Professional Documents
Culture Documents
Chemical Periodicity (Period 3)
Chemical Periodicity (Period 3)
Uploaded by
Subaashini0 ratings0% found this document useful (0 votes)
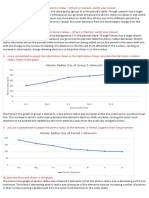
10 views1 pageAtomic size decreases, first ionization energy increases, and electronegativity increases across Period 3 from sodium to argon. Melting point increases from sodium to aluminum due to stronger metallic bonding. Electrical conductivity also increases from sodium to aluminum due to better conduction of valence electrons. The melting point of silicon is highest due to its small size and strong covalent bonding. Phosphorus has a lower melting point than silicon and sulfur because it has a smaller size and weaker van der Waals forces. Chlorine has a higher boiling point than argon but lower than sulfur due to their relative molecular sizes and polarizabilities. Neither silicon nor sulfur conduct electricity as they typically form covalent, molecular structures.
Original Description:
bm
Original Title
10
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentAtomic size decreases, first ionization energy increases, and electronegativity increases across Period 3 from sodium to argon. Melting point increases from sodium to aluminum due to stronger metallic bonding. Electrical conductivity also increases from sodium to aluminum due to better conduction of valence electrons. The melting point of silicon is highest due to its small size and strong covalent bonding. Phosphorus has a lower melting point than silicon and sulfur because it has a smaller size and weaker van der Waals forces. Chlorine has a higher boiling point than argon but lower than sulfur due to their relative molecular sizes and polarizabilities. Neither silicon nor sulfur conduct electricity as they typically form covalent, molecular structures.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
10 views1 pageChemical Periodicity (Period 3)
Chemical Periodicity (Period 3)
Uploaded by
SubaashiniAtomic size decreases, first ionization energy increases, and electronegativity increases across Period 3 from sodium to argon. Melting point increases from sodium to aluminum due to stronger metallic bonding. Electrical conductivity also increases from sodium to aluminum due to better conduction of valence electrons. The melting point of silicon is highest due to its small size and strong covalent bonding. Phosphorus has a lower melting point than silicon and sulfur because it has a smaller size and weaker van der Waals forces. Chlorine has a higher boiling point than argon but lower than sulfur due to their relative molecular sizes and polarizabilities. Neither silicon nor sulfur conduct electricity as they typically form covalent, molecular structures.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 1
Chemical Periodicity (Period 3):
1. State and explain the following trends:
a) atomic size across Period 3
b) first ionisation energy across Period 3
c) electronegativity across Period 3
d) melting point from sodium to aluminium
e) electrical conductivity from sodium to aluminium
2. Explain the following:
a) the melting point of silicon is the highest in Period 3
b) the melting point of phosphorus is lower than silicon
c) the melting point of phosphorus is lower than that of sulphur
d) the boiling point of chlorine is greater than that of argon, but lower than that of
sulphur
e) neither silicon nor sulphur conduct electricity
You might also like
