Professional Documents
Culture Documents
Bchm2000 PX
Bchm2000 PX
Uploaded by
Roger Jayson MercadoOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Bchm2000 PX
Bchm2000 PX
Uploaded by
Roger Jayson MercadoCopyright:
Available Formats
Biochemistry 2000
(1)
Sample Questions x
Kinetic graphs
For an enzyme (5 M) , the following initial velocities have been reported
depending on the substrate concentration:
v0, mM s-1
10.83
18.57
26.76
32.50
39.00
43.33
48.75
54.17
56.88
[Substrate], mM
0.02
0.04
0.07
0.1
0.15
0.2
0.3
0.5
0.7
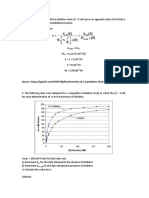
(a) Draw a Michaelis-Menten plot for this enzyme.
(b) Draw a Lineweaver-Burke plot for this enzyme.
(c) Determine KM and vmax for this enzyme
(d) Indicate in both graphs (a & b) where vmax and KM can be recognized.
(e) Calculate the catalytic constant and the catalytic efficiency for this
enzyme.
(2)
You are trying to reproduce experimental data from the previous student in
the lab. He/she had reported that the enzyme under investigation has a
kcat of 1875 s-1 and a catalytic efficiency of 7.5 107 M-1 s-1.
(a) What is the KM of this enzyme?
(b) In repeating the measurement, you want to have a vmax of 10 mM s-1
(as this is easy to measure). How much enzyme should you use?
Provide your result in M.
(c) Calculate at which substrate concentrations in M you should measure
the initial rate v0 to obtain the following values:
v0, mM s-1
[Substrate], M
2.50
4.50
6.00
9.00
(d) Create a predicted Michaelis-Menten plot for your enzyme.
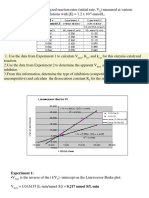
(e) Draw a predicted Lineweaver-Burke plot for your enzyme.
Biochemistry 2000
Sample Questions x
Kinetic graphs
Answers:
(1)
(a)
70
vmax
v0, mM/s
60
50
40
30
20
10
0
0.0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9
KM
[Substrate], mM
(b)
0.10
-1
1/v0, mM s
0.08
0.06
0.04
1/vmax
0.02
-1/KM
-20
-10
10
20
30
40
50
60
1/[Substrate], mM-1
(c)
KM and vmax can be most precisely determined from the intercepts in the
Lineweaver-Burke plot:
1/vmax = 0.015385 mM-1 s vmax = 65 mM s-1
-1/KM = -10 mM-1
KM = 0.1 mM
(d)
kcat = vmax / [E]total = 65 mM s-1 / 5 M = 65 mM s-1 / 0.005 mM = 13000 s-1
catalytic efficiency: kcat/KM = 13000 s-1 / 0.1 mM = 13000 s-1 / 0.0001 M =
1.3 108 M s-1
Biochemistry 2000
Sample Questions x
Kinetic graphs
vmax
25
KM
50
75 100 125 150 175 200 225 250
[Substrate], M
(e)
1/[Substrate],
M-1
0.1200
0.0489
0.0267
0.0044
1/v0, mM-1 s
0.4000
0.2222
0.1667
0.1111
0.45
0.40
0.35
0.30
-1
11
10
9
8
7
6
5
4
3
2
1
0
0
1/v0, mM s
v0, mM/s
(2) (a) From kcat/KM and kcat, you can calculate KM as
KM = 1/(kcat/KM) * kcat = KM/kcat * kcat = 1/7.5 107 M s-1* 1875 s-1 = 2.5 10-5 M
= 0.025 mM = 25 M
(b)
From kcat and vmax, you can calculate [E]total
vmax / [E]total = kcat
[E]total = vmax/kcat = 10 mM s-1 / 1875 s-1 = 0.00533 mM = 5.33 M
(c)
Rearranging the Michaelis-Menten equation: [S] = v0 * KM / (vmax v0)
Inserting the given values for KM, vmax and v0 will give you [S] in mM.
