 pagesAS1 FormatgurusamyNo ratings yet
- Neral Capítulo.1 PDFDocument8 pagesNeral Capítulo.1 PDFemhidalgoNo ratings yet
- Thrust AugmentationDocument16 pagesThrust AugmentationSenthamil Arasan0% (1)
- Jawaban PropulsiDocument35 pagesJawaban PropulsiyandaNo ratings yet
- Chapter 4 - Airfoil and Geometry SelectionDocument75 pagesChapter 4 - Airfoil and Geometry SelectionSaad MasoodNo ratings yet
- Aerodynamic Performance Analysis of A Non Planar C-Wing Using Experimental and Numerical ToolsDocument28 pagesAerodynamic Performance Analysis of A Non Planar C-Wing Using Experimental and Numerical ToolsGiang Nguyễn TrườngNo ratings yet
- Industrial AerodynamicsDocument32 pagesIndustrial AerodynamicsSenthamil ArasanNo ratings yet
- Main Practice 1Document7 pagesMain Practice 1sureshrnalNo ratings yet
- AF Structure ContDocument21 pagesAF Structure ContlongmilkNo ratings yet
- Flight Dynamics 2 Marks by Nambi RajanDocument4 pagesFlight Dynamics 2 Marks by Nambi RajanNambi RajanNo ratings yet
- Rr312102 Aircraft PerformanceDocument8 pagesRr312102 Aircraft PerformanceSrinivasa Rao GNo ratings yet
- Types of Wind TunnelsDocument3 pagesTypes of Wind TunnelsAfaq0% (1)
- Experimental Fluid Mechanics: The Commonwealth and International Library: Thermodynamics and Fluid Mechanics DivisionFrom EverandExperimental Fluid Mechanics: The Commonwealth and International Library: Thermodynamics and Fluid Mechanics DivisionRating: 4 out of 5 stars4/5 (2)
- Nptel - Ac.in Courses IIT-MADRAS Applied Thermodynamics Module 4 6 AsdcDocument4 pagesNptel - Ac.in Courses IIT-MADRAS Applied Thermodynamics Module 4 6 AsdcSanjeev PrakashNo ratings yet
- Limited Pressure CycleDocument4 pagesLimited Pressure Cyclecasu19No ratings yet
- Stirling CycleDocument3 pagesStirling CycleRon QuerubinNo ratings yet
- Stirling CycleDocument6 pagesStirling CycleChristian_Joe__9147No ratings yet
- 2 Carnot CycleDocument6 pages2 Carnot CycleSamujawal Kumar SumanNo ratings yet
- IC Engines 2012 Edition Theory & QuestionsDocument178 pagesIC Engines 2012 Edition Theory & Questionskumarrohit91No ratings yet
- Pagine Per L'Insegnante: Idee Per Una Lezione DigitaleDocument6 pagesPagine Per L'Insegnante: Idee Per Una Lezione DigitaleVasile TurtaNo ratings yet
- CFD Exam PaperDocument2 pagesCFD Exam PaperAbhishek Awasthi100% (1)
- Fluid Properties ME322 (Semester 372) - 1Document23 pagesFluid Properties ME322 (Semester 372) - 1AhmadNo ratings yet
- Les Équations de Maxwell Sont Elles Complètes - Germain - Rousseau - 2001Document9 pagesLes Équations de Maxwell Sont Elles Complètes - Germain - Rousseau - 2001Margot MA TAUPINNo ratings yet
- Module 3 - Flexural Strength in Beams (WSD & Cracked Sections - Tansformed Section Method)Document5 pagesModule 3 - Flexural Strength in Beams (WSD & Cracked Sections - Tansformed Section Method)Mika Vernadeth SingNo ratings yet
- Properties and Microstructures of Lightweight Alumina Containing Different Types of Nano-AluminaDocument46 pagesProperties and Microstructures of Lightweight Alumina Containing Different Types of Nano-AluminaIury VazNo ratings yet
- Phase Rule Water & CO2systemsDocument9 pagesPhase Rule Water & CO2systemsAtul GautamNo ratings yet
- 10 1002@biot 201600054Document36 pages10 1002@biot 201600054ANZWAYNENo ratings yet
- Material - S45C NormalizedDocument1 pageMaterial - S45C NormalizedhanazahrNo ratings yet
- Lecture 6 Notes - Advanced Arch Dams Part 2Document10 pagesLecture 6 Notes - Advanced Arch Dams Part 2Chalang AkramNo ratings yet
- Lug Analysis - MechaniCalcDocument31 pagesLug Analysis - MechaniCalcmuhammad hadiNo ratings yet
- Fluid Mechanic.151 200Document50 pagesFluid Mechanic.151 200Shanmuga Ramanan100% (2)
- TSM Ch0 ContentsDocument10 pagesTSM Ch0 ContentsddyzleeNo ratings yet
- Tpe PDFDocument6 pagesTpe PDFChourouk MeskiniNo ratings yet
- Shwan Unsat18 118Document7 pagesShwan Unsat18 118Amir BakhtiarNo ratings yet
- Problem Sheet - 3 PDFDocument2 pagesProblem Sheet - 3 PDFJyotirmoy DekaNo ratings yet
- Researchpaper Waffle Slab Analysis by Different MethodsDocument4 pagesResearchpaper Waffle Slab Analysis by Different MethodsYasser AlghrafyNo ratings yet
- Entropy and ProbabilityDocument8 pagesEntropy and ProbabilityFer MoncadaNo ratings yet
- Turbulence Modeling SlideDocument25 pagesTurbulence Modeling SlideNguyenThanhVanNo ratings yet
- Mechanical Measurements PDFDocument3 pagesMechanical Measurements PDFSv SaisudheerNo ratings yet
- TCE E Consul Lting Eng Gineers L Limited: Design Nofstr AS Ructural Per Is: 8 L Steel E 00-2007 Element TSDocument1 pageTCE E Consul Lting Eng Gineers L Limited: Design Nofstr AS Ructural Per Is: 8 L Steel E 00-2007 Element TSNeeraj DubeyNo ratings yet
- Natural VentilationDocument34 pagesNatural VentilationHarshitBabooNo ratings yet