- General Chemistry Principles and Modern Applications 11Th Edition Petrucci Solutions Manual Full Chapter PDFDocument52 pagesGeneral Chemistry Principles and Modern Applications 11Th Edition Petrucci Solutions Manual Full Chapter PDFDebraPricemkw100% (13)
- Francis X. McConville - The Pilot Plant Real Book A Unique Handbook For The Chemical Process Industry (2002, FXM Engineering and Design) PDFDocument309 pagesFrancis X. McConville - The Pilot Plant Real Book A Unique Handbook For The Chemical Process Industry (2002, FXM Engineering and Design) PDFHaziq100% (3)
- Practice Test (Dolgos) - Periodic Table - W KeyDocument7 pagesPractice Test (Dolgos) - Periodic Table - W Keychandro57100% (1)
- D Block ElementsDocument22 pagesD Block Elementsketan kambleNo ratings yet
- CH 17 w16 PDFDocument53 pagesCH 17 w16 PDFrachel lorenNo ratings yet
- Periodicity - Practice 19 Sept PAPERDocument4 pagesPeriodicity - Practice 19 Sept PAPEREvandra Ramadhan Azriel SanyotoNo ratings yet
- TEST 2 Topic 3Document3 pagesTEST 2 Topic 3Ashutosh SharmaNo ratings yet
- A. Li + BR B. Li + CL C. K + BR D. K + CL (Total 1 Mark)Document21 pagesA. Li + BR B. Li + CL C. K + BR D. K + CL (Total 1 Mark)raja_tanukuNo ratings yet
- Japanese Reports Electrical Fire CausesDocument55 pagesJapanese Reports Electrical Fire Causesal bauerNo ratings yet
- IB SL Topic (3) Periodicity Review QuestionsDocument7 pagesIB SL Topic (3) Periodicity Review Questionsbindu shineNo ratings yet
- F23 Topic 3 Periodicity QuizCDocument6 pagesF23 Topic 3 Periodicity QuizCNiambi WillsNo ratings yet
- Miscellaneous ElementsDocument314 pagesMiscellaneous ElementsOnline EducatorNo ratings yet
- IB Chemistry SL - Chapter 3 Review QuestionsDocument4 pagesIB Chemistry SL - Chapter 3 Review Questionsshawnfi jusrmk100% (1)
- Forces of Attraction - TutorialDocument3 pagesForces of Attraction - TutorialMel ManningNo ratings yet
- 2 QP ElectrochemistryDocument6 pages2 QP ElectrochemistrysachinNo ratings yet
- Chemistry Questions: Structure of AtomDocument61 pagesChemistry Questions: Structure of AtomladkibadianjanihaiNo ratings yet
- Material Dec14 FinalDocument1 pageMaterial Dec14 FinalRajesh PathakNo ratings yet
- D and F Block PYQ FOR 2024 CLASS 12 EXAMS BY Gtctuition For YtDocument34 pagesD and F Block PYQ FOR 2024 CLASS 12 EXAMS BY Gtctuition For YtDr SaabNo ratings yet
- JEE Main Physics Previous Year Questions With Solutions On SemiconductorsDocument4 pagesJEE Main Physics Previous Year Questions With Solutions On SemiconductorsConimicNo ratings yet
- CH205 Test 1 2021Document6 pagesCH205 Test 1 2021avnishnandNo ratings yet
- Roots Classes: Physics TestDocument3 pagesRoots Classes: Physics TestAnupam DubeyNo ratings yet
- AA Chem CW (2nd Term) (9) 2nd - InddDocument3 pagesAA Chem CW (2nd Term) (9) 2nd - InddTing TCNo ratings yet
- STD XI Question Bank Quarterly 2021Document3 pagesSTD XI Question Bank Quarterly 2021Shriya RameshNo ratings yet
- Pioneer Junior College Higher 2 Chemistry (9647) Inorganic Chemistry The Periodic Table: Chemical PeriodicityDocument22 pagesPioneer Junior College Higher 2 Chemistry (9647) Inorganic Chemistry The Periodic Table: Chemical PeriodicityTimothy HandokoNo ratings yet
- H2 Inorganic ChemistryDocument7 pagesH2 Inorganic ChemistrykitoniumNo ratings yet
- Atomic Radius and Electronegativity - QuestionsDocument4 pagesAtomic Radius and Electronegativity - QuestionsAnonymous Mj0pfScNo ratings yet
- Che Chapter 10 - PeriodictyDocument9 pagesChe Chapter 10 - PeriodictylisaNo ratings yet