Mulitplying by a factor of 1000 converts [S] in M.
v0, mM s-1
[Substrate], M
8.333333
2.50
20.45455
4.50
37.5
6.00
225
9.00
(d)
0.25
0.20
0.15
1/vmax
0.10
0.05
-1/KM
-0.04 -0.02
0.00
0.02
0.04
0.06
0.08
-1
1/[Substrate], M
0.10
0.12
0.14
You might also like
- A.1. Competitive InhibitionDocument5 pagesA.1. Competitive InhibitionFlorecita Cabañog100% (2)
- Enzyme KineticsDocument3 pagesEnzyme KineticsZeny Naranjo100% (2)
- 3Document4 pages3biotech_vidhya100% (1)
- BIO307 Lecture 5 (Enzyme Kinetics I)Document11 pagesBIO307 Lecture 5 (Enzyme Kinetics I)Phenyo Mmereki100% (1)
- Test Blue Print in BiochemistryDocument3 pagesTest Blue Print in BiochemistryJoherNo ratings yet
- 7-Bioc431 Enzymes Kinetics S15Document34 pages7-Bioc431 Enzymes Kinetics S15Shariq Mansoor KhanNo ratings yet
- Name: Kristine Joy Atos Block: BSN 1-D Practice ProblemsDocument6 pagesName: Kristine Joy Atos Block: BSN 1-D Practice ProblemsJenz Hope Segui Novela100% (3)
- Enzyme InhibitionDocument7 pagesEnzyme InhibitionJane Docdoc100% (1)
- Problem Set - Enzymes From LehningerDocument11 pagesProblem Set - Enzymes From LehningervioletbrownNo ratings yet
- ENZYMES Problems and SolutionsDocument6 pagesENZYMES Problems and SolutionsChadby GraNaNo50% (2)
- Assign#2 Enzyme KineticsDocument3 pagesAssign#2 Enzyme KineticsKaren Castro100% (2)
- 5 Enzyme Kinetics-InhibitionDocument40 pages5 Enzyme Kinetics-InhibitionJoel SmolanoffNo ratings yet
- Sample ProblemDocument1 pageSample ProblemfintastellaNo ratings yet
- EnzymeKinetics by P.C. Misra Professor, Department of Biochemistry Lucknow University, Lucknow-226 007Document21 pagesEnzymeKinetics by P.C. Misra Professor, Department of Biochemistry Lucknow University, Lucknow-226 007Dr. SHIVA AITHALNo ratings yet
- Lecture Notes Enzyme 2 Enzyme Kinetics WebDocument29 pagesLecture Notes Enzyme 2 Enzyme Kinetics WebAldren RebaLdeNo ratings yet
- MIT Lecture 6 5.07 Biochemistry LectureDocument16 pagesMIT Lecture 6 5.07 Biochemistry LectureakiridoNo ratings yet
- Voet - Chapt - 12 Properties of EnzymesDocument102 pagesVoet - Chapt - 12 Properties of Enzymestelmo flowNo ratings yet
- Kinetic Vs Chemical MechanismDocument34 pagesKinetic Vs Chemical MechanismIgnacio Bascuñán OyarceNo ratings yet
- Problem Set On Enzyme Kinetics - FS - 2012 - 2013Document2 pagesProblem Set On Enzyme Kinetics - FS - 2012 - 2013Chay Alcantara100% (1)
- Enzyme KineticsDocument72 pagesEnzyme Kineticsitokki otoya100% (1)
- Excel 2007 TutorialDocument5 pagesExcel 2007 TutorialromelcarvajalNo ratings yet
- Lecture 17-Multisubstrate Enzyme RXN KineticsDocument13 pagesLecture 17-Multisubstrate Enzyme RXN KineticsAmogha G C 1SI19CH002No ratings yet
- Sample Exam 4 Fall 11Document14 pagesSample Exam 4 Fall 11janohxNo ratings yet
- Enzyme Kinetics Examples and ProblemsDocument4 pagesEnzyme Kinetics Examples and Problemskiiadizon07100% (1)
- Chem 40 Enzyme KineticsDocument85 pagesChem 40 Enzyme KineticsJustine Grace Mariano100% (1)
- Question CH06+answer PDFDocument8 pagesQuestion CH06+answer PDFCris-Anne Juangco III100% (1)
- Chapter 2 - Basics of EnzymesDocument40 pagesChapter 2 - Basics of EnzymesSakinah MuhamadNo ratings yet
- Buffer SolutionDocument14 pagesBuffer SolutionSony0% (1)
- Chapter 10 Chemical Kinetics IIDocument131 pagesChapter 10 Chemical Kinetics IIChicken ChickenNo ratings yet
- Study GuideDocument3 pagesStudy GuideSam Khader100% (1)
- Equations in Enzyme KineticsDocument29 pagesEquations in Enzyme KineticsAndrew Sam100% (2)
- Enzyme Kinetics Questions and Answers: 2 Year Undergraduates-Biology 2018-2019Document8 pagesEnzyme Kinetics Questions and Answers: 2 Year Undergraduates-Biology 2018-2019Emmanuel JoyNo ratings yet
- A.2. Noncompetitive InhibitionDocument6 pagesA.2. Noncompetitive InhibitionFlorecita CabañogNo ratings yet
- Nzymes: By: Mrs. Kalaivani Sathish. M. Pharm, Assistant Professor, Pims - PanipatDocument63 pagesNzymes: By: Mrs. Kalaivani Sathish. M. Pharm, Assistant Professor, Pims - Panipaturmila pandeyNo ratings yet
- Sutherland 1991Document7 pagesSutherland 1991Isal AbdussalamNo ratings yet
- Optimization of Cellulase Enzyme From Vegetable Waste by Using Trichoderma Atroviride in Solid State FermentationDocument6 pagesOptimization of Cellulase Enzyme From Vegetable Waste by Using Trichoderma Atroviride in Solid State FermentationIOSRjournalNo ratings yet
- Che503 PS3 PDFDocument2 pagesChe503 PS3 PDFCarissa TejioNo ratings yet
- Multiple Choice Questions: 1. GlycolysisDocument4 pagesMultiple Choice Questions: 1. GlycolysisNguyễn TânNo ratings yet
- 2-Bacterial Growth Kinetics - F11Document15 pages2-Bacterial Growth Kinetics - F11Suvidha Chib100% (1)
- CH 3 - ProblemsDocument6 pagesCH 3 - ProblemsKhris Griffis100% (5)
- Biochemistry - Final Exam Biology 020.305 December 15, 2011 Instructions For ExaminationDocument11 pagesBiochemistry - Final Exam Biology 020.305 December 15, 2011 Instructions For ExaminationJenna ReynoldsNo ratings yet
- BCH 314 Tutorial 1Document9 pagesBCH 314 Tutorial 1victorNo ratings yet
- Ex 4 Effect of PH On Enzyme ActivityDocument2 pagesEx 4 Effect of PH On Enzyme ActivityRochel CaduyacNo ratings yet
- Chapter 8 Lecture-Energy, Enzymes, and Metabolism-MODIFIED2Document49 pagesChapter 8 Lecture-Energy, Enzymes, and Metabolism-MODIFIED2E'Lasia LarkinNo ratings yet
- BIOC 307 Old Exam 1Document17 pagesBIOC 307 Old Exam 1Katie RoseNo ratings yet
- Amylase Assay 2Document9 pagesAmylase Assay 2Rahman ImudaNo ratings yet
- HOW TO - Calculate KM and Vmax With ExcelDocument3 pagesHOW TO - Calculate KM and Vmax With Excelminjeshp67% (3)
- Michaelis Menten EquationDocument9 pagesMichaelis Menten Equationsadaf zaidiNo ratings yet
- 4 Bioenergetics and Oxidative Metabolism IIDocument3 pages4 Bioenergetics and Oxidative Metabolism IILinus LiuNo ratings yet
- CH 13Document32 pagesCH 13Dhei DheitNo ratings yet
- Vietnam National University Ho Chi Minh International UniversityDocument5 pagesVietnam National University Ho Chi Minh International UniversityHuỳnh Như100% (1)
- CH 8 - ProblemsDocument4 pagesCH 8 - ProblemsKhris GriffisNo ratings yet
- Kinetics OverviewDocument168 pagesKinetics OverviewAlbert Daniel GarciaNo ratings yet
- The Influence of Conformational Isomerism On Drug ActionDocument6 pagesThe Influence of Conformational Isomerism On Drug Actionhectorlope45No ratings yet
- 15 Chem KinetDocument51 pages15 Chem KinetRoua Ali100% (2)
- Quantitative Human Physiology: An IntroductionFrom EverandQuantitative Human Physiology: An IntroductionRating: 2 out of 5 stars2/5 (1)
- Selected Topics in the History of Biochemistry. Personal Recollections. Part IIIFrom EverandSelected Topics in the History of Biochemistry. Personal Recollections. Part IIIRating: 1 out of 5 stars1/5 (1)