- 5.periodicity - AnswersDocument7 pages5.periodicity - AnswersAnshu MovvaNo ratings yet
- Quiz IDocument1 pageQuiz ISurya RNo ratings yet
- TAQ 1-6 PaperDocument21 pagesTAQ 1-6 PaperDavid MuneneNo ratings yet
- Cu OverviewDocument38 pagesCu OverviewDejan DjokićNo ratings yet
- T.C. Forensic - JAPANESE REPORTS ON ELECTRICAL FIRE CAUSES - Abstract 3Document5 pagesT.C. Forensic - JAPANESE REPORTS ON ELECTRICAL FIRE CAUSES - Abstract 3ecordova1No ratings yet
- Atomic Structure TutorialDocument3 pagesAtomic Structure Tutorialchong56No ratings yet
- DPP - 8Document2 pagesDPP - 8Neha AttriNo ratings yet
- Answers by K-SEPTEMBER TEST 2023 - 1Document4 pagesAnswers by K-SEPTEMBER TEST 2023 - 1bikramjitgujjarNo ratings yet
- Mushili Science 9a2?Document3 pagesMushili Science 9a2?Sam Chibuye JnrNo ratings yet
- Tutorial N°2Document6 pagesTutorial N°2isam-eddine.babouriNo ratings yet
- Eletrical McqsDocument7 pagesEletrical McqsPravin Hande100% (1)
- Ana ChemDocument14 pagesAna ChemSamuel MagannonNo ratings yet
- Unit 6 Practice Test (Periodic Table)Document5 pagesUnit 6 Practice Test (Periodic Table)Xazerco LaxNo ratings yet
- Chem11 SM 1 7Document2 pagesChem11 SM 1 7dudoocandrawNo ratings yet
- EndSem MM454-Apr2014 Soln PDFDocument6 pagesEndSem MM454-Apr2014 Soln PDFPrakash ChandraNo ratings yet
- Answer Sheet of Chemistry HW 10th Class From Periodic Table PDFDocument3 pagesAnswer Sheet of Chemistry HW 10th Class From Periodic Table PDFAbdul Sami MomandNo ratings yet
- Important MCQ - Materials ScienceDocument13 pagesImportant MCQ - Materials SciencearjitlgspNo ratings yet
- Topic 1: Atomic Structure and The Periodic Table - Part 1Document4 pagesTopic 1: Atomic Structure and The Periodic Table - Part 1MohamudNo ratings yet
- Tetreahedron Educlasses: One MarksDocument2 pagesTetreahedron Educlasses: One MarkssadhuNo ratings yet
- CHE 123 - Worksheet - Atomic Structure - IonizationDocument6 pagesCHE 123 - Worksheet - Atomic Structure - IonizationJanet UsherNo ratings yet
- Test Yourself: - 1 A B C DDocument4 pagesTest Yourself: - 1 A B C Dtess_15No ratings yet
- Examples of Multiple Choice QuestionsDocument3 pagesExamples of Multiple Choice QuestionsBryan NozaledaNo ratings yet
- Engineering Failure Analysis: Nam-Hyuck Lee, Sin Kim, Byung-Hak Choe, Kee-Bong Yoon, Dong-Il KwonDocument5 pagesEngineering Failure Analysis: Nam-Hyuck Lee, Sin Kim, Byung-Hak Choe, Kee-Bong Yoon, Dong-Il KwonAnand VarmaNo ratings yet
- Atomic Structure QuestionsDocument4 pagesAtomic Structure QuestionsHovan Tall Nut TanNo ratings yet
- Inorg RevDocument4 pagesInorg RevMarvin JeaNo ratings yet
- 11th Chemistry Final TermDocument8 pages11th Chemistry Final TermpallavichandraNo ratings yet
- DiffusionDocument2 pagesDiffusionKrishna Kumar MishraNo ratings yet
- Assignment 1 - CSE StreamDocument1 pageAssignment 1 - CSE Streammonkeydluffy0505kaizokuNo ratings yet
- 9th Class (Test Chemistry Chapter 2)Document3 pages9th Class (Test Chemistry Chapter 2)waqasNo ratings yet
- Period 3 ElementsDocument36 pagesPeriod 3 ElementsTichafara Paul ShumbaNo ratings yet
- Test Chem 9Document3 pagesTest Chem 9ZaryabRidNo ratings yet
- Tanapp Tcm18-410hurDocument5 pagesTanapp Tcm18-410hurNguh FabriceNo ratings yet
- 4 5998822681074471515Document9 pages4 5998822681074471515Siif siin KajemsNo ratings yet
- DiffusionDocument5 pagesDiffusionCatalin SfatNo ratings yet
- Metallurgical Achievements: Selection of Papers Presented at the Birmingham Metallurgical Society's Diamond Jubilee Session, 1963-1964From EverandMetallurgical Achievements: Selection of Papers Presented at the Birmingham Metallurgical Society's Diamond Jubilee Session, 1963-1964W. O. AlexanderNo ratings yet
- Waste ManagementDocument6 pagesWaste ManagementpraveenitplNo ratings yet
- Pre-Lab ReportDocument6 pagesPre-Lab ReportTintin Brusola SalenNo ratings yet
- 4W1570 Final ReportDocument44 pages4W1570 Final ReportKhurram ShahzadNo ratings yet
- Problem Set 1 - CH148Document11 pagesProblem Set 1 - CH148Allyssa BadilloNo ratings yet
- Cement Process PDFDocument36 pagesCement Process PDFAstha Yadav100% (3)
- Luff SchoorlDocument13 pagesLuff Schoorlmarijuana.hesNo ratings yet
- CTM400 - 19 - 02JUN05 Rep 9 0Document420 pagesCTM400 - 19 - 02JUN05 Rep 9 0Antony Moreno100% (4)
- Kastolite 23 LI DataDocument2 pagesKastolite 23 LI DataAjit YadavNo ratings yet
- Chemcure - 15Document2 pagesChemcure - 15ICPL-RWPNo ratings yet
- AIChE 2011 NSDC Problem StatementDocument16 pagesAIChE 2011 NSDC Problem Statementcmm4671No ratings yet
- Jacketed Vessel DesignDocument5 pagesJacketed Vessel DesignfunkkkkyNo ratings yet
- Patrick Lee - Fermions and Spin LiquidDocument58 pagesPatrick Lee - Fermions and Spin LiquidKonnasderNo ratings yet
- Plastics and Insulating MaterialsDocument1 pagePlastics and Insulating MaterialsAngel GutierrezNo ratings yet
- Manual Inquiry enDocument37 pagesManual Inquiry enDancescu AlexandruNo ratings yet
- GUJCET2016MT02 SolutionDocument14 pagesGUJCET2016MT02 SolutionvuppalasampathNo ratings yet
- MSDS-CSP e - 4038 Acri 700 FinishDocument5 pagesMSDS-CSP e - 4038 Acri 700 FinishAnkit VyasNo ratings yet
- Laboratory ReportDocument5 pagesLaboratory ReportRicanie CadornaNo ratings yet
- DGA Comparison Between Ester and Mineral Oils: C. Perrier, M. Marugan, M. Saravolac A. BeroualDocument4 pagesDGA Comparison Between Ester and Mineral Oils: C. Perrier, M. Marugan, M. Saravolac A. BeroualJhon Fredy Vallejo GiraldoNo ratings yet
- Welding MachineDocument3 pagesWelding MachinetuanNo ratings yet
- CO2 Fire Extinguishing SystemDocument4 pagesCO2 Fire Extinguishing Systemsymyaurel0% (1)
- Azas-Azas Umum Toksikologi (Nasib Racun Dalam Tubuh)Document27 pagesAzas-Azas Umum Toksikologi (Nasib Racun Dalam Tubuh)Nitasolikhah0703No ratings yet
- Tugas Bahasa Inggris: Faturrachman Rengga Wisnu Resza Bunga NisaDocument4 pagesTugas Bahasa Inggris: Faturrachman Rengga Wisnu Resza Bunga NisaFaturrachman Nanu Rengga WisnuNo ratings yet
- Effect of Boron and Carbon Addition On Microstructure and Mec 2015 MaterialsDocument14 pagesEffect of Boron and Carbon Addition On Microstructure and Mec 2015 Materialsnarayananx5No ratings yet
- 2022L - Revest de Resistência À Erosão Por Cavitação Usando Aço Inox e Ligas de Cobalto Depositados Por Gmaw e CW GmawDocument14 pages2022L - Revest de Resistência À Erosão Por Cavitação Usando Aço Inox e Ligas de Cobalto Depositados Por Gmaw e CW GmawIgor Alexsander Barbosa MagnoNo ratings yet
- Crystallization of Paraxylene With Scraped Surface Heat ExchangersDocument192 pagesCrystallization of Paraxylene With Scraped Surface Heat ExchangersJorge Gutierrez AranibarNo ratings yet
- Structure of AtomDocument26 pagesStructure of AtomsyNo ratings yet
- Silver-Brite Hi-Heat: Protective & Marine CoatingsDocument4 pagesSilver-Brite Hi-Heat: Protective & Marine CoatingsAida LinaresNo ratings yet
- High Tensile C Purlin: Steel Grade Equivalent To ASTM 446 Grade DDocument2 pagesHigh Tensile C Purlin: Steel Grade Equivalent To ASTM 446 Grade DRodolfo ZazuetaNo ratings yet